
Concept explainers
(a)
Interpretation:
Fischer projection of the given molecule is to be drawn.
Concept introduction:
A molecule containing a chain of carbon atoms is frequently represented in its zigzag conformation. If it contains multiple asymmetric centers, it is more convenient to draw its Fisher projection. Fischer projections are generally drawn with the longest carbon chain vertical. The two groups attached to each carbon except the first and last are shown on horizontal bonds. The carbon atoms in the middle of the chain are represented by the intersections of vertical and horizontal lines. The vertical lines represent bonds that are oriented away from the viewer while the horizontal bonds are oriented toward the viewer. The most oxidized group must be present at the top of the vertical line.
In the zig-zag conformation, if two groups on adjacent carbon atoms are on the same side of the plane and are shown by wedge bonds, then those groups will be on the opposite sides of the vertical chain in the Fischer projection.
In the zig-zag conformation, if two groups on adjacent carbon atoms are on the opposite sides of the plane and are shown by a wedge and a dash bond, then, in the Fischer projection, those groups will be on the same side of the vertical chain.
In the zig-zag conformation, if two groups on alternate carbon atoms are on opposite sides of the plane and are shown by a wedge and a dash bond, then, in the Fischer projection, those groups will be on the opposite sides of the vertical chain.
Answer to Problem 5.52P
The Fischer projection of the given molecule is
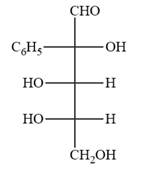
Explanation of Solution
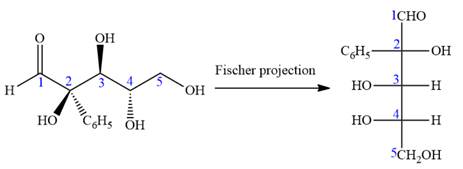
The given molecule is
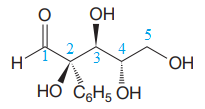
The given molecule consists of a five carbon chain, each of which is numbered. C2, C3, and C4 are the asymmetric carbon atoms. We begin to draw the Fischer projection with the framework shown below:
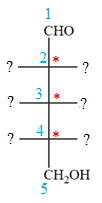
The asymmetric centers are denoted by asterisks. Two of the bonds on each asymmetric center have been left with question marks because two substituents must still be added to each so that the stereochemical configuration at those carbon atoms in the Fischer projection matches with what was given in the dash-wedge notation.
Notice that in the zig-zag conformation, the OH groups on C2 and C3 carbon atoms lie on the same side of the plane and are shown by a wedge bond. Thus, these two groups must lie on opposite sides in the Fischer projection, as shown below:
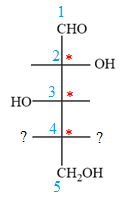
The OH groups on C3 and C4 carbon atoms lie on the opposite side of the plane and are shown by a wedge and a dash bond. Thus, these two groups must lie on the same side in the Fischer projection, as shown below:
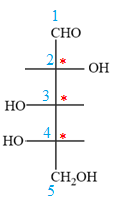
The OH groups on C2 and C4 carbon atoms lie on the opposite side of the plane and are shown by a wedge and a dash bond. Thus, these two groups must lie on the opposite side in the Fischer projection.
Thus, the OH groups on C3 and C4 carbon atoms must lie on the same side of the vertical chain, and OH group on C2 must lie on the opposite side. Then fill in the remaining groups on C2, C3, and C4 carbon atoms to complete the structure as below:
Thus, the structure above is the correct conversion from a given zig-zag conformation to a Fischer projection.
In the zig-zag conformation, if two groups on adjacent carbon atoms are on the opposite sides of the plane and are shown by a wedge and dash bond, then those groups will be on the same side of the vertical chain in the Fischer projection. In the zig-zag conformation, if two groups on adjacent carbon atoms are on opposite sides of the plane and are shown by a wedge and dash bond, then those groups will be on the same side of the vertical chain in the Fischer projection.
Interpretation:
(b)
Fischer projection of the given molecule is to be drawn.
Concept introduction:
A molecule containing a chain of carbon atoms is frequently represented in its zigzag conformation. If it contains multiple asymmetric centers, it is more convenient to draw its Fisher projection. Fischer projections are generally drawn with the longest carbon chain vertical. The two groups attached to each carbon except the first and last are shown on horizontal bonds. The carbon atoms in the middle of the chain are represented by the intersections of vertical and horizontal lines. The vertical lines represent bonds that are oriented away from the viewer while the horizontal bonds are oriented toward the viewer. The most oxidized group must be present at the top of the vertical line.
In the zig-zag conformation, if two groups on adjacent carbon atoms are on the same side of the plane and are shown by wedge bonds, then those groups will be on the opposite sides of the vertical chain in the Fischer projection.
In the zig-zag conformation, if two groups on adjacent carbon atoms are on the opposite sides of the plane and are shown by a wedge and a dash bond, then, in the Fischer projection, those groups will be on the same side of the vertical chain.
In the zig-zag conformation, if two groups on alternate carbon atoms are on opposite sides of the plane and are shown by a wedge and a dash bond, then, in the Fischer projection, those groups will be on the opposite sides of the vertical chain.
Answer to Problem 5.52P
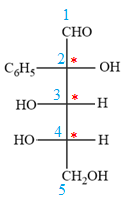
The Fischer projection of the given molecule is
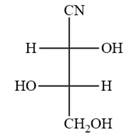
Explanation of Solution
The given molecule is
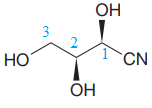
The given molecule consists of a three carbon chain, each of which is numbered. C1 and C2 carbon atoms are the asymmetric carbon atoms. We begin to draw the Fischer projection with the framework shown below:
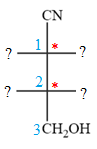
The asymmetric centers are denoted by asterisks. Two of the bonds on each asymmetric center have been left with question marks because two substituents must still be added to each so that the stereochemical configuration at those carbon atoms in the Fischer projection matches with what was given in the dash-wedge notation.
Notice that in the zig-zag conformation, the OH groups on C1 and C2 carbon atoms lie on the same side of the plane and are shown by a wedge bond. Thus, these two groups must lie on opposite side in the Fischer projection, as shown below:
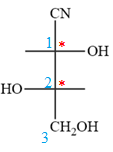
Then fill in the remaining groups on C1 and C2 carbon atoms to complete the structure as below:
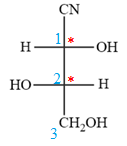
Thus, the structure above is the correct conversion from a given zig-zag conformation to a Fischer projection.
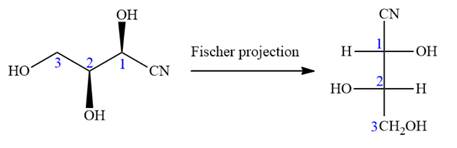
In the zig-zag conformation, if two groups on adjacent carbon atoms are on the opposite sides of the plane and are shown by a wedge and dash bond, then those groups will be on the same side of the vertical chain in the Fischer projection. In the zig-zag conformation, if two groups on adjacent carbon atoms are on opposite sides of the plane and are shown by a wedge and dash bond, then those groups will be on the same side of the vertical chain in the Fischer projection.
Interpretation:
(c)
Fischer projection of the given molecule is to be drawn.
Concept introduction:
A molecule containing a chain of carbon atoms is frequently represented in its zigzag conformation. If it contains multiple asymmetric centers, it is more convenient to draw its Fisher projection. Fischer projections are generally drawn with the longest carbon chain vertical. The two groups attached to each carbon except the first and last are shown on horizontal bonds. The carbon atoms in the middle of the chain are represented by the intersections of vertical and horizontal lines. The vertical lines represent bonds that are oriented away from the viewer while the horizontal bonds are oriented toward the viewer. The most oxidized group must be present at the top of the vertical line.
In the zig-zag conformation, if two groups on adjacent carbon atoms are on the same side of the plane and are shown by wedge bonds, then those groups will be on the opposite sides of the vertical chain in the Fischer projection.
In the zig-zag conformation, if two groups on adjacent carbon atoms are on the opposite sides of the plane and are shown by a wedge and a dash bond, then, in the Fischer projection, those groups will be on the same side of the vertical chain.
In the zig-zag conformation, if two groups on alternate carbon atoms are on opposite sides of the plane and are shown by a wedge and a dash bond, then, in the Fischer projection, those groups will be on the opposite sides of the vertical chain.
Answer to Problem 5.52P
The Fischer projection of the given molecule is
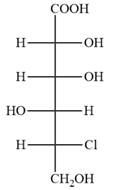
Explanation of Solution
The given molecule is
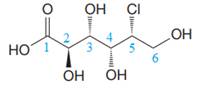
The given molecule consists of a six carbon chain, each of which is numbered. C2, C3, C4, and C5 carbon atoms are the asymmetric carbon atoms. We begin to draw the Fischer projection with the framework shown below:

The asymmetric centers are denoted by asterisks. Two of the bonds on each asymmetric center have been left with question marks because two substituents must still be added to each so that the stereochemical configuration at those carbon atoms in the Fischer projection matches with what was given in the dash-wedge notation.
Notice that in the zig-zag conformation, the OH groups on C2 and C3 carbon atoms lie on the opposite side of the plane and are shown by a wedge and a dash bond. Thus, these two groups must lie on same side in the Fischer projection, as shown below:
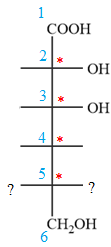
Notice that in the zig-zag conformation, the OH groups on C3 and C4 carbon atoms lie on the same side of the plane and are shown by dash bonds. Thus, these two groups must lie on the opposite sides of the vertical chain in the Fischer projection, as shown below:
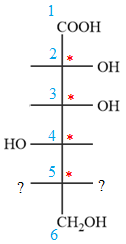
In the zig-zag conformation, the OH group and chlorine atom on C4 and C5 carbon atoms lie on the same side of the plane and are shown by dash bonds. Thus, these two groups must lie on the opposite sides of the vertical chain in the Fischer projection, as shown below:
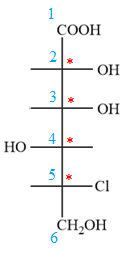
Then place the remaining groups on C1 and C2 carbon atoms to complete the structure as below:
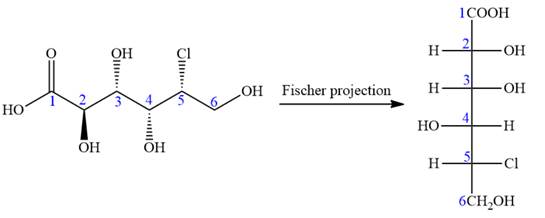
In the zig-zag conformation, if two groups on adjacent carbon atoms are on the opposite sides of the plane and are shown by a wedge and dash bond, then those groups will be on the same side of the vertical chain in the Fischer projection. In the zig-zag conformation, if two groups on adjacent carbon atoms are on opposite sides of the plane and are shown by a wedge and dash bond, then those groups will be on the same side of the vertical chain in the Fischer projection.
Want to see more full solutions like this?
Chapter 5 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Can the molecule on the right-hand side of this organic reaction be made in good yield from no more than two reactants, in one step, by moderately heating the reactants? ?A Δ O • If your answer is yes, then draw the reactant or reactants in the drawing area below. You can draw the reactants in any arrangement you like. • If your answer is no, check the box under the drawing area instead. Explanation Check Click and drag to start drawing a structure. 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibilit ku F11arrow_forward१ eq ine teaching and × + rn/takeAssignment/takeCovalentActivity.do?locator-assignment-take [Review Topics] [References] Write an acceptable IUPAC name for the compound below. (Only systematic names, not common names are accepted by this question.) Keep the information page open for feedback reference. The IUPAC name is In progress mit Answer Retry Entire Group 5 more group attempts remaining Cengage Learning | Cengage Technical Support Save and Exitarrow_forwardDraw the molecules.arrow_forward
- Draw the mechanism for the acid-catalyzed dehydration of 2-methyl-hexan-2-ol with arrows please.arrow_forward. Draw the products for addition reactions (label as major or minor) of the reaction between 2-methyl-2-butene and with following reactants : Steps to follow : A. These are addition reactions you need to break a double bond and make two products if possible. B. As of Markovnikov rule the hydrogen should go to that double bond carbon which has more hydrogen to make stable products or major product. Here is the link for additional help : https://study.com/academy/answer/predict-the-major-and-minor-products-of-2-methyl- 2-butene-with-hbr-as-an-electrophilic-addition-reaction-include-the-intermediate- reactions.html H₂C CH3 H H3C CH3 2-methyl-2-butene CH3 Same structure CH3 IENCESarrow_forwardDraw everything on a piece of paper including every single step and each name provided using carbons less than 3 please.arrow_forward
- Topics] [References] Write an acceptable IUPAC name for the compound below. (Only systematic names, not common names are accepted by this question.) Keep the information page open for feedback reference. H The IUPAC name isarrow_forward[Review Topics] [References] Write an acceptable IUPAC name for the compound below. (Only systematic names, not common names are accepted by this question.) Keep the information page open for feedback reference. The IUPAC name is Submit Answer Retry Entire Group 9 more group attempts remainingarrow_forwardPlease draw.arrow_forward
- A chromatogram with ideal Gaussian bands has tR = 9.0 minutes and w1/2 = 2.0 minutes. Find the number of theoretical plates that are present, and calculate the height of each theoretical plate if the column is 10 centimeters long.arrow_forwardAn open tubular column has an inner diameter of 207 micrometers, and the thickness of the stationary phase on the inner wall is 0.50 micrometers. Unretained solute passes through in 63 seconds and a particular solute emerges at 433 seconds. Find the distribution constant for this solute and find the fraction of time spent in the stationary phase.arrow_forwardConsider a chromatography column in which Vs= Vm/5. Find the retention factor if Kd= 3 and Kd= 30.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
