
ORGANIC CHEMISTRY BOOK& SG/SM
6th Edition
ISBN: 9781264094493
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9.8, Problem 11P
Draw the products formed when each alcohol undergoes dehydration with
a. 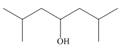 b.
b. 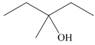 c.
c. 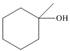
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Propose an efficient synthesis for the following transformation:
EN
The transformation above can be performed with some reagent or combination of the reagents listed below. Give the necessary
reagents in the correct order, as a string of letters (without spaces or punctuation, such as "EBF"). If there is more than one correct
solution, provide just one answer.
A. t-BuOK
B. Na2Cr2O7, H2SO4, H2O
C. NBS, heat
F. NaCN
D. MeOH
E. NaOH
G. MeONa
H. H2O
I. 1) O3; 2) DMS
Stereochemistry
Identifying the enantiomer of a simple organic molecule
1/5
Check the box under each structure in the table that is an enantiomer of the molecule shown below. If none of them are, check the none of t
above box under the table.
Br
ま
HO
H
0
Molecule 1
Molecule 2
Molecule 3
OH
H
Br
H
H"
Br
OH
Br
Molecule 4
Br
H
OH
+ +
OH
Molecule 5
Br
H
OH
none of the above
Molecule 6
Br
H...
OH
Please answer the questions and provide detailed explanations.
Chapter 9 Solutions
ORGANIC CHEMISTRY BOOK& SG/SM
Ch. 9.1 - Problem 9.1 Label each ether and alcohol in...Ch. 9.3 - Give the IUPAC name for each compound.Ch. 9.3 - Problem 9.3 Give the structure corresponding to...Ch. 9.3 - Name each of the following ethers.Ch. 9.3 - Name each epoxide.
a. (two ways) b. c. (two...Ch. 9.6 - Problem 9.8 Draw the organic product of each...Ch. 9.6 - Prob. 9PCh. 9.6 - Problem 9.10 Draw the products of each reaction.
...Ch. 9.8 - Problem 9.11 Draw the products formed when each...Ch. 9.8 - Prob. 12P
Ch. 9.11 - Problem 9.18 Draw the products of each reaction,...Ch. 9.11 - Problem 9.19 What is the major product formed...Ch. 9.12 - Prob. 19PCh. 9.12 - Prob. 20PCh. 9.12 - Problem 9.22 Draw the organic products formed in...Ch. 9.12 - Problem 9.23 Draw two steps to convert into each...Ch. 9.13 - Prob. 23PCh. 9.13 - Problem 9.25 Draw the products of each reaction,...Ch. 9.13 - Draw the products formed when (S)-butan-2-ol is...Ch. 9.13 - Draw the product formed when (CH3)2CHOH is treated...Ch. 9.14 - What alkyl halides are formed when each ether is...Ch. 9.14 - Explain why the treatment of anisole with HBr...Ch. 9.15 - Name each thiol.
a. b.
Ch. 9.15 - Draw the product of each reaction. ac b.d.Ch. 9.15 - Give the IUPAC name for each sulfide.
a. b.
Ch. 9.15 - Draw the product of each reaction.
a. b.
Ch. 9.16 - Prob. 33PCh. 9.16 - The cis and trans isomers of 2, 3-dimethyloxirane...Ch. 9.16 - Problem 9.36 Draw the product of each...Ch. 9 - 9.37 Name each compound depicted in the...Ch. 9 - Answer each question using the ball-and-stick...Ch. 9 - Prob. 38PCh. 9 - 9.40 Give IUPAC name for each...Ch. 9 - Prob. 40PCh. 9 - Prob. 41PCh. 9 - 9.46 What alkenes are formed when each alcohol is...Ch. 9 - Prob. 46PCh. 9 - Prob. 51PCh. 9 - 9.57 Draw a stepwise, detailed mechanism for the...Ch. 9 - Prob. 53PCh. 9 - 9.59 Draw two different routes to each of the...Ch. 9 - Prob. 55PCh. 9 -
9.61 Draw the products formed when each ether is...Ch. 9 - 9.62 Draw a stepwise mechanism for each...Ch. 9 - Draw a stepwise, detailed mechanism for the...Ch. 9 - Prob. 59PCh. 9 - Draw the products of each reaction. a.c. b.d.Ch. 9 - Prob. 64PCh. 9 - Prob. 75P
Additional Science Textbook Solutions
Find more solutions based on key concepts
How does the removal of hydrogen atoms from nutrient molecules result in a loss of energy from the nutrient mol...
SEELEY'S ANATOMY+PHYSIOLOGY
On what molecule does the anticodon appear? Explain the role of this molecule in protein synthesis.
Human Physiology: An Integrated Approach (8th Edition)
Identify each of the following reproductive barriers as prezygotic or postzygotic. a. One lilac species lives o...
Campbell Essential Biology with Physiology (5th Edition)
Give the IUPAC name for each compound.
Organic Chemistry
2. Define equilibrium population. Outline the conditions that must be met for a population to stay in genetic e...
Biology: Life on Earth (11th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Question 16 0/1 pts Choose the correct option for the following cycloaddition reaction. C CF3 CF3 CF3 CF3 The reaction is suprafacial/surafacial and forbidden The reaction is antarafacial/antarafacial and forbidden The reaction is antarafacial/antarafacial and allowed The reaction is suprafacial/surafacial and allowedarrow_forward1. Give the structures of the products obtained when the following are heated. Include stereochemistry where relevant. A NO2 + NO2 B + C N=C CEN + { 2. Which compounds would you heat together in order to synthesize the following?arrow_forwardExplain how myo-inositol is different from D-chiro-inositol. use scholarly sources and please hyperlink.arrow_forward
- What is the molarisuty of a 0.396 m glucose solution if its density is 1.16 g/mL? MM glucose 180.2 /mol.arrow_forwardProvide the proper IUPAC or common name for the following compound. Dashes, commas, and spaces must be used correctly. Br ......Im OHarrow_forwardCan you please help me solve this problems. The top one is just drawing out the skeletal correct and then the bottom one is just very confusing to me and its quite small in the images. Can you enlarge it and explain it to me please. Thank You much (ME EX1) Prblm #33arrow_forward
- I'm trying to memorize VESPR Shapes to solve problems like those. I need help making circles like the second image in blue or using an x- and y-axis plane to memorize these and solve those types of problems, especially the ones given in the top/first image (180, 120, 109.5). Can you help me with this? or is their any other efficient method do soarrow_forwardCan you please explain this problems to me? I'm very confused about it. Please provide a detailed, step-by-step explanation for me! (ME EX1) Prblm 27arrow_forwardQuestion 6 of 7 (1 point) | Question Attempt: 1 of 1 = 1 ✓2 ✓ 3 ✓ 4 ✓ 5 6 ✓ 7 This organic molecule is dissolved in a basic aqueous solution: Jen ✓ ? A short time later sensitive infrared spectroscopy reveals the presence of a new C-OH stretch absorption. That is, must now be a new molecule present with at least one C- OH bond. there 18 In the drawing area below, show the detailed mechanism that could convert the molecule above into the new molecule Ar © + Click and drag to start drawing a structure. Add/Remove step Click and drawing Save For Later Submit Assignmentarrow_forward
- Can you please explain this problem to me? I'm very confused about it. Please provide a detailed, step-by-step explanation for me! (ME EX1) Prblm 22arrow_forwardCan you please explain this problems to me? I'm very confused about it. Please provide a detailed, step-by-step explanation for me! (ME EX1) Prblm 30arrow_forwardThis organic molecule is dissolved in a basic aqueous solution: O ? olo RET A short time later sensitive infrared spectroscopy reveals the presence of a new C-OH stretch absorption. That is, there Ar must now be a new molecule present with at least one C - OH bond. In the drawing area below, show the detailed mechanism that could convert the molecule above into the new molecule. $ Add/Remove steparrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY