
An aqueous slurry at 30°C containing 20.0 wt% solids is fed to an evaporator in which enough w ater is vaporized at 1 atm to produce a product slurry containing 35.0 wt% solids. Heat is supplied to the evaporator by feeding saturated steam at 2.6 bar absolute into a coil immersed in the liquid. The steam condenses in the coil, and the slurry boils at the normal boiling point of pure water. The heat capacity of the solids may be taken to be half that of liquid water.
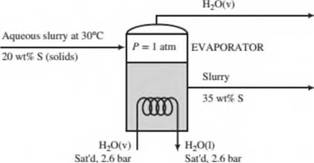
- Calculate the required steam feed rate (kg/h) for a slurry feed rate of 1.00 × 103 kg/h.
- Vapor recompression is often used in the operation of an evaporator. Suppose that the vapor (steam) generated in the evaporator described above is compressed to 2.6 bar and simultaneously heated to the saturation temperature at 2.6 bar, so that no condensation occurs. The compressed steam and additional saturated steam at 2.6 bar arc then fed to the evaporator coil, in which isobaric condensation occurs. How much additional steam is required?
- What more would you need to know to determine whether or not vapor recompression is economically advantageous in this process?
Trending nowThis is a popular solution!
Learn your wayIncludes step-by-step video

Chapter 8 Solutions
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
Additional Science Textbook Solutions
Introduction To Programming Using Visual Basic (11th Edition)
Starting Out with Java: From Control Structures through Data Structures (4th Edition) (What's New in Computer Science)
INTERNATIONAL EDITION---Engineering Mechanics: Statics, 14th edition (SI unit)
Modern Database Management
Management Information Systems: Managing The Digital Firm (16th Edition)
Starting Out with Programming Logic and Design (5th Edition) (What's New in Computer Science)
- Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. Select to Edit Arrows H H Select to Add Arrows > H CFCI: Select to Edit Arrows H Select to Edit Arrowsarrow_forwardShow work with explanation needed. don't give Ai generated solutionarrow_forwardShow work. don't give Ai generated solutionarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





