
Loose Leaf For Introduction To Chemical Engineering Thermodynamics
8th Edition
ISBN: 9781259878084
Author: Smith Termodinamica En Ingenieria Quimica, J.m.; Van Ness, Hendrick C; Abbott, Michael; Swihart, Mark
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 13, Problem 13.14P
Air, even more than carbon dioxide, is inexpensive and nontoxic. Why is it not the gas of choice for making soda water and (cheap) champagne effervescent? Table 13.2 may provide useful data.
Table 13.2: Henry’s Constants for Gases Dissolved in Water at 25°C
| Gas | 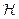 bar bar |
| Acetylene | 1,350 |
| Air | 72,950 |
| Carbon dioxide | 1,670 |
| Carbon monoxide | 54,600 |
| Ethane | 30,600 |
| Ethylene | 11,550 |
| Helium | 126,600 |
| Hydrogen | 71,600 |
| Hydrogen sulfide | 550 |
| Methane | 41,850 |
| Nitrogen | 87,650 |
| Oxygen | 44,380 |
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
9.4. A PID temperature controller is at steady state with an output pressure of 9 psig. The set
point and process temperature are initially the same. At time = 0, the set point is increased
at the rate of 0.5°F/min. The motion of the set point is in the direction of lower temperatures.
If the current settings are
PART 3 LINEAR CLOSED-LOOP SYSTEMS
Ke
=
2 psig/°F
Ti =
1.25 min
TD = 0.4 min
plot the output pressure versus time.
9.6. A PI controller has the transfer function
Determine the values of K, and T.
5s + 10
Ge
S
9.5. The input & to a PI controller is shown in Fig. P9-5. Plot the output of the controller if
Ke 2 and 0.50 min.
-
E
0.5
0
-0.5
0
FIGURE P9-5
2
4
t, min
Chapter 13 Solutions
Loose Leaf For Introduction To Chemical Engineering Thermodynamics
Ch. 13 - Prob. 13.1PCh. 13 - Assuming Raoult’s law to be valid, prepare a Pxy...Ch. 13 - Assuming Raoult’s law to apply to the system...Ch. 13 - Prob. 13.4PCh. 13 - Prove: An equilibrium liquid/vapor system...Ch. 13 - Of the following binary liquid/vapor systems,...Ch. 13 - A single-stage liquid/vapor separation for the...Ch. 13 - Do all four parts of Prob. 13.7, and compare the...Ch. 13 - A mixture containing equimolar amounts of...Ch. 13 - Do all four parts of Prob. 13.9. and compare the...
Ch. 13 - A binary mixture of mole fraction z1is flashed to...Ch. 13 - Humidity, relating to the quantity of moisture in...Ch. 13 - A concentrated binary solution containing mostly...Ch. 13 - Air, even more than carbon dioxide, is inexpensive...Ch. 13 - Helium-laced gases are used as breathing media for...Ch. 13 - A binary system of species 1 and 2 consists of...Ch. 13 - For the system ethyl ethanoate(l)/n-heptane(2) at...Ch. 13 - A liquid mixture of cyclohexanone(1)/phenol(2) for...Ch. 13 - A binary system of species 1 and 2 consists of...Ch. 13 - For the acetone(l)/methanol(2) system, a vapor...Ch. 13 - The following is a rule of thumb: For a binary...Ch. 13 - A process stream contains light species 1 and...Ch. 13 - If a system exhibits VLE, at least one of the...Ch. 13 - Flash calculations are simpler for binary systems...Ch. 13 - Prob. 13.25PCh. 13 - (a) A feed containing equimolar amounts of...Ch. 13 - A binary mixture of benzene(1) and toluene(2) is...Ch. 13 - Ten (10) kmolhr-1 of hydrogen sulfide gas is...Ch. 13 - Physiological studies show the neutral comfort...Ch. 13 - Prob. 13.30PCh. 13 - Prob. 13.31PCh. 13 - Prob. 13.32PCh. 13 - If Eq. (13.24) is valid for isothermal VLE in a...Ch. 13 - Prob. 13.34PCh. 13 - The excess Gibbs energy for binary systems...Ch. 13 - For the ethanol(l )/chloroform(2) system at 50°C,...Ch. 13 - VLE data for methyl tert-butyl...Ch. 13 - Prob. 13.38PCh. 13 - Prob. 13.39PCh. 13 - Following are VLE data for the system...Ch. 13 - Prob. 13.41PCh. 13 - Prob. 13.42PCh. 13 - Problems 13.43 through 13.54 require parameter...Ch. 13 - Problems 13.43 through 13.54 require parameter...Ch. 13 - Prob. 13.45PCh. 13 - Problems 13.43 through 13.54 require parameter...Ch. 13 - Prob. 13.47PCh. 13 - Prob. 13.48PCh. 13 - Prob. 13.49PCh. 13 - Prob. 13.50PCh. 13 - Problems 13.43 through 13.54 require parameter...Ch. 13 - Prob. 13.52PCh. 13 - The following expressions have been reported for...Ch. 13 - Possible correlating equations for In 1 in a...Ch. 13 - Prob. 13.57PCh. 13 - Binary VLE data are commonly measured at constant...Ch. 13 - Consider the following model for GE/RT of a binary...Ch. 13 - A breathalyzer measures volume-% ethanol in gases...Ch. 13 - Table 13.10 gives values of parameters for the...Ch. 13 - Prob. 13.62PCh. 13 - A single P-x1- y1data point is available for a...Ch. 13 - A single P- x1, data point is available for a...Ch. 13 - The excess Gibbs energy for the system...Ch. 13 - Prob. 13.66PCh. 13 - A system formed of methane(l) and a light oil(2)...Ch. 13 - Use Eq. (13.13) to reduce one of the following...Ch. 13 - For one of the following substances, determine...Ch. 13 - Departures from Raoult's law are primarily from...Ch. 13 - The relative volatility a12is commonly used in...Ch. 13 - Prob. 13.74PCh. 13 - Prob. 13.75PCh. 13 - Prob. 13.76P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemical-engineering and related others by exploring similar questions and additional content below.Similar questions
- 9.3. An ideal PD controller has the transfer function P Ke (TDs + 1) E An actual PD controller had the transfer function P = Ke E TDS +1 (TDIẞ)s +1 where ẞis a large constant in an industrial controller. If a unit-step change in error is introduced into a controller having the second transfer function, show that P(1) = Kc (1 + Ae˜¯BD) where A is a function of ẞwhich you are to determine. For ẞ=5 and K = 0.5, plot P(t) versus tl tp. As ẞ, show that the unit-step response approaches that for the ideal controller.arrow_forward9.1. A pneumatic PI temperature controller has an output pressure of 10 psig when the set point and process temperature coincide. The set point is suddenly increased by 10°F (i.e., a step change in error is introduced), and the following data are obtained: Time, s psig 0- 10 0+ 8 20 7 60 90 5 3.5 Determine the actual gain (psig per degree Fahrenheit) and the integral time.arrow_forward2. A unit-step change in error is introduced into a PID controller. If Ke TD = 0.5, plot the response of the controller P(t). = =10, 1, andarrow_forward
- A distribution of values is normal with a mean of 211 and a standard deviation of 50.4. Find the probability that a randomly selected value is between 59.8 and 155.6. P(59.8 X 155.6) = Enter your answer as a number accurate to 4 decimal places. Answers obtained using exact z-scores or z- scores rounded to 3 decimal places are accepted.arrow_forwardTopic: Production of propylene glycol from glycerol derived from palm oil. QUESTION:Estimate capital items, operating costs and economics of the plant. Finally, report the estimatedreturn.The Detailed Factorial Method with approximately 25% accuracy must be used for detailedeconomic evaluation.Plant lifetime is fixed at 15 years.1) Depreciation and TaxesCalculate the depreciation using a suitable method (e.g., straight-line, declining balance) andincorporate tax implications based on current tax laws applicable to chemical plants. Use following attached Process Flow Diagram as reference for this question.arrow_forwardTopic: Production of propylene glycol from glycerol derived from palm oil. QUESTION:Estimate capital items, operating costs and economics of the plant. Finally, report the estimatedreturn.The Detailed Factorial Method with approximately 25% accuracy must be used for detailedeconomic evaluation.Plant lifetime is fixed at 15 years.1) Revenue EstimationEstimate the annual revenue based on the production capacity, product selling price, and marketdemand. Groups should also consider potential market fluctuations. Use following attached Process Flow Diagram as reference for this question.arrow_forward
- Topic: Production of propylene glycol from glycerol derived from palm oil. QUESTION:Estimate capital items, operating costs and economics of the plant. Finally, report the estimatedreturn.The Detailed Factorial Method with approximately 25% accuracy must be used for detailedeconomic evaluation.Plant lifetime is fixed at 15 years.TASKS:1) Capital Cost EstimationProvide a detailed breakdown of the initial capital investment, including land, building,equipment, and installation costs. Include any assumptions made in the estimation. Use following attached Process Flow Diagram as reference for this question.arrow_forwardTopic: Production of propylene glycol from glycerol derived from palm oil. QUESTION:Estimate capital items, operating costs and economics of the plant. Finally, report the estimatedreturn.The Detailed Factorial Method with approximately 25% accuracy must be used for detailedeconomic evaluation.Plant lifetime is fixed at 15 years.1) Breakeven Year CalculationUsing the cash flow analysis, calculate the breakeven year when the cumulative cash inflowequals the initial investment. Groups should graphically represent the breakeven point. Use following attached Process Flow Diagram as reference for this question.arrow_forwardTopic: Production of propylene glycol from glycerol derived from palm oil. QUESTION:Estimate capital items, operating costs and economics of the plant. Finally, report the estimatedreturn.The Detailed Factorial Method with approximately 25% accuracy must be used for detailedeconomic evaluation.Plant lifetime is fixed at 15 years.1) Cash Flow AnalysisDevelop a projected cash flow statement for the first 10 years of plant operation, consideringall the costs and revenues. Include working capital, loans, and interest payments if applicable. Use following attached Process Flow Diagram as reference for this question.arrow_forward
- Topic: Production of propylene glycol from glycerol derived from palm oil.QUESTION:Estimate capital items, operating costs and economics of the plant. Finally, report the estimatedreturn.The Detailed Factorial Method with approximately 25% accuracy must be used for detailedeconomic evaluation.Plant lifetime is fixed at 15 years.1) Operational Cost AnalysisCalculate the yearly operational costs, including raw materials, labor, utilities, maintenance,and other recurring expenses. Provide a clear explanation of how these costs are derived. Use following attached Process Flow Diagram as reference for this question.arrow_forwardChemical Engineering Questionarrow_forwardA steam boiler or steam generator is a device used to produce steam by transferring heat to water. In our case, the combustion chamber is fueled with propane (C3H8) at a flowrate of 50.0 mol/h in an excess air of 50%. Assume that both propane and air are fed at 25ºC and the combustion gases leave the chamber at 200ºC. Pressure can be assumed to be atmospheric.* Determine: 1. The heat obtained assuming complete combustion. Compare the results using elements or compounds 2. The steam flowrate that could be generated if the heat is directed to obtain superheated steam at 2 bar and 160ºC from saturated liquid water at this pressure solvearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education
Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
 Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning
Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The
Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The

Introduction to Chemical Engineering Thermodynami...
Chemical Engineering
ISBN:9781259696527
Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind...
Chemical Engineering
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY

Elements of Chemical Reaction Engineering (5th Ed...
Chemical Engineering
ISBN:9780133887518
Author:H. Scott Fogler
Publisher:Prentice Hall


Industrial Plastics: Theory and Applications
Chemical Engineering
ISBN:9781285061238
Author:Lokensgard, Erik
Publisher:Delmar Cengage Learning

Unit Operations of Chemical Engineering
Chemical Engineering
ISBN:9780072848236
Author:Warren McCabe, Julian C. Smith, Peter Harriott
Publisher:McGraw-Hill Companies, The
Homogeneous and Heterogeneous Equilibrium - Chemical Equilibrium - Chemistry Class 11; Author: Ekeeda;https://www.youtube.com/watch?v=8V9ozZSKl9E;License: Standard YouTube License, CC-BY