
EBK ORGANIC CHEMISTRY
7th Edition
ISBN: 9780133556186
Author: Bruice
Publisher: VST
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 11.4, Problem 13P
(a)
Interpretation Introduction
Interpretation:
The reason should be explained for difficulty in the synthesis of unsymmetrical ether.
Concept introduction:
Ethers:
An oxygen atom connected to two alkyl or aryl groups is called as ether. The general formula of ether is
Ethyl alcohol is reaction with acid which yields diethyl ether which is shown below.
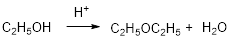
(b)
Interpretation Introduction
Interpretation:
The ethyl propyl ether should be synthesized.
Concept introduction:
Ethers:
An oxygen atom connected to two alkyl or aryl groups is called as ether. The general formula of ether is
Ethyl alcohol is reaction with acid which yields diethyl ether which is shown below.
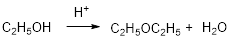
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw the major product of this reaction. Ignore inorganic byproducts.
Assume that the water side product is continuously removed to drive the reaction toward products.
O
CH3CH2NH2, TSOH
Select to Draw
>
Predict the major organic product(s) for the following reaction.
Predict the major organic product(s) for the following reactions.
Chapter 11 Solutions
EBK ORGANIC CHEMISTRY
Ch. 11.1 - Why are NH3 and CH3NH2 no longer nucleophiles when...Ch. 11.1 - Prob. 2PCh. 11.1 - Prob. 5PCh. 11.2 - Prob. 7PCh. 11.3 - Prob. 9PCh. 11.3 - Show how 1-propanol can be converted into the...Ch. 11.4 - Which of the following alcohols dehydrates the...Ch. 11.4 - Prob. 12PCh. 11.4 - Prob. 13PCh. 11.4 - Propose a mechanism for each of the following...
Ch. 11.4 - Draw the product of each of the following...Ch. 11.4 - Explain why the following alcohols, when heated...Ch. 11.4 - What stereoisomers are formed in the following...Ch. 11.4 - Prob. 18PCh. 11.4 - What alcohol would you treat with phosphorus...Ch. 11.5 - Prob. 20PCh. 11.6 - Prob. 22PCh. 11.7 - Prob. 24PCh. 11.7 - Would you expect the reactivity of a five-membered...Ch. 11.7 - Prob. 26PCh. 11.7 - What products are obtained from the reaction of...Ch. 11.7 - Prob. 28PCh. 11.7 - Prob. 29PCh. 11.7 - Prob. 30PCh. 11.8 - Prob. 31PCh. 11.8 - Prob. 32PCh. 11.8 - How do the major products obtained from...Ch. 11.8 - Explain why the two arene oxides in Problem 38...Ch. 11.8 - Three arene oxides can be obtained from...Ch. 11.9 - Explain why the half-life (the time it takes for...Ch. 11.10 - Prob. 38PCh. 11.10 - Prob. 39PCh. 11.10 - Prob. 40PCh. 11.10 - Prob. 41PCh. 11.10 - Prob. 42PCh. 11.11 - Using an alkyl halide and a thiol as starting...Ch. 11.11 - The following three nitrogen mustards were studied...Ch. 11.11 - Why is melphalan a good cancer drug?Ch. 11.11 - Prob. 47PCh. 11 - Prob. 48PCh. 11 - Which compound is more likely to be carcinogenic?Ch. 11 - Prob. 50PCh. 11 - Prob. 51PCh. 11 - Write the appropriate reagent over each arrow.Ch. 11 - What alkenes would you expect to be obtained from...Ch. 11 - Prob. 54PCh. 11 - When heated with H2SO4, both...Ch. 11 - What is the major product obtained from the...Ch. 11 - When deuterated phenanthrene oxide undergoes a...Ch. 11 - An unknown alcohol with a molecular formula of...Ch. 11 - Prob. 59PCh. 11 - Prob. 60PCh. 11 - Propose a mechanism for the following reaction:Ch. 11 - What product would be formed if the four-membered...Ch. 11 - Which of the following ethers would be obtained in...Ch. 11 - Using the given starting material any necessary...Ch. 11 - Prob. 65PCh. 11 - When 3-methyl-2-butanol is heated with...Ch. 11 - Propose a mechanism for each of the following...Ch. 11 - How could you synthesize isopropyl propyl ether,...Ch. 11 - When the following seven-membered ring alcohol is...Ch. 11 - Ethylene oxide reacts readily with HO because of...Ch. 11 - Prob. 71PCh. 11 - Propose a mechanism for each of the following...Ch. 11 - Explain why the acid-catalyzed dehydration of an...Ch. 11 - Triethylene glycol is one of the products obtained...Ch. 11 - Prob. 75PCh. 11 - Prob. 76PCh. 11 - When ethyl ether is heated with excess HI for...Ch. 11 - Propose a mechanism for the following reaction:Ch. 11 - Prob. 79PCh. 11 - An ion with a positively charged nitrogen atom in...Ch. 11 - Propose a mechanism for each of the following...Ch. 11 - Prob. 82PCh. 11 - The following reaction takes place several times...Ch. 11 - A vicinal diol has OH groups on adjacent carbons....Ch. 11 - Prob. 85PCh. 11 - Prob. 86PCh. 11 - Two stereoisomers are obtained from the reaction...Ch. 11 - Propose a mechanism for each or the following...Ch. 11 - Triethylenemelamine (TEM) is an antitumor agent....
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Provide the complete mechanism for the reactions below. You must include appropriate arrows,intermediates, and formal charges.arrow_forwardIndicate the products obtained by reacting fluorobenzene with a sulfonitric mixture.arrow_forwardIf I have 1-bromopropene, to obtain compound A, I have to add NaOH and another compound. Indicate which compound that would be. C6H5 CH3arrow_forward
- If I have 1-bromopropene and I want to obtain (1,1-dipropoxyethyl)benzene, indicate the compound that I should add in addition to NaOH.arrow_forwardDraw the major product of this reaction. Ignore inorganic byproducts. Ο HSCH2CH2CH2SH, BF3 Select to Draw I Submitarrow_forwardFeedback (7/10) Draw the major product of this reaction. Ignore inorganic byproducts. Assume that the water side product is continuously removed to drive the reaction toward products. Incorrect, 3 attempts remaining Ο (CH3CH2)2NH, TSOH Select to Draw V N. 87% Retryarrow_forward
- If I want to obtain (1,1-dipropoxyethyl)benzene from 1-bromopropene, indicate the product that I have to add in addition to NaOH.arrow_forwardIndicate the products obtained when fluorobenzene reacts with a sulfonitric acid mixture (HNO3 + H2SO4). Indicate the majority if necessary.arrow_forwardIndicate the products obtained when chlorobenzene acid reacts with a sulfonitric acid mixture (HNO3 + H2SO4). Indicate the majority if necessary.arrow_forward
- Indicate the products obtained by reacting benzenesulfonic acid with a sulfonitric acid mixture (HNO3 + H2SO4). Indicate the majority if necessary.arrow_forwardIndicate the products obtained by reacting ethylbenzene with a sulfonitric acid mixture (HNO3 + H2SO4). Indicate the majority if necessary.arrow_forwardIndicate the products obtained when tert-butylbenzene reacts with a sulfonitric acid mixture (HNO3 + H2SO4). Indicate the majority if necessary.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning