
Concept explainers
(a)
Interpretation:
The molecular formula of the hydrocarbon has to be derived.
Concept Introduction:
The compounds that contain carbon and hydrogen atoms are known as hydrocarbon compounds. There are two classes of hydrocarbon compounds which are saturated and
Saturated hydrocarbon compound has only one type that is
(a)
Explanation of Solution
The number of moles of hydrocarbon is calculated by the formula shown below.
The mass of hydrocarbon is
The molar mass of hydrocarbon is
Substitute the values in the above formula.
The volume of
Thus, the number of moles of hydrogen occupied by
The moles of hydrogen per moles of hydrocarbon are calculated as shown below.
Thus, one mole of hydrocarbon reacts with two moles of hydrogen. This indicates the presence of two double or one triple bond in the hydrocarbon.
The mass percent of carbon is
The mass percent of hydrogen is
The mass of carbon is calculated as shown below.
The mass of hydrogen is calculated as shown below.
The number of moles of carbon is calculated by the formula shown below.
The mass of carbon is
The molar mass of carbon is
Substitute the values in the above formula.
The number of moles of hydrogen is calculated by the formula shown below.
The mass of hydrogen is
The molar mass of hydrogen n is
Substitute the values in the above formula.
The molar ratio of
Thus, the molecular formula of the hydrocarbon is
(b)
Interpretation:
The structural formula for two possible isomers has to be drawn.
Concept Introduction:
The molecules which have same molecular formula and same connectivity of atoms but different arrangement of atoms in space are known as stereoisomers. Stereocentre is the region or atom in a molecule due to which molecule is showing stereoisomerism. The interchange of groups at stereocentre generates stereoisomer.
(b)
Explanation of Solution
The molecular formula of the hydrocarbon is
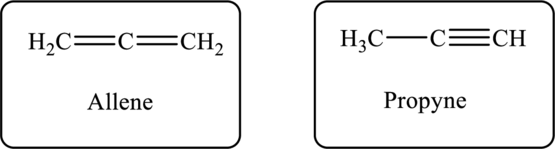
Figure 1
(c)
Interpretation:
The balanced chemical equation has to be used to show the bromination and hydrogenation reaction for one of the isomers.
Concept Introduction:
The addition of a bromine atom in the given compound is known as bromination. Bromination occurs through an electrophilic substitution reaction. Bromine atom acts as an electrophile which causes the formation of sigma bond in the reaction.
(c)
Explanation of Solution
Propyne undergoes bromination to form tetrabromo product. The balanced chemical equation is shown below.
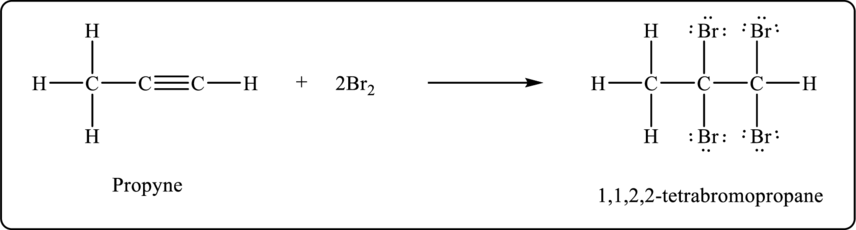
Figure 2
Propyne undergoes hydrogenation to form propane. The balanced chemical equation is shown below.
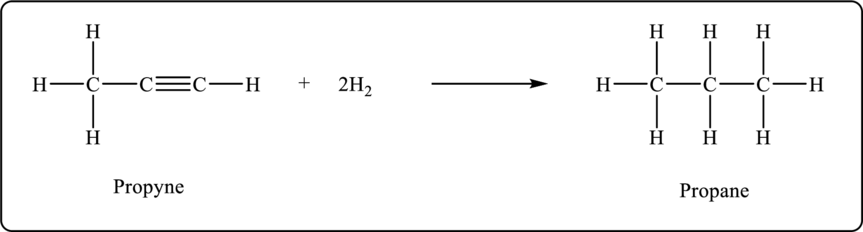
Figure 3
Want to see more full solutions like this?
Chapter 10 Solutions
Bundle: Chemistry: The Molecular Science, 5th, Loose-Leaf + OWLv2 with Quick Prep 24-Months Printed Access Card
- Please help me solve this reaction.arrow_forwardIndicate the products obtained by mixing 2,2-dimethylpropanal with acetaldehyde and sodium ethoxide in ethanol.arrow_forwardSynthesize 2-Ethyl-3-methyloxirane from dimethyl(propyl)sulfonium iodide using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forward
- Synthesize 2-Hydroxy-2-phenylacetonitrile from phenylmethanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forward
- If possible, please provide the formula of the compound 3,3-dimethylbut-2-enal.arrow_forwardSynthesize 1,4-dibromobenzene from acetanilide (N-phenylacetamide) using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardIndicate the products obtained by mixing (3-oxo-3-phenylpropyl)triphenylphosphonium bromide with sodium hydride.arrow_forward
- We mix N-ethyl-2-hexanamine with excess methyl iodide and followed by heating with aqueous Ag2O. Indicate the major products obtained.arrow_forwardIndicate the products obtained by mixing acetophenone with iodine and NaOH.arrow_forwardIndicate the products obtained by mixing 2-Propanone and ethyllithium and performing a subsequent acid hydrolysis.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning




