
Concept explainers
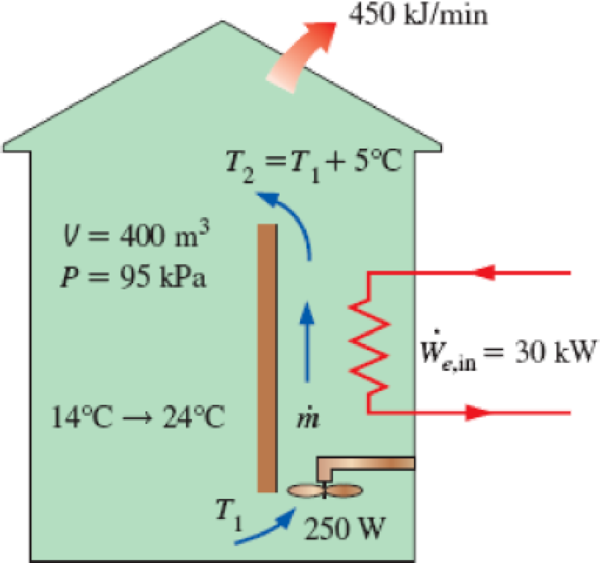
A building with an internal volume of 400 m3 is to be heated by a 30-kW electric resistance heater placed in the duct inside the building. Initially, the air in the building is at 14°C, and the local atmospheric pressure is 95 kPa. The building is losing heat to the surroundings at a steady rate of 450 kJ/min. Air is forced to flow through the duct and the heater steadily by a 250-W fan, and it experiences a temperature rise of 5°C each time it passes through the duct, which may be assumed to be adiabatic.
- (a) How long will it take for the air inside the building to reach an average temperature of 24°C?
- (b) Determine the average mass flow rate of air through the duct.
FIGURE P5–173
(a)
The time taken to attain the building’s average temperature of
Answer to Problem 173RP
The time taken to attain the building’s average temperature of
Explanation of Solution
Consider the entire building as system and the air circulates the in the building itself. There is no leakage to the surrounding.
The air flows at steady state through one inlet and one exit system (pipe and duct flow). Hence, the inlet and exit mass flow rates are equal.
Write the energy balance equation.
Here, the heat transfer is
In this system two work inputs are involved namely, the work input to the electric heater
The Equations (I) reduced as follows.
Here, there is no mass leakage from the building to the surrounding. The mass of air circulates in the building itself. Hence, inlet and exit enthalpies are neglected.
The change in internal energy is expresses as follow.
Here, the specific heat at constant volume is
Neglect the inlet and exit enthalpies and substitute
Equation (II).
Express the Equation (III) with respect to change of time and rearrange it to obtain
Write the formula for mass of air
The mass flow rate
Here, the change in time or time interval is
Refer Table A-1, “Molar mass, gas constant, and critical-point properties”.
The gas constant of air
Refer Table A-2, “Ideal-gas specific heats of various common gases”.
The specific heat at constant volume
Conclusion:
Substitute
Substitute
Substitute
Thus, the time taken to attain the building’s average temperature of
(b)
The average mass flow rate of air through the duct.
Answer to Problem 173RP
The average mass flow rate of air through the duct is
Explanation of Solution
Consider the heating duct with fan and heater only as the system. The air passes through in it steadily.
The system is at steady state. Hence, the rate of change in net energy of the system becomes zero.
The heating duct is an adiabatic duct. Hence, there is no heat loss.
The Equations (II) reduced as follows.
Express the Equation (VII) with respect to change of time as follows.
The change in enthalpy is expresses as follow.
Here, the specific heat at constant pressure is
Substitute
Refer Table A-2, “Ideal-gas specific heats of various common gases”.
The specific heat at constant pressure
Conclusion:
It is given that the temperature rise is
Substitute
Thus, The average mass flow rate of air through the duct is
Want to see more full solutions like this?
Chapter 5 Solutions
Thermodynamics: An Engineering Approach
- 1 Pleasearrow_forwardA spring cylinder system measures the pressure. Determine which spring can measure pressure between 0-1 MPa with a large excursion. The plate has a diameter of 20 mm. Also determine the displacement of each 0.1 MPa step.Spring power F=c x fF=Springpower(N)c=Spring constant (N/mm)f=Suspension (mm) How do I come up with right answer?arrow_forwardA lift with a counterweight is attached to the ceiling. The attachment is with 6 stainless and oiled screws. What screw size is required? What tightening torque? - The lift weighs 500 kg and can carry 800 kg. - Counterweight weight 600 kg - Durability class 12.8 = 960 MPa- Safety factor ns=5+-Sr/Fm= 0.29Gr =0.55arrow_forward
- Knowing that a force P of magnitude 750 N is applied to the pedal shown, determine (a) the diameter of the pin at C for which the average shearing stress in the pin is 40 MPa, (b) the corresponding bearing stress in the pedal at C, (c) the corresponding bearing stress in each support bracket at C. 75 mm 300 mm- mm A B P 125 mm 5 mm C Darrow_forwardAssume the B frame differs from the N frame through a 90 degree rotation about the second N base vector. The corresponding DCM description is: 1 2 3 4 5 6 9 # adjust the return matrix values as needed def result(): dcm = [0, 0, 0, 0, 0, 0, 0, 0, 0] return dcmarrow_forwardFind the reaction at A and B The other response I got was not too accurate,I need expert solved answer, don't use Artificial intelligence or screen shot it solvingarrow_forward
- A six cylinder petrol engine has a compression ratio of 5:1. The clearance volume of each cylinder is 110CC. It operates on the four-stroke constant volume cycle and the indicated efficiency ratio referred to air standard efficiency is 0.56. At the speed of 2400 rpm. 44000KJ/kg. Determine the consumes 10kg of fuel per hour. The calorific value of fuel average indicated mean effective pressure.arrow_forwardThe members of a truss are connected to the gusset plate as shown in (Figure 1). The forces are concurrent at point O. Take = 90° and T₁ = 7.5 kN. Part A Determine the magnitude of F for equilibrium. Express your answer to three significant figures and include the appropriate units. F= 7.03 Submit ? kN Previous Answers Request Answer × Incorrect; Try Again; 21 attempts remaining ▾ Part B Determine the magnitude of T2 for equilibrium. Express your answer to three significant figures and include the appropriate units. Figure T₂ = 7.03 C T2 |? KN Submit Previous Answers Request Answer × Incorrect; Try Again; 23 attempts remaining Provide Feedbackarrow_forwardConsider the following acid-base reaction: Fe3+(aq) +3H2O -Fe(OH)3 (s) + 3H* ← A. Using thermodynamics, calculate the equilibrium constant K at 25°C (The AG° of formation of Fe(OH)3(s) is -699 kJ/mol). B. Using the value of K you calculated in part a, if a solution contains 10-4 M Fe3+ and has a pH of 7.5, will Fe(OH)3(s) precipitate? Show all calculations necessary to justify your answer. Note that the reaction as written is for precipitation, not dissolution like Ksp-arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





