
Principles of General, Organic, Biological Chemistry
2nd Edition
ISBN: 9780073511191
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 11, Problem 11.70AP
(a)
Interpretation Introduction
Interpretation:
The name for the given compound has to be assigned.
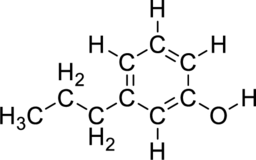
Concept Introduction:
Nomenclature of Benzene derivatives:
- First identify the number of substituent present in the benzene ring.
- If benzene ring has only one substituent, then name the substituent and add this word to benzene.
- If benzene ring has two substituent, then prefix ortho(1,2), meta(1,3), para(1,4) are used to locate the position of the substituent and ortho, meta, para are generally represented as o, m, p respectively.
- If benzene ring has three or more substituent, then the numbering should be given as the lowest possible number around the ring.
- Substituent names should be assigned in alphabetical order.
- If substituent is part of common roots, then the name should be assigned as their derivatives.
(b)
Interpretation Introduction
Interpretation:
The name for the given compound has to be assigned.
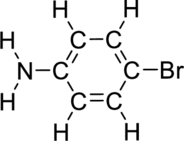
Concept Introduction:
Refer to part (a).
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Help fix my arrows please
no Ai walkthroughs
Given the data attached, provide a drawing of the corresponding structure.
Chapter 11 Solutions
Principles of General, Organic, Biological Chemistry
Ch. 11.1 - Convert each condensed structure to a complete...Ch. 11.1 - Determine whether each molecular formula...Ch. 11.1 - Give the molecular formula for each of the...Ch. 11.2 - Give the IUPAC name for each alkene. a. (CH3CH2)2C...Ch. 11.2 - Prob. 11.5PCh. 11.2 - Give the structure corresponding to each name. a....Ch. 11.3 - Prob. 11.7PCh. 11.3 - Bombykol is secreted by the female silkworm moth...Ch. 11.3 - Prob. 11.9PCh. 11.3 - Prob. 11.10P
Ch. 11.3 - Prob. 11.11PCh. 11.5 - Prob. 11.12PCh. 11.5 - Prob. 11.13PCh. 11.6 - Prob. 11.14PCh. 11.6 - Prob. 11.15PCh. 11.7 - Prob. 11.16PCh. 11.7 - Prob. 11.17PCh. 11.9 - Prob. 11.18PCh. 11.9 - Draw the structure corresponding to each name. a....Ch. 11.10 - Prob. 11.20PCh. 11.10 - Prob. 11.21PCh. 11.10 - Prob. 11.22PCh. 11 - Prob. 11.23UKCCh. 11 - Prob. 11.24UKCCh. 11 - Prob. 11.25UKCCh. 11 - Prob. 11.26UKCCh. 11 - Answer the following questions about compound A,...Ch. 11 - Prob. 11.28UKCCh. 11 - Prob. 11.29UKCCh. 11 - Prob. 11.30UKCCh. 11 - Prob. 11.31UKCCh. 11 - Prob. 11.32UKCCh. 11 - Prob. 11.33APCh. 11 - Prob. 11.34APCh. 11 - Prob. 11.35APCh. 11 - Prob. 11.36APCh. 11 - Prob. 11.37APCh. 11 - Falcarinol is a natural pesticide found in carrots...Ch. 11 - Prob. 11.39APCh. 11 - Prob. 11.40APCh. 11 - Prob. 11.41APCh. 11 - Prob. 11.42APCh. 11 - Prob. 11.43APCh. 11 - Give the structure corresponding to each IUPAC...Ch. 11 - Leukotriene C4 is a key compound that causes the...Ch. 11 - Prob. 11.46APCh. 11 - Prob. 11.47APCh. 11 - Prob. 11.48APCh. 11 - Prob. 11.49APCh. 11 - Prob. 11.50APCh. 11 - Prob. 11.51APCh. 11 - Prob. 11.52APCh. 11 - Prob. 11.53APCh. 11 - Prob. 11.54APCh. 11 - Prob. 11.55APCh. 11 - Prob. 11.56APCh. 11 - Prob. 11.57APCh. 11 - Draw the products formed in each reaction.Ch. 11 - Prob. 11.59APCh. 11 - Prob. 11.60APCh. 11 - Prob. 11.61APCh. 11 - Prob. 11.62APCh. 11 - Prob. 11.63APCh. 11 - Prob. 11.64APCh. 11 - Prob. 11.65APCh. 11 - Prob. 11.66APCh. 11 - Prob. 11.67APCh. 11 - Prob. 11.68APCh. 11 - Prob. 11.69APCh. 11 - Prob. 11.70APCh. 11 - Prob. 11.71APCh. 11 - Prob. 11.72APCh. 11 - Prob. 11.73APCh. 11 - Prob. 11.74APCh. 11 - Prob. 11.75APCh. 11 - Prob. 11.76APCh. 11 - Prob. 11.77APCh. 11 - Prob. 11.78APCh. 11 - Prob. 11.79APCh. 11 - Prob. 11.80APCh. 11 - Prob. 11.81APCh. 11 - Prob. 11.82APCh. 11 - Prob. 11.83APCh. 11 - Prob. 11.84APCh. 11 - Prob. 11.85APCh. 11 - Prob. 11.86APCh. 11 - Are cis-2-hexene and trans-3-hexene constitutional...Ch. 11 - Prob. 11.88CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- help 20arrow_forwardProvide the drawing of the unknown structure that corresponds with this data.arrow_forward20.44 The Diels-Alder reaction is not limited to making six-membered rings with only car- bon atoms. Predict the products of the following reactions that produce rings with atoms other than carbon in them. OCCH OCCH H (b) CH C(CH₂)s COOCH མ་ནས་བ (c) N=C H -0.X- (e) H C=N COOCHS + CH2=CHCH₂ →→arrow_forward
- 3) Draw a detailed mechanism and predict the product of the reaction shown? 1) EtMgBr 2) H3O+arrow_forwardHow to draw the mechanism for this reaction?arrow_forward> H₂C=C-CH2-CH3 B. H₂O Pt C. + H2 + H₂O H D. 16. Give the IUPAC name for each of the following: B. Cl Cl c. Cl Cl 17. Draw the line-angle formula for each of the following compounds: 1. phenol 2. 1,3-dichlorobenzene 3. 4-ethyltoluene < Previous Submit Assignment Next ▸arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co


Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning