
Organic Chemistry As a Second Language: Second Semester Topics
4th Edition
ISBN: 9781119110651
Author: David R. Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6.5, Problem 6.38P
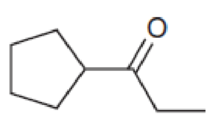
Predict the major product that is expected when each of the following compounds is treated with ethylene thioglycol (HSCH2CH2SH) in the presence of catalytic acid, followed by Raney nickel.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
A ketone undergoes acid-catalyzed bromination, acid-catalyzed chlorination, racemization, and acid-catalyzed deuterium exchange at the alpha-carbon. All of these reactions have similar rate constants. What does this tell you about the mechanisms of these reactions?
Predict the coupling products of organometallic substitutions, and use them in syntheses.
Which of the reaction conditions could afford the following transformation?
Chapter 6 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Ch. 6.1 - Predict the major product for each of the...Ch. 6.1 - Predict the major product for each of the...Ch. 6.1 - Predict the major product for each of the...Ch. 6.1 - Predict the major product for each of the...Ch. 6.1 - Predict the major product for each of the...Ch. 6.1 - Predict the major product for each of the...Ch. 6.1 - Identify the reagents you would use to achieve...Ch. 6.1 - Identify the reagents you would use to achieve...Ch. 6.1 - Identify the reagents you would use to achieve...Ch. 6.1 - Identify the reagents you would use to achieve...
Ch. 6.3 - Predict the major product of each of the following...Ch. 6.3 - Predict the major product of each of the following...Ch. 6.3 - Predict the major product of each of the following...Ch. 6.3 - Predict the major product of each of the following...Ch. 6.3 - Predict the major product of each of the following...Ch. 6.3 - Propose a mechanism for each of the following...Ch. 6.3 - Propose a mechanism for each of the following...Ch. 6.3 - Propose a mechanism for each of the following...Ch. 6.4 - Prob. 6.24PCh. 6.4 - Prob. 6.25PCh. 6.4 - Prob. 6.26PCh. 6.4 - Prob. 6.27PCh. 6.4 - Predict the major product of each of the following...Ch. 6.4 - Predict the major product of each of the following...Ch. 6.4 - Predict the major product of each of the following...Ch. 6.4 - Predict the major product of each of the following...Ch. 6.5 - Prob. 6.33PCh. 6.5 - Prob. 6.34PCh. 6.5 - Prob. 6.35PCh. 6.5 - Predict the major product that is expected when...Ch. 6.5 - Prob. 6.37PCh. 6.5 - Predict the major product that is expected when...Ch. 6.5 - Identify the reagents you would use to achieve...Ch. 6.5 - Identify the reagents you would use to achieve...Ch. 6.5 - Identify the reagents you would use to achieve...Ch. 6.5 - Identify the reagents you would use to achieve...Ch. 6.5 - Identify the reagents you would use to achieve...Ch. 6.5 - Identify the reagents you would use to achieve...Ch. 6.5 - Identify the reagents you would use to achieve...Ch. 6.5 - Identify the reagents you would use to achieve...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.7 - Prob. 6.62PCh. 6.7 - Prob. 6.63PCh. 6.7 - Prob. 6.64PCh. 6.7 - Prob. 6.65PCh. 6.7 - Prob. 6.67PCh. 6.7 - Prob. 6.68PCh. 6.7 - Prob. 6.69PCh. 6.7 - Predict the major product for each of the...Ch. 6.7 - Predict the major product for each of the...Ch. 6.7 - Predict the major product for each of the...Ch. 6.8 - Predict the major product for each of the...Ch. 6.8 - Predict the major product for each of the...Ch. 6.8 - Predict the major product for each of the...Ch. 6.8 - Predict the major product for each of the...Ch. 6.8 - Predict the major product for each of the...Ch. 6.9 - Prob. 6.81PCh. 6.9 - Prob. 6.82PCh. 6.9 - Prob. 6.83PCh. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - Propose an efficient synthesis for the following...Ch. 6.9 - Propose an efficient synthesis for the following...Ch. 6.9 - Propose an efficient synthesis for the following...Ch. 6.9 - Propose an efficient synthesis for the following...Ch. 6.9 - Propose an efficient synthesis for the following...Ch. 6.9 - Propose an efficient synthesis for the following...Ch. 6.9 - Propose an efficient synthesis for the following...Ch. 6.9 - Propose an efficient synthesis for the following...Ch. 6.9 - Propose an efficient synthesis for the following...Ch. 6.9 - Propose an efficient synthesis for the following...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Synthesize each of die following compounds by routes that involve allylic or benzylic brominacion by NBS and an...
Organic Chemistry
In qualitative analysis, Ca2+ and Ba2+ are separated from Na+, K+, and Mg2+ by adding aqueous (NH4)2CO3 to a so...
General Chemistry: Atoms First
a. Which 2+ ion has two 3d electrons? Which has eight 3d electrons? b. Which is the more likely configuration f...
Inorganic Chemistry
4.55 Using the activity series (Table 4.5), write balanced chemical equations for the following reactions. If ...
Chemistry: The Central Science (14th Edition)
41. A reaction in which A, B, and C react to form products is first order in A, second order in B, and zero ord...
Chemistry: Structure and Properties (2nd Edition)
23. Give the symbol and name for (a) an isotope with a mass number of 37 and an atomic number of 17 and (b) an ...
Chemistry For Changing Times (14th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Give the reaction conditions which could be used to bring about the transformation shown in two stepsarrow_forwardShow how you might synthesize the following compounds, using acetylene and anysuitable alkyl halides as your starting materials. If the compound given cannot besynthesized by this method, explain why. cyclodecynearrow_forwardThe p−toluenesulfonate shown undergoes an intramolecular Williamson reaction on treatment with base to give a spirocyclic ether. Demonstrate your understanding of the terminology used in the preceding sentence by writing the structure, including stereochemistry, of the product.arrow_forward
- Treatment of a cyclic ketone with diazomethane is a method for accomplishing a ring-expansion reaction. The reaction involves the initial nucleophilic attack by diazomethane on the carbonyl carbon to form a tetrahedral intermediate. Collapse of this intermediate is accompanied by bond migration and loss of N,. For example, treatment of cyclohexanone with diazomethane yields cycloheptanone. CH2N2, ether + N2 Draw the structure of the organic product(s) of the ring expansion of this compound: • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading. • Draw one structure per sketcher Add additional ketchers usina the dron down menu in the bottom right co Previous Nextarrow_forwardProvide a synthesis for of the following reactions. Show each intermediate structure between steps. No arrow-pushing mechanisms are requirEdarrow_forwarda) Give the mechanistic symbols (SN1, SN2, E1, E2) that are most consistent with each of the following statements:arrow_forward
- Provide the structure of the two organic product(s) which results when 2-bromo-2-methylbutane is treated with sodium ethoxide.arrow_forwardPropose the structure for compound that fit the following descriptions: (a)An Alkylhalide that gives a mixture of three alkenes on E2 reaction (b) An organo halide that will not undergo nucleophilic substitution (c)An Alkyl halide that give that Non-zaitsev product on E2 reaction. (d)an alcohol that react with Hcl at 0 centigrade .arrow_forwardFor each reaction, give the expected substitution product, and predict whether the mechanism will be predominantly first order (SN1) or second-order (SN2). (a) isobutyl bromide + sodium methoxidearrow_forward
- How will you synthesize cyclohexanecarboxaldehyde (cyclohexylmethanal) from the following reagents? (There are no restrictions on the reagents or the number of steps). (a) Cyclohexanone (b) Ethynylcyclohexane (c) Methyl cyclohexylformate (Remember: Formic acid is the IUPAC recognized name for Methanoic acid) (d) Cyclohexanecarboxylic acid (Cyclohexylmethanoic acid) (e) Vinylcyclohexanearrow_forwardThe following molecule undergoes an intramolecular reaction in the presence of pyrrolidinium acetate, the protonated form of pyrrolidine. Draw the product of this reaction, assuming that a dehydration reaction takes place.arrow_forwardThe compound eutypine is an antibacterial agent isolated from the fungus Eutypa lata. This fungus results in a disease common to vineyards called eutyposis. Give a sequence of reactions that will take the following reactant and give eutypine when the other reactants used in the sequence are acetylene and acetone.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Characteristic Reactions of Benzene and Phenols; Author: Linda Hanson;https://www.youtube.com/watch?v=tjEqEjDd87E;License: Standard YouTube License, CC-BY
An Overview of Aldehydes and Ketones: Crash Course Organic Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=-fBPX-4kFlw;License: Standard Youtube License