
Biochemistry: Concepts and Connections (2nd Edition)
2nd Edition
ISBN: 9780134641621
Author: Dean R. Appling, Spencer J. Anthony-Cahill, Christopher K. Mathews
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14, Problem 3P
Freshly prepared mitochondria were incubated with ß-hydroxybutyrate, oxidized cytochrome c, ADP, Pi, and cyanide. ß-hydroxybutyrate is oxidized by an NAD+ -dependent dehydrogenase. 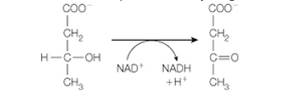
- The experimenter measured the rate of oxidation of ß-hydroxybutyrate and the rate of formation of ATP.
- Indicate the probable flow of electrons in this system.
- How many moles of ATP would you expect to be formed per mole of ß-hydroxybutyrate oxidized in this system?
- Why is ß-hydroxybutyrate added rather than NADH?
- What is the function of the cyanide?
- Write a balanced equation for the overall reaction occurring in this system (electron transport and ATP synthesis).
- Calculate the net standard free energy change (ΔGO') in this system, using
E'o values from Table 14.1 and a ΔGO' value for ATP hydrolysis of -32.2 kJ / mol.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Assessment
+1501 pts
/1600
Resources
Solution
? Hint
Sub
bo Each pictured Lewis structure is invalid. Identify the error in each case.
O Macmillan Learning
:0▬▬0:
Answer Bank
wrong electron total
:0-
:F======F:
octet-rule violation
N
:0:
[s] mM
V (M/s)
Uninhibited
0.333
1.65 x 107
1.05 x 107
V (M/s) x 10'
Inhibitor A
V (M/s) x 107
Inhibitor B
0.794 x 107
0.40
1.86 x 107
1.21 x 107
0.893 x 107
0.50
2.13 x 107
1.43 x 107
1.02 x 107
0.666
2.49 x 107
1.74 x 107
1.19 x 107
1.0
2.99 x 107
2.22 x 107
1.43 x 107
2.0
3.72 x 107
3.08 x 107
1.79 x 107
For a Michaelis-Menten reaction, k₁-5 x 10'/M-s, k.-2 x 10%/s, and k₂-4 x 10²/s.
a)
Calculate the Ks and KM for this reaction.
b)
Does substrate binding achieve equilibrium or steady state?
Chapter 14 Solutions
Biochemistry: Concepts and Connections (2nd Edition)
Ch. 14 - Prob. 1PCh. 14 - When pure reduced cytochrome c is added to...Ch. 14 - Freshly prepared mitochondria were incubated with...Ch. 14 - Prob. 4PCh. 14 - Prob. 5PCh. 14 - Prob. 6PCh. 14 - Intramitochondrial ATP concentrations are about 5...Ch. 14 - Prob. 8PCh. 14 - Prob. 9PCh. 14 - Years ago there was interest in using uncouplers...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- Assume that an enzyme-catalyzed reaction follows the scheme shown: E+S SES →E + P k₁ = 1 x 109/M-s k-1=2.5 x 10%/s k₂ = 3.4 x 107/s What is the dissociation constant for the enzyme-substrate, K,? What is the Michaelis constant, Km, for this enzyme? What is the turnover number, Keat, for this enzyme? What is the catalytic efficiency for the enzyme? If the initial Et concentration is 0.25mM, what is Vmax?arrow_forwardAn enzyme lowers the activation energy, (AG) of a reaction from 50.0 kcal/mol to 40.0 kcal/mol. Calulate the catalytic power at 310K. (R-1.987x10 kcal/mol)arrow_forwardDraw a typical axodendritic synapse, including a specific neurotransmitter of your choice, its associated postsynaptic receptors (indicating whether they are ionotropic or metabotropic), and any associated reuptake transporters or degradation enzymes. Please include a description of what specific steps would occur as an action potential reaches the axonal terminal.arrow_forward
- Give a full arrow pushing mechanism of the spontaneous redox reaction between NAD+/NADH and oxaloacetate/malate. Please include diagram drawing of the mechanism! (Thank You!)arrow_forward18. Which one of the compounds below is the major organic product obtained from the following series of reactions? 1. BH3 2. H2O2, NaOH H₂CrO4 CH2N2 oro ororos A B C D Earrow_forward17. Which one of the compounds below is the major organic product obtained from the following series of reactions? CI benzyl alcohol OH PBr3 Mg 1. CO2 SOCl2 ? ether 2. H+, H₂O CI Cl HO OH CI Cl A B C D Earrow_forward
- 14. What is the IUPAC name of this compound? A) 6-hydroxy-4-oxohexanenitrile B) 5-cyano-3-oxo-1-pentanol C) 5-cyano-1-hydroxy-3-pentanone D) 1-cyano-5-hydroxy-3-pentanone E) 5-hydroxy-3-oxopentanenitrile HO. CNarrow_forward13. What is the IUPAC name of this compound? A) 5-hydroxy-3,3-dimethylpentanoic acid B) 3,3-dimethylpentanoic acid C) 3,3-dimethyl-1-oxo-1,5-pentanediol D) 1,5-dihydroxy-3,3-dimethylpentanal E) 4-hydroxy-2,2-dimethylbutanoic acid HO OHarrow_forwardHelp me understand how carbon disulfide leads to toxicity in the brain, using terms like distal axonopathy, neurofilaments, covalent cross-linking, adducts, etc.,...please intuitively explain what is happening and where and the effects of it. For example, I know that CS2 reacts with amide and sulfhydryl groups on proteins, but what proteins exactly and where are they located?arrow_forward
- What is the standard free energy change (in kJ/mole) of the spontaneous reaction between Oxygen and NADH to form H2O2 and NAD+?arrow_forwardRedox Chemistry: Give standard free energy changes expected for the following reactions:-Succinate -> fumarate (using FAD/FADH2)-Oxaloacetate -> Malate (using NAD/NADH)-NADH --> NAD+ (using FMN/FMNH2)-CoQ --> CoQH2 (using Cytochrome C)arrow_forwardGive examples of balanced redox reactions that match the following:-Catabolic-Anabolic-Oxidative-Reductivearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning
Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
 Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning
Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning

Human Physiology: From Cells to Systems (MindTap ...
Biology
ISBN:9781285866932
Author:Lauralee Sherwood
Publisher:Cengage Learning

Concepts of Biology
Biology
ISBN:9781938168116
Author:Samantha Fowler, Rebecca Roush, James Wise
Publisher:OpenStax College

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning

Biology (MindTap Course List)
Biology
ISBN:9781337392938
Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:Cengage Learning


Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:9781305389892
Author:Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:Cengage Learning
Mitochondrial mutations; Author: Useful Genetics;https://www.youtube.com/watch?v=GvgXe-3RJeU;License: CC-BY