
Introduction To General, Organic, And Biochemistry
12th Edition
ISBN: 9781337571357
Author: Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6.5, Problem 6.9QC
Problem 6-9
Sodium hydrogen sulfate, NaHSO4, which dissolves in water to release H+ ion, is used to adjust the pH of the water in swimming pools. Suppose we add 560. g of NaHSO4 to a swimming pool that contains 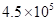 L of water at 25°C. What is the Na+ ion concentration in ppm?
L of water at 25°C. What is the Na+ ion concentration in ppm?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Pleasssssseeee solve this question in cheeemsirty, thankss sir
Show work. Don't give Ai generated solution
Pleasssssseeee solve this question in cheeemsirty, thankss sir
Chapter 6 Solutions
Introduction To General, Organic, And Biochemistry
Ch. 6.5 - Problem 6-1 How would we prepare 250 mL of a 4.4%...Ch. 6.5 - Prob. 6.2QCCh. 6.5 - Problem 6-3 How would we prepare 2.0 L of a 1.06 M...Ch. 6.5 - Prob. 6.4QCCh. 6.5 - Problem 6-5 If a 0.300 M glucose solution is...Ch. 6.5 - Problem 6-6 A certain wine contains 0.010 M NaHSO3...Ch. 6.5 - Prob. 6.7QCCh. 6.5 - Problem 6-8 A concentrated solution of 15% w/v KOH...Ch. 6.5 - Problem 6-9 Sodium hydrogen sulfate, NaHSO4, which...Ch. 6.8 - Prob. 6.10QC
Ch. 6.8 - Prob. 6.11QCCh. 6.8 - Prob. 6.12QCCh. 6.8 - Problem 6-13 What is the osmolarity of a 3.3% w/v...Ch. 6.8 - Prob. 6.14QCCh. 6 - 6-15 Answer true or false. (a) A solute is the...Ch. 6 - 6-16 Answer true or false. (a) Solubility is a...Ch. 6 - 6-17 Vinegar is a homogeneous aqueous solution...Ch. 6 - 6-18 Suppose you prepare a solution by dissolving...Ch. 6 - 6-19 In each of the following, tell whether the...Ch. 6 - 6-20 Give a familiar example of solutions of each...Ch. 6 - 6-21 Are mixtures of gases true solutions or...Ch. 6 - 6-22 Answer true or false. (a) Water is a good...Ch. 6 - 6-23 We dissolved 0.32 g of aspartic acid in 115.0...Ch. 6 - Prob. 10PCh. 6 - 6-25 A small amount of solid is added to a...Ch. 6 - 6-26 On the basis of polarity and hydrogen...Ch. 6 - Prob. 13PCh. 6 - 6-28 Which pairs of liquids are likely to be...Ch. 6 - Prob. 15PCh. 6 - 6-30 Near a power plant, warm water is discharged...Ch. 6 - 6-31 If a bottle of beer is allowed to stand for...Ch. 6 - 6-32 Would you expect the solubility of ammonia...Ch. 6 - Prob. 19PCh. 6 - Prob. 20PCh. 6 - 6-35 Describe how we would prepare the following...Ch. 6 - Prob. 22PCh. 6 - 6-37 Calculate the w/v percentage of each of these...Ch. 6 - 6-38 Describe how we would prepare 250 mL of 0.10...Ch. 6 - 6-39 Assuming that the appropriate volumetric...Ch. 6 - 6-40 What is the molarity of each solution? (a) 47...Ch. 6 - 6-41 A teardrop with a volume of 0.5 mL contains...Ch. 6 - Prob. 28PCh. 6 - 6-43 The label on a sparkling cider says it...Ch. 6 - Prob. 30PCh. 6 - 6-45 The label on ajar of jam says it contains 13...Ch. 6 - 6-46 A particular toothpaste contains 0.17 g NaF...Ch. 6 - 6-47 A student has a bottle labeled 0.750% albumin...Ch. 6 - 6-48 How many grams of solute are present in each...Ch. 6 - 6-49 A student has a stock solution of 30.0% w/v...Ch. 6 - Prob. 36PCh. 6 - Prob. 37PCh. 6 - Prob. 38PCh. 6 - 6-53 Dioxin is considered to be poisonous in...Ch. 6 - 6-54 An industrial wastewater contains 3.60 ppb...Ch. 6 - 6-55 According to the label on a piece of cheese,...Ch. 6 - Prob. 42PCh. 6 - Prob. 43PCh. 6 - Prob. 44PCh. 6 - Prob. 45PCh. 6 - 6-60 Predict which of these covalent compounds is...Ch. 6 - On the basis of Tables 6.1 and 6.2 , classify the...Ch. 6 - Prob. 48PCh. 6 - Prob. 49PCh. 6 - Prob. 50PCh. 6 - 6-67 Calculate the freezing points of solutions...Ch. 6 - 6-68 If we add 175 g of ethylene glycol, C2H6O2,...Ch. 6 - Prob. 53PCh. 6 - 6-70 In winter, after a snowstorm, salt (NaCI) is...Ch. 6 - 6-71 A 4 M acetic acid (CH3COOH) solution lowers...Ch. 6 - Prob. 56PCh. 6 - 6-73 In each case, tell which side (if either)...Ch. 6 - 6-74 An osmotic semipermeable membrane that allows...Ch. 6 - 6-75 Calculate the osmolarity of each of the...Ch. 6 - Prob. 60PCh. 6 - Prob. 61PCh. 6 - Prob. 62PCh. 6 - Prob. 63PCh. 6 - Prob. 64PCh. 6 - Prob. 65PCh. 6 - 6-78 (Chemical Connections 6A) Oxides of nitrogen...Ch. 6 - Prob. 67PCh. 6 - Prob. 68PCh. 6 - Prob. 69PCh. 6 - 6-82 (Chemical Connections 6C) A solution contains...Ch. 6 - 6-83 (Chemical Connections 6C) The concentration...Ch. 6 - 6-84 (Chemical Connections 6D) What is the...Ch. 6 - Prob. 73PCh. 6 - Prob. 74PCh. 6 - Prob. 75PCh. 6 - Prob. 76PCh. 6 - Prob. 77PCh. 6 - Prob. 78PCh. 6 - 6-91 When a cucumber is put into a saline solution...Ch. 6 - Prob. 80PCh. 6 - 6-93 Two bottles of water are carbonated, with CO2...Ch. 6 - Prob. 82PCh. 6 - Prob. 83PCh. 6 - 6-96 We know that a 0.89% saline (NaCI) solution...Ch. 6 - Prob. 85PCh. 6 - Prob. 86PCh. 6 - 6-99 A concentrated nitric acid solution contains...Ch. 6 - 6-100 Which will have greater osmotic pressure?...Ch. 6 - Prob. 89PCh. 6 - Prob. 90PCh. 6 - 6-103 A swimming pool containing 20,000. L of...Ch. 6 - Prob. 92PCh. 6 - Prob. 93PCh. 6 - Prob. 94PCh. 6 - Prob. 95PCh. 6 - Prob. 96PCh. 6 - Prob. 97PCh. 6 - Prob. 98PCh. 6 - 6-111 As noted in Section 6-8C, the amount of...Ch. 6 - 6-112 List the following aqueous solutions in...Ch. 6 - 6-113 List the following aqueous solutions in...Ch. 6 - Prob. 102P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The Ksp for lead iodide ( Pbl₂) is 1.4 × 10-8. Calculate the solubility of lead iodide in each of the following. a. water Solubility = mol/L b. 0.17 M Pb(NO3)2 Solubility = c. 0.017 M NaI mol/L Solubility = mol/Larrow_forwardPleasssssseeee solve this question in cheeemsirty, thankss sirarrow_forwardPleasssssseeee solve this question in cheeemsirty, thankss sirarrow_forward
- Only 100% sure experts solve it correct complete solutions need to get full marks it's my quiz okkkk.take your time but solve full accurate okkk chemistry expert solve itarrow_forwardPleasssssseeee solve this question in cheeemsirty, thankss sirarrow_forwardShow work. don't give Ai generated solutionarrow_forward
- Show work. Don't give Ai generated solutionarrow_forwardPart A Give the IUPAC name and a common name for the following ether: CH3-CH2-O-CH2-CH2-CH3 Spell out the full names of the compound in the indicated order separated by a comma. Submit My Answers Give Up Part B Give the IUPAC name and a common name for the following ether: Spell out the full names of the compound in the indicated order separated by a comma. Submit My Answers Give Uparrow_forwardFrenkel and Schottky are intrinsic or extrinsic defects, point or linear defects.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY