
Concept explainers
Interpretation:
The pH values after the addition of each proportion of the acid to the base is to be determined. Also, the titration curve needs to be drawn.
Concept introduction:
A strong acid or a strong base can completely dissociate into its ions when they are in an aqueous solution. Pyridine is a weak base while hydrochloric acid is a strong acid. When these two chemical species are reacted with each other, they form their ionic forms and an equilibrium state.
Explanation of Solution
Initial pH of the analyte solution; Pyridine is a weak base that forms equilibrium when dissolved in water. The equilibrium is as follows.
The molarity of pyridine is 0.1 M thus, the ICE table can be created as follows:
| Reaction | Pyridine base | Pyridine ion | OH- |
| Initial | 0.1 | 0 | 0 |
| Change | -x | +x | +x |
| Equilibrium | (0.1-x) | x | x |
Here,
Thus,
The pH of the solution will be:
Addition of
Total amount of base to be neutralized
Amount of acid added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Pyridine base | H+ | Pyridine ion |
| Initial | 0.0025 | 0 | 0 |
| Change | -0.0004 | -0.0004 | 0.0004 |
| Equilibrium | 0.0021 | 0 | 0.0004 |
In the Henderson-Hasselbalch equation, the pKa is used. Therefore, the pKa for pyridine need to be calculated using its Kb.
Applying the Henderson-Hasselbalch equation,
Addition of
Total amount of pyridine to be neutralized
Amount of acid added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Pyridine base | H+ | Pyridine ion |
| Initial | 0.0025 | 0 | 0 |
| Change | -0.0008 | -0.0008 | 0.0008 |
| Equilibrium | 0.0017 | 0 | 0.0008 |
Applying the Henderson-Hasselbalch equation,
Addition of
Total amount of pyridine to be neutralized
Amount of acid added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Pyridine base | H+ | Pyridine ion |
| Initial | 0.0025 | 0 | 0 |
| Change | -0.00125 | -0.00125 | 0.00125 |
| Equilibrium | 0.00125 | 0 | 0.00125 |
Applying the Henderson-Hasselbalch equation,
Addition of
Total amount of pyridine to be neutralized
Amount of acid added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Pyridine base | H+ | Pyridine base |
| Initial | 0.0025 | 0 | 0 |
| Change | -0.002 | -0.002 | 0.002 |
| Equilibrium | 0.0005 | 0 | 0.002 |
Applying the Henderson-Hasselbalch equation,
Addition of
Total amount of pyridine to be neutralized
Amount of acid added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Pyridine base | H+ | Pyridine ion |
| Initial | 0.0025 | 0 | 0 |
| Change | -0.0024 | -0.0024 | +0.0024 |
| Equilibrium | 0.0001 | 0 | 0.0024 |
Applying the Henderson-Hasselbalch equation,
Addition of
Total amount of pyridine to be neutralized
Amount of acid added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Pyridine base | H+ | Pyridine ion |
| Initial | 0.0025 | 0 | 0 |
| Change | -0.0025 | -0.0025 | +0.0025 |
| Equilibrium | 0.0000 | 0 | 0.0025 |
Concentration of base after addition of base
Concentration of conjugate ion
At this point, there is no excess acid or base. Therefore, the only possible reaction here is the dissociation of the conjugate acid of the pyridine base.
Thereafter, using the Ka value for pyridine, the amount of hydrogen ions in the solution can be determined to get the pH value at this point.
| Reaction | Pyridine base | Pyridine ion | H+ |
| Initial | 0.05 | 0 | 0 |
| Change | X | x | x |
| Equilibrium | (0.05-x) | x | x |
Then the pH can be calculated as follows:
The value of x can be neglected from denominator as the acid dissociation constant has very small value.
Thus,
Addition of 26.0 mL of the acid:
Total amount of pyridine to be neutralized
Amount of acid added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Pyridine base | H+ | Pyridine ion |
| Initial | 0.0025 | 0.0026 | 0 |
| Change | -0.0025 | -0.0025 | +0.0025 |
| Equilibrium | 0.0000 | 0.0001 | 0.0025 |
The pH of the solution only depends on the concentration of HCl thus,
The pH of the solution will be:
Addition of
Amount of acid added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Pyridine base | H+ | Pyridine ion |
| Initial | 0.0025 | 0.0028 | 0 |
| Change | -0.0025 | -0.0025 | +0.0025 |
| Equilibrium | 0 | 0.0003 | 0.0025 |
Here, the pH will only depend on the concentration of hydrogen ion.
Concentration of hydrogen ion
Addition of
Amount of ammonium added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Pyridine base | H+ | Pyridine ion |
| Initial | 0.0025 | 0.0003 | 0 |
| Change | -0.0025 | -0.0025 | +0.0025 |
| Equilibrium | 0 | 0.0005 | 0.0025 |
The pH will only depend on the concentration of hydrogen ion.
Concentration of hydrogen ion
Thus,
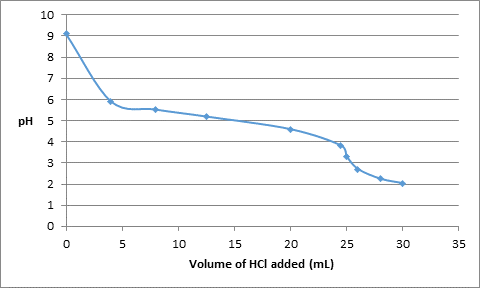
Thus, the data obtained from the calculations is as follows:
| Volume of HCl added (mL) | pH |
| 0 | 9.11 |
| 4.0 | 5.92 |
| 8.0 | 5.53 |
| 12.5 | 5.2 |
| 20.0 | 4.6 |
| 24.5 | 3.82 |
| 25.0 | 3.3 |
| 26.0 | 2.71 |
| 28.0 | 2.25 |
| 30.0 | 2.04 |
The titration curve can be represented as follows:
Want to see more full solutions like this?
Chapter 8 Solutions
EBK CHEMICAL PRINCIPLES
- Draw the product of the E2 reaction shown below. Include the correct stereochemistry. Ignore and inorganic byproducts.arrow_forwardDraw the major producrs of this SN1 reaction. Ignore any inorganic byproducts. Use a dash or wedge bond to indicate the sereochemistry of substituents on asymmetric centers where appllicable.arrow_forward5) Oxaloacetic Acid is an important intermediate in the biosynthesis of citric acid. Synthesize oxaloacetic acid using a mixed Claisen Condensation reaction with two different esters and a sodium ethoxide base. Give your answer as a scheme Hint 1: Your final acid product is producing using a decarboxylation reaction. Hint 2: Look up the structure of oxalic acid. HO all OH oxaloacetic acidarrow_forward
- 20. The Brusselator. This hypothetical system was first proposed by a group work- ing in Brussels [see Prigogine and Lefever (1968)] in connection with spatially nonuniform chemical patterns. Because certain steps involve trimolecular reac tions, it is not a model of any real chemical system but rather a prototype that has been studied extensively. The reaction steps are A-X. B+X-Y+D. 2X+ Y-3X, X-E. 305 It is assumed that concentrations of A, B, D, and E are kept artificially con stant so that only X and Y vary with time. (a) Show that if all rate constants are chosen appropriately, the equations de scribing a Brusselator are: dt A-(B+ 1)x + x²y, dy =Bx-x²y. diarrow_forwardProblem 3. Provide a mechanism for the following transformation: H₂SO A Me. Me Me Me Mearrow_forwardYou are trying to decide if there is a single reagent you can add that will make the following synthesis possible without any other major side products: xi 1. ☑ 2. H₂O хе i Draw the missing reagent X you think will make this synthesis work in the drawing area below. If there is no reagent that will make your desired product in good yield or without complications, just check the box under the drawing area and leave it blank. Click and drag to start drawing a structure. There is no reagent that will make this synthesis work without complications. : ☐ S ☐arrow_forward
- Predict the major products of this organic reaction: H OH 1. LiAlH4 2. H₂O ? Note: be sure you use dash and wedge bonds when necessary, for example to distinguish between major products with different stereochemistry. Click and drag to start drawing a structure. G C टेarrow_forwardFor each reaction below, decide if the first stable organic product that forms in solution will create a new C-C bond, and check the appropriate box. Next, for each reaction to which you answered "Yes" to in the table, draw this product in the drawing area below. Note for advanced students: for this problem, don't worry if you think this product will continue to react under the current conditions - just focus on the first stable product you expect to form in solution. NH2 CI MgCl ? Will the first product that forms in this reaction create a new CC bond? Yes No MgBr ? Will the first product that forms in this reaction create a new CC bond? Yes No G टेarrow_forwardFor each reaction below, decide if the first stable organic product that forms in solution will create a new CC bond, and check the appropriate box. Next, for each reaction to which you answered "Yes" to in the table, draw this product in the drawing area below. Note for advanced students: for this problem, don't worry if you think this product will continue to react under the current conditions - just focus on the first stable product you expect to form in solution. དྲ。 ✗MgBr ? O CI Will the first product that forms in this reaction create a new C-C bond? Yes No • ? Will the first product that forms in this reaction create a new CC bond? Yes No × : ☐ Xarrow_forward
- Predict the major products of this organic reaction: OH NaBH4 H ? CH3OH Note: be sure you use dash and wedge bonds when necessary, for example to distinguish between major products with different stereochemistry. Click and drag to start drawing a structure. ☐ : Sarrow_forwardPredict the major products of this organic reaction: 1. LIAIHA 2. H₂O ? Note: be sure you use dash and wedge bonds when necessary, for example to distinguish between major products with different stereochemistry. Click and drag to start drawing a structure. X : ☐arrow_forwardFor each reaction below, decide if the first stable organic product that forms in solution will create a new C - C bond, and check the appropriate box. Next, for each reaction to which you answered "Yes" to in the table, draw this product in the drawing area below. Note for advanced students: for this problem, don't worry if you think this product will continue to react under the current conditions - just focus on the first stable product you expect to form in solution. NH2 tu ? ? OH Will the first product that forms in this reaction create a new CC bond? Yes No Will the first product that forms in this reaction create a new CC bond? Yes No C $ ©arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





