
Concept explainers
a)
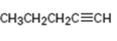
Interpretation:
How to synthesize 1-pentyne from acetylene using any alkyl halide with four or fewer number of carbons is to be shown.
Concept introduction:
Terminal
To state:
How to synthesize 1-pentyne from acetylene using any alkyl halide with four or fewer number of carbons.
b)
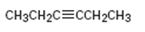
Interpretation:
How to synthesize 3-hexyne from acetylene using any alkyl halide with four or fewer number of carbons is to be shown.
Concept introduction:
Terminal alkynes can be converted into their alkynides by treating with NaNH3 in liquid NH3. The alkynides when treated with alkyl halides with the required number of carbons yield the higher alkyne needed. Acetylene has two acidic hydrogens. Both hydrogens can be replaced by alkyl groups through this process.
To state:
How to synthesize 3-hexyne from acetylene using any alkyl halide with four or fewer number of carbons.
c)
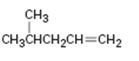
Interpretation:
How to synthesize 4-methyl-1-pentene from acetylene using any alkyl halide with four or fewer number of carbons is to be shown.
Concept introduction:
Terminal alkynes can be converted into their alkynides by treating with NaNH3 in liquid NH3. The alkynides when treated with alkyl halides with the required number of carbons yield the higher alkyne needed. The alkyne can be reduced to the corresponding
To state:
How to synthesize 4-methyl-1-pentene from acetylene using any alkyl halide with four or fewer number of carbons.
d)
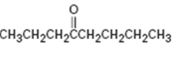
Interpretation:
How to synthesize 4-octanone from acetylene using any alkyl halide with four or fewer number of carbons is to be shown.
Concept introduction:
Terminal alkynes can be converted into their alkynides by treating with NaNH3 in liquid NH3. The alkynides when treated with alkyl halides with the required number of carbons yield the higher alkyne needed. The alkyne undergoes hydration when treated with dilute H3SO4 in the presence of HgSO4 to yield an enol which tautomerizes to a
To state:
How to synthesize 4-octanone from acetylene using any alkyl halide with four or fewer number of carbons.
e)
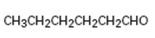
Interpretation:
How to synthesize hexanal from acetylene using any alkyl halide with four or fewer number of carbons is to be shown.
Concept introduction:
Terminal alkynes can be converted into their alkynides by treating with NaNH3 in liquid NH3. The alkynides when treated with alkyl halides with the required number of carbons yield the higher alkynes needed. The alkynes yield enols with OH on terminal carbon in hydroboration-oxidation reaction which tautomerize to yield
To state:
How to synthesize hexanal from acetylene using any alkyl halide with four or fewer number of carbons.
Trending nowThis is a popular solution!

Chapter 9 Solutions
Student Value Bundle: Organic Chemistry, + OWLv2 with Student Solutions Manual eBook, 4 terms (24 months) Printed Access Card (NEW!!)
- (11pts total) Consider the arrows pointing at three different carbon-carbon bonds in the molecule depicted below. Bond B Bond A Bond C a. (2pts) Which bond between A-C is weakest? Which is strongest? Place answers in appropriate boxes. Weakest Bond Strongest Bond b. (4pts) Consider the relative stability of all cleavage products that form when bonds A, B, AND C are homolytically cleaved/broken. Hint: cleavage products of bonds A, B, and C are all carbon radicals. i. Which ONE cleavage product is the most stable? A condensed or bond line representation is fine. ii. Which ONE cleavage product is the least stable? A condensed or bond line representation is fine. c. (5pts) Use principles discussed in lecture, supported by relevant structures, to succinctly explain the why your part b (i) radical is more stable than your part b(ii) radical. Written explanation can be no more than one-two succinct sentence(s)!arrow_forward. 3°C with TH 12. (10pts total) Provide the major product for each reaction depicted below. If no reaction occurs write NR. Assume heat dissipation is carefully controlled in the fluorine reaction. 3H 24 total (30) 24 21 2h • 6H total ● 8H total 34 래 Br2 hv major product will be most Substituted 12 hv Br NR I too weak of a participate in P-1 F₂ hv Statistically most favored product will be major = most subst = thermo favored hydrogen atom abstractor to LL Farrow_forwardFive chemistry project topic that does not involve practicalarrow_forward
- Please correct answer and don't used hand raitingarrow_forwardQ2. Consider the hydrogenation of ethylene C2H4 + H2 = C2H6 The heats of combustion and molar entropies for the three gases at 298 K are given by: C2H4 C2H6 H2 AH comb/kJ mol¹ -1395 -1550 -243 Sº / J K¹ mol-1 220.7 230.4 131.1 The average heat capacity change, ACP, for the reaction over the temperature range 298-1000 K is 10.9 J K¹ mol¹. Using these data, determine: (a) the standard enthalpy change at 800 K (b) the standard entropy change at 800 K (c) the equilibrium constant at 800 K.arrow_forward13. (11pts total) Consider the arrows pointing at three different carbon-carbon bonds in the molecule depicted below. Bond B Bond A Bond C a. (2pts) Which bond between A-C is weakest? Which is strongest? Place answers in appropriate boxes. Weakest Bond Strongest Bond b. (4pts) Consider the relative stability of all cleavage products that form when bonds A, B, AND C are homolytically cleaved/broken. Hint: cleavage products of bonds A, B, and C are all carbon radicals. i. Which ONE cleavage product is the most stable? A condensed or bond line representation is fine. ii. Which ONE cleavage product is the least stable? A condensed or bond line representation is fine. c. (5pts) Use principles discussed in lecture, supported by relevant structures, to succinctly explain the why your part b (i) radical is more stable than your part b(ii) radical. Written explanation can be no more than one-two succinct sentence(s)! Googlearrow_forward
- Print Last Name, First Name Initial Statifically more chances to abstract one of these 6H 11. (10pts total) Consider the radical chlorination of 1,3-diethylcyclohexane depicted below. 4 4th total • 6H total 래 • 4H total 21 total ZH 2H Statistical H < 3° C-H weakest - product abstraction here bund leads to thermo favored a) (6pts) How many unique mono-chlorinated products can be formed and what are the structures for the thermodynamically and statistically favored products? Product 6 Number of Unique Mono-Chlorinated Products Thermodynamically Favored Product Statistically Favored Product b) (4pts) Draw the arrow pushing mechanism for the FIRST propagation step (p-1) for the formation of the thermodynamically favored product. Only draw the p-1 step. You do not need to include lone pairs of electrons. No enthalpy calculation necessary H H-Cl Waterfoxarrow_forward10. (5pts) Provide the complete arrow pushing mechanism for the chemical transformation → depicted below Use proper curved arrow notation that explicitly illustrates all bonds being broken, and all bonds formed in the transformation. Also, be sure to include all lone pairs and formal charges on all atoms involved in the flow of electrons. CH3O II HA H CH3O-H H ①arrow_forwardDo the Lone Pairs get added bc its valence e's are a total of 6 for oxygen and that completes it or due to other reasons. How do we know the particular indication of such.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

