
Organic Chemistry
4th Edition
ISBN: 9780073402772
Author: Janice G. Smith
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 9.79P
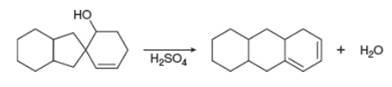
Draw a stepwise, detailed mechanism for the following reaction.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw the mechanism to make the alcohol 2-hexanol.
Draw the Mechanism to make the alcohol 1-hexanol.
Draw the mechanism for the formation of diol by starting with 1-pentanal in...
basic conditions
then
acidic conditions
then draw the mechanism for the formation of a carboxylic acid from your product.
Identify each chiral carbon as either R or S. Identify the overall carbohydrates as L or D
Chapter 9 Solutions
Organic Chemistry
Ch. 9 - Draw all constitutional isomers having molecular...Ch. 9 - Prob. 9.2PCh. 9 - Give the IUPAC name for each compound.Ch. 9 - Problem 9.3 Give the structure corresponding to...Ch. 9 - Prob. 9.5PCh. 9 - Name CH33COCH3, a gasoline additive commonly...Ch. 9 - Name each epoxide.
a. (two ways) b. c. (two...Ch. 9 - Problem 9.6 Rank the following compounds in order...Ch. 9 - Which mechanism is favored by the use of crown...Ch. 9 - Draw the organic product of each reaction.
Ch. 9 - Prob. 9.11PCh. 9 - Prob. 9.12PCh. 9 - Prob. 9.13PCh. 9 - Prob. 9.14PCh. 9 - Problem 9.13 Draw the structure of each...Ch. 9 - What other alkene is also formed along with Y in...Ch. 9 - Prob. 9.17PCh. 9 - Explain how the reaction of CH32CHCHCl CH3 with...Ch. 9 - Prob. 9.19PCh. 9 - Draw the products of each reaction, indicating the...Ch. 9 - Problem 9.19 What is the major product formed...Ch. 9 - Prob. 9.22PCh. 9 - Prob. 9.23PCh. 9 - Problem 9.22 Draw the organic products formed in...Ch. 9 - Problem 9.23 Draw two steps to convert into each...Ch. 9 - Draw the products of each reaction, and indicate...Ch. 9 - Draw the products of each reaction, and include...Ch. 9 - Draw the products formed when (S)-butan-2-ol is...Ch. 9 - Prob. 9.29PCh. 9 - Prob. 9.30PCh. 9 - Explain why the treatment of anisole with HBr...Ch. 9 - Prob. 9.32PCh. 9 - The cis and trans isomers of 2, 3-dimethyloxirane...Ch. 9 - Problem 9.36 Draw the product of each...Ch. 9 - 9.37 Name each compound depicted in the...Ch. 9 - Answer each question using the ball-and-stick...Ch. 9 - Prob. 9.37PCh. 9 - a. Draw the structure of 1, 2, and 3 alcohol with...Ch. 9 - Give IUPAC name for each alcohol. a....Ch. 9 - Name each ether and epoxide. a.b.c.d.Ch. 9 - Prob. 9.41PCh. 9 - Draw the eight constitutional isomers with...Ch. 9 - Rank each group of compounds in order of: a....Ch. 9 - Prob. 9.44PCh. 9 - 9.44 Why is the boiling point of higher than...Ch. 9 - Draw the organic products formed when CH3CH2CH2OH...Ch. 9 - Draw the organic products formed when 1-methyl...Ch. 9 - What alkenes are formed when each alcohol is...Ch. 9 - Prob. 9.49PCh. 9 - Prob. 9.50PCh. 9 - 9.48 Draw the products of each reaction and...Ch. 9 - Prob. 9.52PCh. 9 - 9.52 Draw a stepwise mechanism for the following...Ch. 9 - 9.53 Although alcohol V gives a single alkene W...Ch. 9 - Prob. 9.55PCh. 9 - Prob. 9.56PCh. 9 - Prob. 9.57PCh. 9 - When CH3CH2CH2CH2OH is treated with H2SO4 NaBr,...Ch. 9 - 9.57 Draw a stepwise, detailed mechanism for the...Ch. 9 - Prob. 9.60PCh. 9 - Draw two different routes to each of the following...Ch. 9 - Prob. 9.62PCh. 9 - Prob. 9.63PCh. 9 - 9.62 Draw a stepwise mechanism for each...Ch. 9 - Draw a stepwise, detailed mechanism for the...Ch. 9 - Prob. 9.66PCh. 9 - Draw the products of each reaction. a.c. b.d.Ch. 9 - When each halohydrin is treated with, a product of...Ch. 9 - Prob. 9.69PCh. 9 - Devise a stepwise mechanism for the following...Ch. 9 - Draw the products of each reaction, and indicate...Ch. 9 - Prepare each compound from CH3CH2CH2CH2OH. More...Ch. 9 - Prepare each compound from cyclopentanol. More...Ch. 9 - 9.72 Identify the reagents (a–h) needed to carry...Ch. 9 - Prob. 9.75PCh. 9 - 9.74 Treatment of with affords compound A and ....Ch. 9 - Prob. 9.77PCh. 9 - Prob. 9.78PCh. 9 - 9.77 Draw a stepwise, detailed mechanism for the...Ch. 9 - Dehydration of 1, 2, 2-trimethylcyclohexanol with...Ch. 9 - Prob. 9.81PCh. 9 - 9.80 Draw a stepwise mechanism for the following...Ch. 9 -
9.81 Aziridines are heterocycles that contain an...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Label each statement about the polynucleotide ATGGCG as true or false. The polynucleotide has six nucleotides. ...
General, Organic, and Biological Chemistry - 4th edition
True or false? Some trails are considered vestigial because they existed long ago.
Biological Science (6th Edition)
45. Calculate the mass of nitrogen dissolved at room temperature in an 80.0-L home aquarium. Assume a total pre...
Chemistry: Structure and Properties (2nd Edition)
How could you separate a mixture of the following compounds? The reagents available to you are water, either, 1...
Organic Chemistry (8th Edition)
An obese 55-year-old woman consults her physician about minor chest pains during exercise. Explain the physicia...
Biology: Life on Earth with Physiology (11th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Ethers can be formed via acid-catalyzed acetal formation. Draw the mechanism for the molecule below and ethanol.arrow_forwardHOCH, H HO CH-OH OH H OH 11 CH₂OH F II OH H H 0 + H OHarrow_forwardDraw the mechanism for the formation of diol by starting with one pen and all in... basic conditions then acidic conditions then draw the mechanism for the formation of a carboxylic acid from your product.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY