
(a)
Interpretation:
The curved arrow notations showing all possible
Concept introduction:
Curved arrows are used to represent the movement of electrons in a reaction mechanism. The arrow starts on an electron-rich atom or an electron-rich region such as a pi bond. It ends on an electron poor atom when the movement results in the formation of a new sigma bond. If the result is the formation of a pi bond, the arrow ends in the region between the two atoms that form the bond.
A carbocation is a positively charged carbon atom that is electron-poor, two electrons short of an octet. It is unstable because it is a charged species.
A
A
The numbering in the shift label simply signifies that a hydride or a methyl group migrates from one carbon to an adjacent one. The numbering is not related to the root chain atom numbering.
Answer to Problem 7.36P
The possible
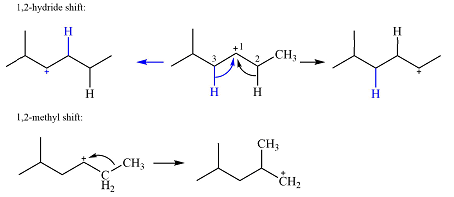
Explanation of Solution
The structure of the carbocation is
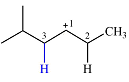
The charge is on the carbon numbered 1. There are two hydrogen atoms on the adjacent carbons C2 and C3. These are the ones that can shift to C1 in the two possible
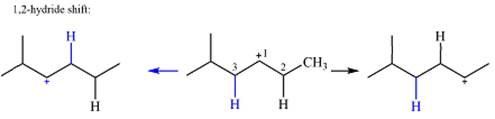
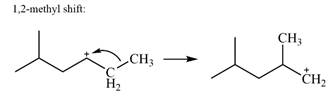
There is only one methyl group on the carbon adjacent to C1, attached to C2. Shifting of this methyl to C1 results in shifting of the charge to C2. Therefore, the
A hydride (
(b)
Interpretation:
The curved arrow notations showing all possible
Concept introduction:
Curved arrows are used to represent the movement of electrons in a reaction mechanism. The arrow starts on an electron-rich atom or an electron-rich region such as a pi bond. It ends on an electron poor atom when the movement results in the formation of a new sigma bond. If the result is the formation of a pi bond, the arrow ends in the region between the two atoms that form the bond.
A carbocation is a positively charged carbon atom that is electron-poor, two electrons short of an octet. It is unstable because it is a charged species.
A
A
The numbering in the shift label simply signifies that a hydride or a methyl group migrates from one carbon to an adjacent one. The numbering is not related to the root chain atom numbering.
Answer to Problem 7.36P
The possible
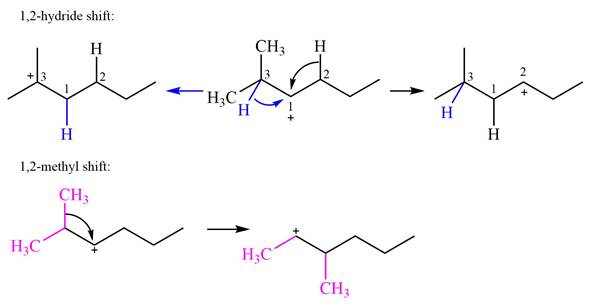
Explanation of Solution
The structure of the given carbocation is
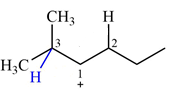
There are two hydrogen atoms on adjacent carbons C2 and C3 that can shift to the positively charged carbon C1 in two possible
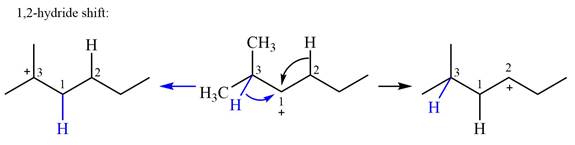
There are two methyl groups attached to a carbon adjacent to C1. Both are on the same carbon C3, therefore, shifting of either one will give the same product.
Therefore, the
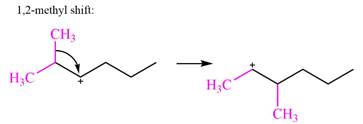
A hydride (
(c)
Interpretation:
The curved arrow notations showing all possible
Concept introduction:
Curved arrows are used to represent the movement of electrons in a reaction mechanism. The arrow starts on an electron-rich atom or an electron-rich region such as a pi bond. It ends on an electron poor atom when the movement results in the formation of a new sigma bond. If the result is the formation of a pi bond, the arrow ends in the region between the two atoms that form the bond.
A carbocation is a positively charged carbon atom that is electron-poor, two electrons short of an octet. It is unstable because it is a charged species.
A
A
The numbering in the shift label simply signifies that a hydride or a methyl group migrates from one carbon to an adjacent one. The numbering is not related to the root chain atom numbering.
Answer to Problem 7.36P
The possible
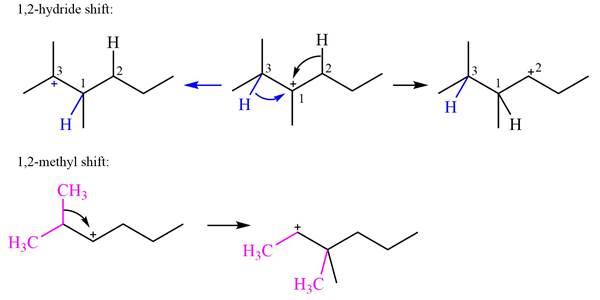
Explanation of Solution
The structure of the given carbocation is
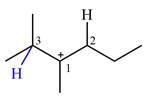
There are two hydrogen atoms on adjacent carbons C2 and C3 that can shift to the positively charged carbon C1 in two possible
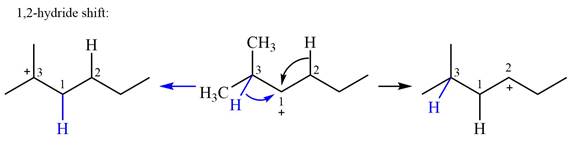
There are two methyl groups attached to a carbon adjacent to C1. Both are on the same carbon C3; therefore, shifting of either one will give the same product.
Therefore, the
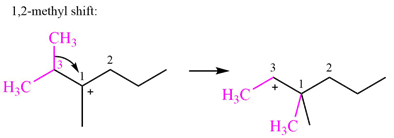
A hydride (
(d)
Interpretation:
The curved arrow notations showing all possible
Concept introduction:
Curved arrows are used to represent the movement of electrons in a reaction mechanism. The arrow starts on an electron-rich atom or an electron-rich region such as a pi bond. It ends on an electron poor atom when the movement results in the formation of a new sigma bond. If the result is the formation of a pi bond, the arrow ends in the region between the two atoms that form the bond.
A carbocation is a positively charged carbon atom that is electron-poor, two electrons short of an octet. It is unstable because it is a charged species.
A
A
The numbering in the shift label simply signifies that a hydride or a methyl group migrates from one carbon to an adjacent one. The numbering is not related to the root chain atom numbering.
Answer to Problem 7.36P
The possible
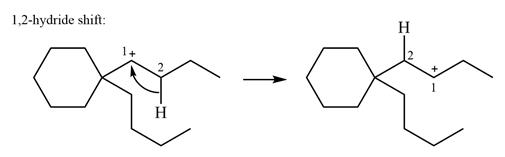
As there are no methyl groups on the carbon adjacent to the charge bearing carbon C1, a
Explanation of Solution
The structure of the given carbocation is
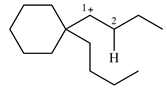
There is only one hydrogen atom on an adjacent carbon, C2, that can shift to the positively charged carbon C1 in a possible
Therefore, the
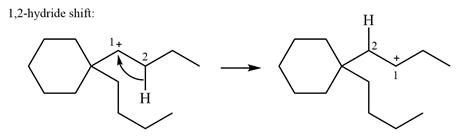
There are no methyl groups attached to the carbon adjacent to C1. Therefore, a
A hydride (
(e)
Interpretation:
The curved arrow notations showing all possible
Concept introduction:
Curved arrows are used to represent the movement of electrons in a reaction mechanism. The arrow starts on an electron-rich atom or an electron-rich region such as a pi bond. It ends on an electron poor atom when the movement results in the formation of a new sigma bond. If the result is the formation of a pi bond, the arrow ends in the region between the two atoms that form the bond.
A carbocation is a positively charged carbon atom that is electron-poor, two electrons short of an octet. It is unstable because it is a charged species.
A
A
The numbering in the shift label simply signifies that a hydride or a methyl group migrates from one carbon to an adjacent one. The numbering is not related to the root chain atom numbering.
Answer to Problem 7.36P
The possible
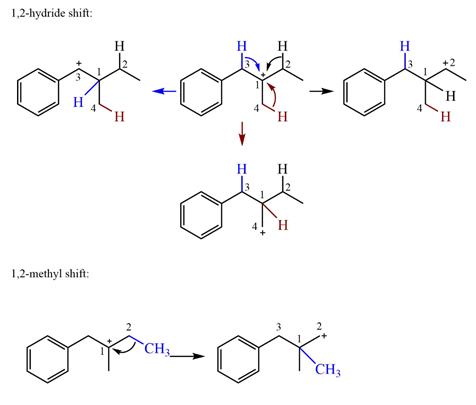
Explanation of Solution
The structure of the given carbocation is
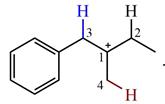
There are three hydrogen atoms on carbon atoms adjacent to the charge carrying carbon. They are on C2, C3, and C4.
Shifting of the hydride on C2 results in the charge shifting to C2, as shown in the product on the right.
Shifting of the hydride on C3 results in the charge shifting to C3, as shown in the product on the left.
Shifting of the hydride on C4 results in the charge shifting to C4, as shown in the product below the given carbocation.
Therefore, the
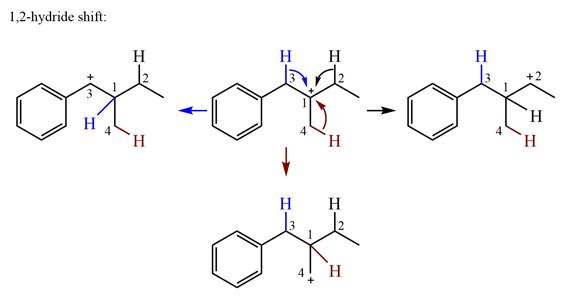
There is one methyl group attached to the carbon adjacent to C1. Shifting of the methyl group on C2 to C1 results in C1 becoming a tertiary carbon and the charge shifting to C2.
Therefore, the possible
A hydride (
(f)
Interpretation:
The curved arrow notations showing all possible
Concept introduction:
Curved arrows are used to represent the movement of electrons in a reaction mechanism. The arrow starts on an electron-rich atom or an electron-rich region such as a pi bond. It ends on an electron poor atom when the movement results in the formation of a new sigma bond. If the result is the formation of a pi bond, the arrow ends in the region between the two atoms that form the bond.
A carbocation is a positively charged carbon atom that is electron-poor, two electrons short of an octet. It is unstable because it is a charged species.
A
A
The numbering in the shift label simply signifies that a hydride or a methyl group migrates from one carbon to an adjacent one. The numbering is not related to the root chain atom numbering.
Answer to Problem 7.36P
The possible
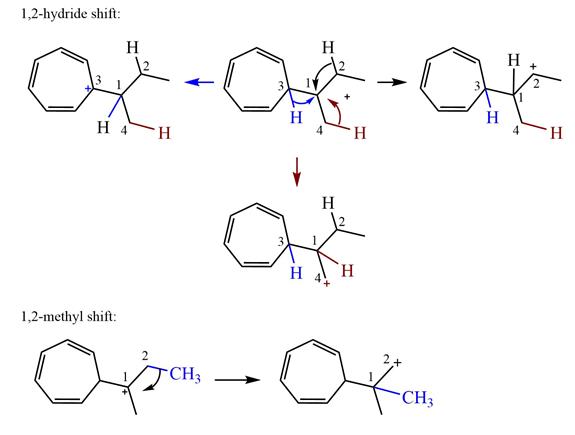
Explanation of Solution
The structure of the given carbocation is
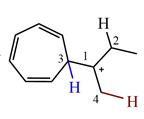
There are three hydrogen atoms on carbon atoms adjacent to the charge carrying carbon. They are on C2, C3, and C4.
Shifting of the hydride on C2 results in the charge shifting to C2, as shown in the product on the right.
Shifting of the hydride on C3 results in the charge shifting to C3, as shown in the product on the left.
Shifting of the hydride on C4 results in the charge shifting to C4, as shown in the product below the given carbocation.
Therefore, the
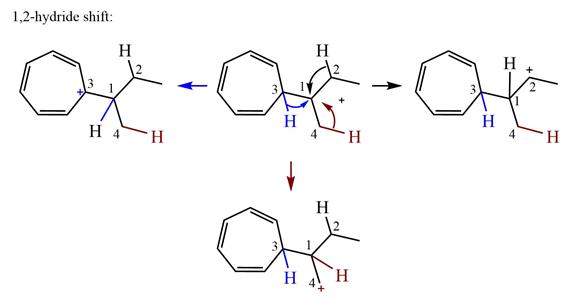
There is one methyl group attached to a carbon adjacent to C1. Shifting of the methyl group on C2 to C1 results in C1 becoming a tertiary carbon and the charge shifting to C2.
Therefore, the possible
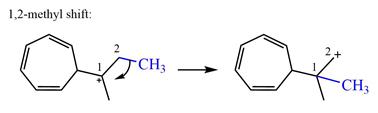
A hydride (
Want to see more full solutions like this?
Chapter 7 Solutions
Organic Chemistry: Principles And Mechanisms (second Edition)
- Provide the missing information. *see imagearrow_forwardDraw the mechanism (including all curved arrows for electron movement) showing how the maleicanhydride is attacked by the anthracene and formation of the final Diels Alder product.arrow_forwardProvide the missing information. *see imagearrow_forward
- Provide the missing information. *see imagearrow_forwardProvide the missing information. *see imagearrow_forwardI have a bottle of butanal that has been improperly used by lab workers. They allowed a traceamount NaOH (aq) to contaminate the bottle. What is now in my bottle of “butanal? What is the molecular name and functional group name? Draw the structure.arrow_forward
- Provide the missing information. *see imagearrow_forwardFirst image: Why can't the molecule C be formed in those conditions Second image: Synthesis for lactone C its not an examarrow_forwardFirst image: I have to show the mecanism for the reaction on the left, where the alcohol A is added fast in one portion Second image: I have to show the mecanism of the reaction at the bottom. Also I have to show by mecanism why the reaction wouldn't work if the alcohol was primaryarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
