
Concept explainers
(a)
Interpretation:
If the given statement is true or false should be determined.
Concept Introduction:
Answer to Problem 29P
The statement istrue.
Explanation of Solution
Alkenes are unsaturated hydrocarbons with carbon-carbon double bonds. The combustion reaction takes place in the presence of air. The combustion of alkenes produces carbon dioxide and water.
Hence, the given statement is true.
(b)
Interpretation:
The given statement is true or false should be determined.
Concept Introduction:
Alkenes or olefins are the unsaturated hydrocarbons. They are organic compounds with one or more carbon-carbon double bonds. Alkenes are more reactive than alkanes. They undergo many characteristic reactions namely Addition reactions, oxidation reactions and combustion reactions.
Answer to Problem 29P
The statement is true.
Explanation of Solution
The addition reaction of alkenes is one double bond of alkene and one single bond of a second reactant break, and three new single bonds are formed such that the addition of the second reactant occurs at the carbon atoms of the double bond. Hence, the two new single bonds are formed at the doubly bonded carbon atoms. Therefore, the given statement is true.
(c)
Interpretation:
The given statement is true or false should be determined.
Concept Introduction:
Alkenes or olefins are the unsaturated hydrocarbons. They are organic compounds with one or more carbon-carbon double bonds. Alkenes are more reactive than alkanes. They undergo many characteristic reactions namely Addition reactions, oxidation reactions and combustion reactions.
Answer to Problem 29P
The statement is true.
Explanation of Solution
Regioselectivity means the preference of making or breaking of bonds over all other possibilities. Markovnikov's rule states that in the addition of a halogen acid to an alkene, hydrogen goes to the carbon atom in a double bond that bears a higher number of hydrogen atoms. Thus, it indicates the position of the attack. Hence, Markovnikov's rule refers to the regioselectivity of the carbon-carbon double bond. Therefore, the given statement is true.
(d)
Interpretation:
The given statement is true or false should be determined.
Concept Introduction:
Alkenes or olefins are the unsaturated hydrocarbons. They are organic compounds with one or more carbon-carbon double bonds. Alkenes are more reactive than alkanes. They undergo many characteristic reactions namely Addition reactions, oxidation reactions and combustion reactions.
Answer to Problem 29P
The statement is true.
Explanation of Solution
Markovnikov's rule states that addition of a halogen acid (HX, x = Cl, Br or I) to an alkene, hydrogen goes to the carbon atom in the double bond that bears a higher number of hydrogen atoms. In additionof HCI, HBr, and HI to an alkene, hydrogen attaches to the carbon of the double bond that already has a greater number of hydrogen atoms bonded to it. Therefore, the given statement is true.
(e)
Interpretation:
The given statement is true or false should be determined.
Concept Introduction:
Alkenes or olefins are the unsaturated hydrocarbons. They are organic compounds with one or more carbon-carbon double bonds. Alkenes are more reactive than alkanes. They undergo many characteristic reactions namely Addition reactions, oxidation reactions and combustion reactions.
Answer to Problem 29P
The statement is false.
Explanation of Solution
A carbocation is a carbon atom with three bonds and is deficient in one pair of the electrons, thus, acquiring a positive charge. Therefore, the given statement is false.
(f)
Interpretation:
The given statement is true or false should be determined.
Concept Introduction:
Alkenes or olefins are the unsaturated hydrocarbons. They are organic compounds with one or more carbon-carbon double bonds. Alkenes are more reactive than alkanes. They undergo many characteristic reactions namely Addition reactions, oxidation reactions and combustion reactions.
Answer to Problem 29P
The statement is true.
Explanation of Solution
The carbocation is derived from ethylene by the attack of a proton as depicted in the equation is CH3 CH2 +.
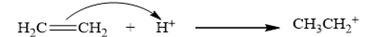
Therefore, the given statement is true.
(g)
Interpretation:
The given statement is true or false should be determined.
Concept Introduction:
Alkenes or olefins are the unsaturated hydrocarbons. They are organic compounds with one or more carbon-carbon double bonds. Alkenes are more reactive than alkanes. They undergo many characteristic reactions namely Addition reactions, oxidation reactions and combustion reactions.
Answer to Problem 29P
The statement is true.
Explanation of Solution
The mechanism of the addition of HX to an alkene is a two-step reaction.
Step 1: The formation of the carbocation. In the first step of the reaction, the proton is added to the double bond, and the carbocation is generated as carbocation intermediate.
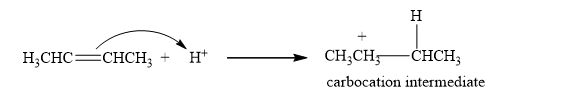
Step 2: The Nucleophilic attack. Then, a halide ion which is a nucleophile attacks the carbocation, forming an additional product.
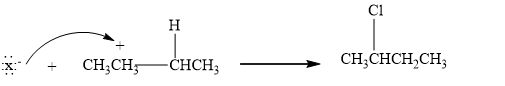
Therefore, the given statement is true.
(h)
Interpretation:
The given statement is true or false should be determined.
Concept Introduction:
Alkenes or olefins are the unsaturated hydrocarbons. They are organic compounds with one or more carbon-carbon double bonds. Alkenes are more reactive than alkanes. They undergo many characteristic reactions namely Addition reactions, oxidation reactions and combustion reactions.
Answer to Problem 29P
The statement is true.
Explanation of Solution
The addition of water is known as hydration. According to Markovnikov's rule, in acid, the catalyzed addition of water to an alkene water molecule is added to the carbon-carbon double bond. Furthermore, in the presence of acid, the alkene is converted to alcohol. Therefore, the given statement is true.
(i)
Interpretation:
The given statement is true or false should be determined.
Concept Introduction:
Alkenes or olefins are the unsaturated hydrocarbons. They are organic compounds with one or more carbon-carbon double bonds. Alkenes are more reactive than alkanes. They undergo many characteristic reactions namely Addition reactions, oxidation reactions and combustion reactions.
Answer to Problem 29P
The statement is true.
Explanation of Solution
The addition of Br2 to a carbon-carbon double bond takes place at room temperature. Thus, if a compound fails to react with Br2, then the compound does not contain a double bond. Therefore, the given statement is true.
(j)
Interpretation:
The given statement is true or false should be determined.
Concept Introduction:
Alkenes or olefins are the unsaturated hydrocarbons. They are organic compounds with one or more carbon-carbon double bonds. Alkenes are more reactive than alkanes. They undergo many characteristic reactions namely Addition reactions, oxidation reactions and combustion reactions.
Answer to Problem 29P
The statement is true.
Explanation of Solution
The addition of H2 is known as reduction. During a reaction with molecular H2, all of the alkenes, are converted to alkanes. Hence, hydrogen is added to the double bond of an alkene. Thus, the given statement is true.
(k)
Interpretation:
The given statement is true or false should be determined.
Concept Introduction:
Alkenes or olefins are the unsaturated hydrocarbons. They are organic compounds with one or more carbon-carbon double bonds. Alkenes are more reactive than alkanes. They undergo many characteristic reactions namely Addition reactions, oxidation reactions and combustion reactions.
Answer to Problem 29P
The statement is false.
Explanation of Solution
Catalytic reduction is the process in which a substance is reduced by hydrogen in the presence of a catalyst. The catalytic reduction of cyclohexene is the addition of H2 across the double bond that is present in the cyclic ring. The resultant product is cyclohexane, not hexane. Thus, the given statement is false.
(l)
Interpretation:
The given statement is true or false should be determined.
Concept Introduction:
Alkenes or olefins are the unsaturated hydrocarbons. They are organic compounds with one or more carbon-carbon double bonds. Alkenes are more reactive than alkanes. They undergo many characteristic reactions namely Addition reactions, oxidation reactions and combustion reactions.
Answer to Problem 29P
The statement is false.
Explanation of Solution
The acid catalyzed hydration of alkene is completed in three steps. The first step includes the addition of a proton (from acid) to the double bond that results in the formation of a carbocation, and hydrogen attaches to one of the doubly bonded carbons. In the second step, an oxygen atom of a water molecule attacks the positively charged carbon, and the water molecule attaches to it, forming an intermediate called an oxonium ion. In the third step, the proton is regenerated again by losing H+ from the oxonium ion, Therefore, in the final product, OH comes from a water molecule, while H comes from the acid used as a catalyst. Thus, the given statement is false.
(m)
Interpretation:
The given statement is true or false should be determined.
Concept Introduction:
Alkenes or olefins are the unsaturated hydrocarbons. They are organic compounds with one or more carbon-carbon double bonds. Alkenes are more reactive than alkanes. They undergo many characteristic reactions namely Addition reactions, oxidation reactions and combustion reactions.
Answer to Problem 29P
The statement is false.
Explanation of Solution
Oxidation is characterized as a gain in oxygen or a loss of hydrogen or a molecule. The alkene on the acid-catalyzed hydration is converted into alcohol. Thus, the conversion of CH2 = CH2 to ethanol is an addition reaction in which a molecule of water is added to the double bond. Thus, there is neither a loss of hydrogen nor a gain of oxygen. Hence, this is an addition reaction and not an oxidation reaction, so, the given statement is false.
(n)
Interpretation:
The given statement is true or false should be determined.
Concept Introduction:
Alkenes or olefins are the unsaturated hydrocarbons. They are organic compounds with one or more carbon-carbon double bonds. Alkenes are more reactive than alkanes. They undergo many characteristic reactions namely Addition reactions, oxidation reactions and combustion reactions.
Answer to Problem 29P
The statement is false.
Explanation of Solution
According to Markovnikov's rule, in the acid-catalyzed hydration of alkene, water is added to the double bond of alkene such that the addition of the H and OH groups occur. Furthermore, it says that H is added to the carbon with more of the hydrogen atoms. The acid catalyzed hydration of 1-butene and 2-butene both result in the formation of 2-butanol. Hence the given statement is false.
Want to see more full solutions like this?
Chapter 12 Solutions
Introduction to General, Organic and Biochemistry
- Give the organic products: (benzyne) Br ? CH3 + K* :NH, liq NH3 HINT: Two products are formed. Each is a substituted aniline; they are isomers of each other. NH2 II I H₂N. CH3 CH3 III Select one: ○ A. I and II ○ B. I and III O C. I and IV O D. II and III O E. III and IV H₂N CH3 IV CH₂-NH2arrow_forwardPredict the major products of this organic reaction: HBr (1 equiv) cold ? Some important notes: • Draw the major product, or products, of this reaction in the drawing area below. • You can draw the products in any arrangement you like. • Pay careful attention to the reaction conditions, and only include the major products. • Be sure to use wedge and dash bonds when necessary, for example to distinguish between major products that are enantiomers. • Note that there is only 1 equivalent of HBr reactant, so you need not consider the case of multiple additions. Erase something Explanation Check 2025 McGraw Hill LLC. All Rights Reserved. Terarrow_forwardQ14. Fill this chart: (please refer to ppt notes/browser to answer these questions) What alcohol is also called wood alcohol? What is the common name of ethanol? Draw the structure of phenol and thiophene? Are bigger chain alcohol like heptanol and octanol are soluble or insoluble in water and explain it ? Are ethers soluble or insoluble in water? What suffix and prefix are used for alcohol while naming alcohol and ether? What the process called when we add water to any alkene to make alcohol? Q16. Draw the diagram of following aromatic compound (practice from previous module) Aniline Phenol Benzoic acid Methyl benzoate Q17. a. Write the oxidation reactions for the 2 propanol. b. Write the oxidation reaction of the ethanol.arrow_forward
- Question 11 of 18 (1 point) Question Attempt: 3 of How many signals do you expect in the 'H NMR spectrum for this molecule? Br Br Write the answer below. Also, in each of the drawing areas below is a copy of the molecule, with Hs shown. In each copy, one of the H atoms is colored red. Highlight in red all other H atoms that would contribute to the same signal as the H already highlighted red. Note for advanced students: In this question, any multiplet is counted as one signal. Number of signals in the 'H NMR spectrum. 1 For the molecule in the top drawing area, highlight in red any other H atoms that will contribute to the same signal as the H atom already highlighted red. If no other H atoms will contribute, check the box at right. No additional Hs to color in top molecule Check For the molecule in the bottom drawing area, highlight in red any other H atoms that will contribute to the same signal as the H atom already highlighted red. If no other H atoms will contribute, check the box…arrow_forwardOrganic Chemistry Esterification reactions 1. Write the steps to prepare ester. 2. Write complete reaction of ethanol and acetic acid to make ester. 3. What does ester smell like? What are the uses of ester. 4. What the role of sulfuric acid in the esterification reactionarrow_forward11. Complete the following esterification reaction with names of all the reactants and products under. Hint: Remove the water and end up with ester R-C-OH + ROH R-C-OR + H₂O A carboxylic acid An alcohol An ester Water BYJU'S H-C-C O-H Нин C-C-C-H HAAA H O-C-C-C-H AAA Ethanoic acid Propanol Water Propyl ethanoate By com CH3COOH + CH3CH2CH2CH₂CH₂OH → Practice for alcohols aldehydes and ketones: 12. Draw the structures from the following names mixed of alcohol/aldehyde and ketone: a. 4-methyl cyclohexanone b. 3-methyl-2-pentenal c. 2,3-dimethylcyclohexanone d. 1,3propanediol or Propane 1,3 diol 13. Write systematic names for the following compounds identify functional group: a. b. (CH3)2CH-C OH c) CH(CH₂)-- OH -,-,arrow_forward
- may you please show all steps! i am having a hard time understanding and applying in this format, thank you!arrow_forward10. Complete the substitution reaction of 2 pentanol with these reagents. Reagents & Reaction Conditions use practice sheet. Please write only major products, minor product like water, other gases are not required. Hint: In substitution of alcohol, we generally substitute OH group with Halogens like cl, Br, F using some reagent containing halogens. Ensure to add halogens to the same carbon number where you are removing OH from Examples Alcohols can be converted to Alkyl Halides with HX acids HBr H₂O HCI + H₂O HI + H₂O CH,CH₂OH + SOCI₂ CH,CH₂OH + PCI₁₂ A BBYJU'S CH CHCI + SO₂+ HCI CH₂CH CIP(OH), + HCI CH,CH₂OH + PCI CHCHCI + POCI + HCI CH,CH₂OH + PBr, CH,CH,Br + P(OH), + HBr 1. Reaction with HBr with 2 Pentanol 2.Reaction with HI with 2 pentanol © Byjus.com 3.Reaction with HCI+ZnCl,, with 2 pentanol (Zncl2 is catalyst no role) 4.Reaction with SOCI,, with 2 Pentanol 5.Reaction with PBr; or PCl, with 2 pentanolarrow_forward3. Is 2-methyl-2-propanol a primary, secondary, or tertiary alcohol? Write out the structures of 2-methyl-2-propanol and also any oxidation products of 2- methyl-2- propanol. If there is more than one oxidation product, give the structure of each of the products. 4. 2-Propanol is the IUPAC systematic name of this alcohol. It has a common name by which it is much better known (You'll see it in the grocery store or pharmacy). Give that common name 5. Aldehydes can be synthesized by the oxidation of. Please choose from below choices A. Primary alcohols B. Secondary alcohols C. Organic acids D. Inorganic acids 6. Tertiary alcohol Can undergo oxidation. yes or no. ? If yes then answer the product.arrow_forward
- Finish the reactions hand written pleasearrow_forwardPart A Identify each alcohol as primary, secondary, or tertiary Drag the appropriate items to their respective bins. CH₂ H₂C- -C-OH HO CH₂ Primary Он OH CH₂ OH CCH₂OH CH₂ сн Secondary Tertiary Reset Help CH,CH₂ (CH)CHCH,OH CH,CH,CH,CCH, CHOH CH₂ Different types of alcohol groups Alcohol and its reaction: 8. Combing two alcohol molecules below and completing the reaction with Product .( Hint Reaction called etherification as ether is formed and name the ether once you complete the reaction. Hint.: R-O-H+H-O-RR-O-R Do the reaction: CH₂OH + CH₂OH---→ + H-O-H 9. Write the reaction of formation of alcohol from alkene by adding water: Addition reaction also called hydration reaction as we are adding water which occur always in presence of acid Hint: Break the double bond and add H and OH if symmetrical then add anywhere if unsymmetrical then follow Markovnikov rule H should go to that double bone carbon which has more hydrogen CH2=CH2 + H₂O-→arrow_forwardComplete the reaction hand written pleasearrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning





