
Concept explainers
(a)
Interpretation:
The IUPAC name of given compound has to be assigned.
Concept Introduction:
Any organic molecule can be named by using certain rules given by IUPAC (International Union for Pure and applied chemistry). IUPAC name consists of three parts in major namely Prefix suffix and root word.
Prefix represents the substituent present in the molecule and its position in the root name.
Suffix denotes the presence of
Root word represents the longest continuous carbon skeleton of the organic molecule.
IUPAC rules for naming ether:
- ✓ The base name is found from the longest carbon chain present in ether.
- ✓ The suffix –yl has to be changed to –oxy in order to obtain the alkoxy group name. For example, ethyl becomes as ethoxy, methyl becomes as methoxy etc.
- ✓ Alkoxy name has to be placed first with the number (carbon atom to which the alkoxy group is attached) followed by the base name.
(a)
Answer to Problem 14.104EP
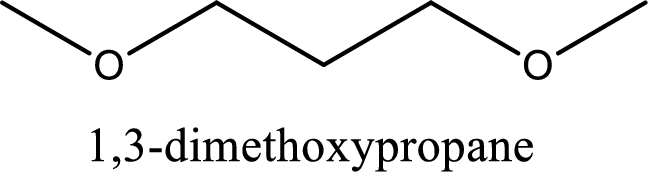
IUPAC name of the given compound is 1,3-dimethoxypropane.
Explanation of Solution
The structure of given compound is shown below,
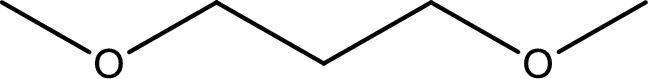
First step is to identify the longest carbon chain. In this case it is a three carbon chain. Hence, the base name is propane.
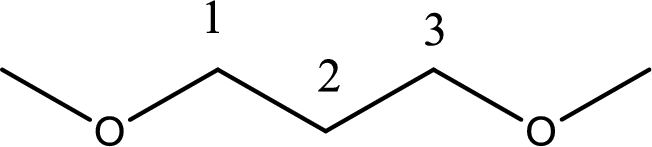
Next step is to identify the alkoxy group. In the given compound, the alkoxy group is found to be two methoxy groups.
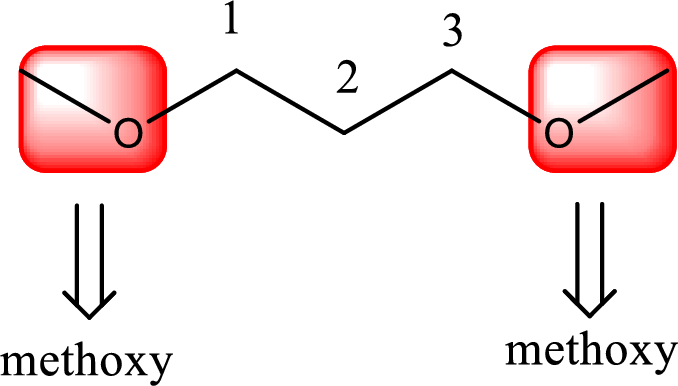
Alkoxy name is placed before the base name with the appropriate number that gives information about to which carbon atom the alkoxy group is attached. In this case as two methoxy groups are present prefix di- is added before methoxy. This gives the IUPAC name as 1,3-dimethoxypropane.
IUPAC name for the given compound is assigned.
(b)
Interpretation:
The IUPAC name of given compound has to be assigned.
Concept Introduction:
Concept Introduction:
IUPAC rules for naming alcohols that contain single hydroxyl group:
- • Longest carbon chain has to be identified that contains hydroxyl group also. The chain name is obtained by replacing the letter “-e” in
alkane with “-ol”. If the compound contains a unsaturated bond, then the respective name has to be changed with regard to alkane. - • The numbering has to be given so that the hydroxyl group gets the least numbering.
- • Name and location of any other substituent present in the chain has to be identified.
- • If in a ring the hydroxyl group is present, then that carbon is numbered 1 and the numbering then proceeds counterclockwise or clockwise in a way that substituents present if any gets the least numbering.
- • Hydroxyl group as a substituent in a molecule is named as hydroxy group rather than hydroxyl group.
- • If the compound contains bulky groups on same side of the double bond, then it is a cis isomer and if the bulkyl groups are present on opposite side of the double bond, then it is a trans isomer.
- • In case of cycloalkane compounds, if the substitutions are present on same side of the ring of carbon atoms, it is a cis isomer. If the substitutions are present above and below the ring, then it is a trans isomer.
IUPAC rules for naming alcohols that contain more than one hydroxyl group:
- • The same rules said above is followed but the prefix di-, tri-, tetra etc is added corresponding to the number of hydroxyl groups that is present.
(b)
Answer to Problem 14.104EP
IUPAC name of the given compound is 1-butanol.
Explanation of Solution
Given structure is,
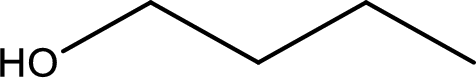
The longest continuous carbon chain with the hydroxyl group is found to be four carbon chain. Therefore, the parent alkane is butane. As a hydroxyl group is present the name of the alcohol can be given as butanol.
The numbering has to be given so that the hydroxyl group gets the least numbering. In this case, the hydroxyl group is present in the first carbon atom. Therefore, the IUPAC name can be given as 1-butanol.
IUPAC name for the given compound is assigned.
(c)
Interpretation:
The IUPAC name of given compound has to be assigned.
Concept Introduction:
Any organic molecule can be named by using certain rules given by IUPAC (International Union for Pure and applied chemistry). IUPAC name consists of three parts in major namely Prefix suffix and root word.
Prefix represents the substituent present in the molecule and its position in the root name.
Suffix denotes the presence of functional group if any in the molecule. It can be an alkene, alkyne, alcohol, carboxylic acid, alcohol etc.
Root word represents the longest continuous carbon skeleton of the organic molecule.
IUPAC rules for naming ether:
- ✓ The base name is found from the longest carbon chain present in ether.
- ✓ The suffix –yl has to be changed to –oxy in order to obtain the alkoxy group name. For example, ethyl becomes as ethoxy, methyl becomes as methoxy etc.
- ✓ Alkoxy name has to be placed first with the number (carbon atom to which the alkoxy group is attached) followed by the base name.
(c)
Answer to Problem 14.104EP
IUPAC name of the given compound is 1-ethoxypropane.
Explanation of Solution
The structure of given compound is shown below,

First step is to identify the longest carbon chain. In this case it is a three carbon chain. Hence, the base name is propane.
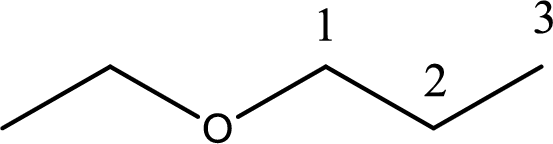
Next step is to identify the alkoxy group. In the given compound, the alkoxy group is found to be ethoxy as it contains two carbon atoms.
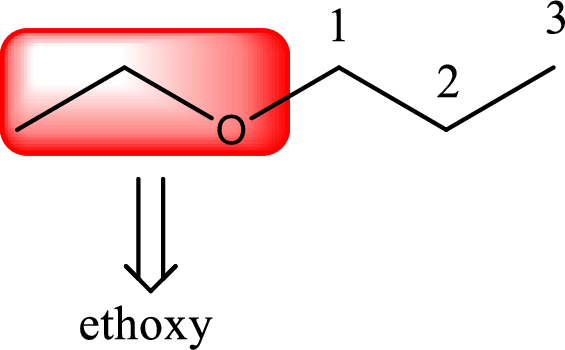
Alkoxy name is placed before the base name with the appropriate number that gives information about to which carbon atom the alkoxy group is attached. This gives the IUPAC name as 1-ethoxypropane.
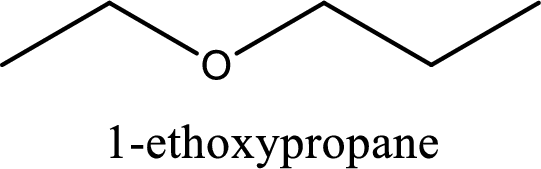
IUPAC name for the given compound is assigned.
(d)
Interpretation:
The IUPAC name of given compound has to be assigned.
Concept Introduction:
IUPAC rules for naming alcohols that contain single hydroxyl group:
- • Longest carbon chain has to be identified that contains hydroxyl group also. The chain name is obtained by replacing the letter “-e” in alkane with “-ol”. If the compound contains a unsaturated bond, then the respective name has to be changed with regard to alkane.
- • The numbering has to be given so that the hydroxyl group gets the least numbering.
- • Name and location of any other substituent present in the chain has to be identified.
- • If in a ring the hydroxyl group is present, then that carbon is numbered 1 and the numbering then proceeds counterclockwise or clockwise in a way that substituents present if any gets the least numbering.
- • Hydroxyl group as a substituent in a molecule is named as hydroxy group rather than hydroxyl group.
- • If the compound contains bulky groups on same side of the double bond, then it is a cis isomer and if the bulkyl groups are present on opposite side of the double bond, then it is a trans isomer.
- • In case of cycloalkane compounds, if the substitutions are present on same side of the ring of carbon atoms, it is a cis isomer. If the substitutions are present above and below the ring, then it is a trans isomer.
IUPAC rules for naming alcohols that contain more than one hydroxyl group:
- • The same rules said above is followed but the prefix di-, tri-, tetra etc is added corresponding to the number of hydroxyl groups that is present.
(d)
Answer to Problem 14.104EP
IUPAC name of the given compound is 2-methoxyphenol.
Explanation of Solution
The structure of given compound is shown below,
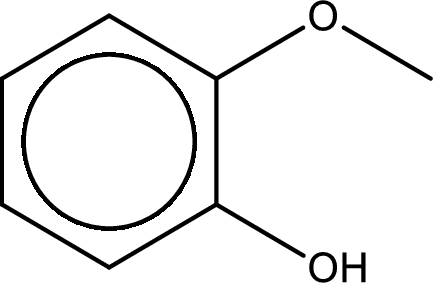
The given compound has two functional group namely, hydroxyl group and a alkoxy group. Alcohol has got more preference over ether. Therefore, the IUPAC name is given by using the parent as alcohol. In this case the parent alcohol is found to be phenol. The substituent that is present in the phenol is a methoxy group in the second carbon atom. Therefore, the IUPAC name of the given compound can be written as 2-methoxyphenol.
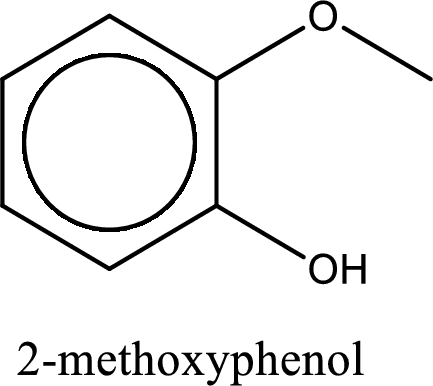
IUPAC name for the given compound is assigned.
Want to see more full solutions like this?
Chapter 14 Solutions
General, Organic, and Biological Chemistry
- You have identified a new species of a Gram-positive bacteria. You would like to screen their genome for all proteins that are covalently linked to the cell wall. You have annotated the genome, so that you identified all the promoters, operons, and genes sequences within the operons. Using these features, what would you screen for to identify a set of candidates for proteins covalently linked to the bacterial cell wall.arrow_forwardBelow is a diagram from a genomic locus of a bacterial genome. Each arrow represents a coding region, and the arrowheads indicate its orientation in the genome. The numbers are randomly assigned. Draw the following features on the diagram, and explain your rationale for each feature: 10 12 合會會會會長 6 a) Expected transcriptions, based on known properties of bacterial genes and operons. How many proteins are encoded in each of the transcripts? b) Location of promoters (include rationale) c) Location of transcriptional terminators (include rationale) d) Locations of Shine-Dalgarno sequences (include rationale)arrow_forwardSample excuse letter in school class for the reasons of headaches and dysmenorrhea caused by menstrual cyclearrow_forward
- How do the muscles on the foot work to balance on an ice skate, specifically the triangle of balance and how does it change when balancing on an ice skate? (Refer to anatomy, be specific)arrow_forwardWhich of the following is NOT an example of passive immunization? A. Administration of tetanus toxoid B. Administration of hepatitis B immunoglobulin C. Administration of rabies immunoglobulin D. Transfer of antibodies via plasma therapyarrow_forwardTranscription and Translation 1. What is the main function of transcription and translation? (2 marks) 2. How is transcription different in eukaryotic and prokaryotic cells? (2 marks) 3. Explain the difference between pre-mRNA and post-transcript mRNA. (2 marks) 4. What is the function of the following: (4 marks) i. the cap ii. spliceosome iii. Poly A tail iv. termination sequence 5. What are advantages to the wobble feature of the genetic code? (2 marks) 6. Explain the difference between the: (3 marks) i. A site & P site ii. codon & anticodon iii. gene expression and gene regulation 7. Explain how the stop codon allows for termination. (1 mark) 8. In your own words, summarize the process of translation. (2 marks)arrow_forward
- In this activity you will research performance enhancers that affect the endocrine system or nervous system. You will submit a 1 page paper on one performance enhancer of your choice. Be sure to include: the specific reason for use the alleged results on improving performance how it works how it affect homeostasis and improves performance any side-effects of this substancearrow_forwardNeurons and Reflexes 1. Describe the function of the: a) dendrite b) axon c) cell body d) myelin sheath e) nodes of Ranvier f) Schwann cells g) motor neuron, interneuron and sensory neuron 2. List some simple reflexes. Explain why babies are born with simple reflexes. What are they and why are they necessary. 3. Explain why you only feel pain after a few seconds when you touch something very hot but you have already pulled your hand away. 4. What part of the brain receives sensory information? What part of the brain directs you to move your hand away? 5. In your own words describe how the axon fires.arrow_forwardMutations Here is your template DNA strand: CTT TTA TAG TAG ATA CCA CAA AGG 1. Write out the complementary mRNA that matches the DNA above. 2. Write the anticodons and the amino acid sequence. 3. Change the nucleotide in position #15 to C. 4. What type of mutation is this? 5. Repeat steps 1 & 2. 6. How has this change affected the amino acid sequence? 7. Now remove nucleotides 13 through 15. 8. Repeat steps 1 & 2. 9. What type of mutation is this? 0. Do all mutations result in a change in the amino acid sequence? 1. Are all mutations considered bad? 2. The above sequence codes for a genetic disorder called cystic fibrosis (CF). 3. When A is changed to G in position #15, the person does not have CF. When T is changed to C in position #14, the person has the disorder. How could this have originated?arrow_forward
- hoose a scientist(s) and research their contribution to our derstanding of DNA structure or replication. Write a one page port and include: their research where they studied and the time period in which they worked their experiments and results the contribution to our understanding of DNA cientists Watson & Crickarrow_forwardhoose a scientist(s) and research their contribution to our derstanding of DNA structure or replication. Write a one page port and include: their research where they studied and the time period in which they worked their experiments and results the contribution to our understanding of DNA cientists Watson & Crickarrow_forward7. Aerobic respiration of a protein that breaks down into 12 molecules of malic acid. Assume there is no other carbon source and no acetyl-CoA. NADH FADH2 OP ATP SLP ATP Total ATP Show your work using dimensional analysis here: 3arrow_forward
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning- Essentials of Pharmacology for Health ProfessionsNursingISBN:9781305441620Author:WOODROWPublisher:Cengage





