
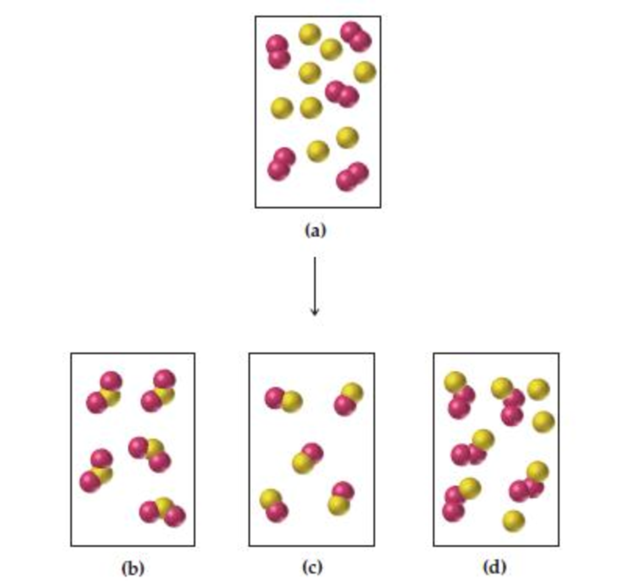
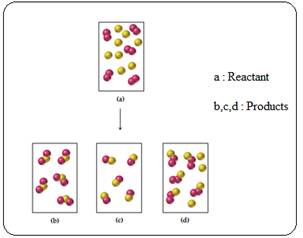
Assume that the mixture of substances in drawing (a) undergoes a reaction. Which of the drawings (b)–(d) represent a product mixture consistent with the law of conservation of mass?
Interpretation: The correct representation of product mixture consistent with the law of conservation of mass should be determined from the given representations.
Concept introduction:
Law of conservation of mass states that, the number of atoms remains constant throughout the reaction, simply it can be stated as follows, “during a chemical reaction atoms are neither be created nor be destroyed”.
Answer to Problem 5.16UKC
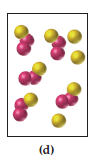
The correct representation of product mixture consistent with the law of conservation of mass is,
Explanation of Solution
Given representation of reaction is,
Law of conservation of mass states that, the number of atoms remains constant throughout the reaction.
In the representation (a) totally 18 spheres are there which representing the reactant molecules, and in the representation of product mixture (d) consistent with the law of conservation of mass, because except the other product mixture representation such as b and c only the representation d has equal number of spheres as in reactant mixture.
Therefore,
The correct representation of product mixture consistent with the law of conservation of mass is,

The correct representation of product mixture consistent with the law of conservation of mass is determined.
Want to see more full solutions like this?
Chapter 5 Solutions
FUND.OF GEN CHEM CHAP 1-13 W/ACCESS
- 13. What is the IUPAC name of this compound? A) 5-hydroxy-3,3-dimethylpentanoic acid B) 3,3-dimethylpentanoic acid C) 3,3-dimethyl-1-oxo-1,5-pentanediol D) 1,5-dihydroxy-3,3-dimethylpentanal E) 4-hydroxy-2,2-dimethylbutanoic acid HO OHarrow_forwardHelp me understand how carbon disulfide leads to toxicity in the brain, using terms like distal axonopathy, neurofilaments, covalent cross-linking, adducts, etc.,...please intuitively explain what is happening and where and the effects of it. For example, I know that CS2 reacts with amide and sulfhydryl groups on proteins, but what proteins exactly and where are they located?arrow_forwardWhat is the standard free energy change (in kJ/mole) of the spontaneous reaction between Oxygen and NADH to form H2O2 and NAD+?arrow_forward
- Redox Chemistry: Give standard free energy changes expected for the following reactions:-Succinate -> fumarate (using FAD/FADH2)-Oxaloacetate -> Malate (using NAD/NADH)-NADH --> NAD+ (using FMN/FMNH2)-CoQ --> CoQH2 (using Cytochrome C)arrow_forwardGive examples of balanced redox reactions that match the following:-Catabolic-Anabolic-Oxidative-Reductivearrow_forwardIf there are 20uM of a GLUT2 transporter on the surface of a cell, each able to move 8 per second, and 50mM glucose outside of the cell, what is the flux into the cell in mM/sec?arrow_forward
- A transporter is responsible for antiporting calcium and glucose. The transporter brings glucose into the cell and sends calcium out of the cell. If blood [calcium] = 2.55mM and intracellular [calcium] = 7uM, blood [glucose] = 5.2mM, and intracellular [glucose] = 40uM, what is the free energy of transport? Assume a membrane potential of 62mV (negative inside).arrow_forwardAn ATP-coupled transporter is used to import 1 phosphate from the extracellular environment. Intracellular phosphate exists at 65mM, while it is 2mM outside.Assume a free energy change of ATP hydrolysis of -42.7 kJ/mol. What is the net free energy change of the coupled reaction? Assume a membrane potential of 70mV.arrow_forwardAnother transporter brings 3 chloride ions into the cell. Outside, chloride has a concentration of 107mM, and 4mM inside the cell. Assuming a membrane potential of 62mV (negative inside), what is the free energy of transport of these ions?arrow_forward
- For the Oxaloacetate -> Malate reaction, assume the normal ratio of NAD/NADH, what is the maximum ratio of Malate/Oxaloacetate that will allow reaction progress?arrow_forwardA particular particle is trying to cross a membrane by simple diffusion from a high concentration of 20mM to a low concentration of 20uM. If a membrane is 15uM in width, and the diffusion coefficient of the particle is 5 uM/sec, what is the influx in uM/sec?arrow_forwardMechanisms: 1. Give a full arrow-pushing mechanism for the hydrolysis of the gamma phosphate of ATP by an ATPase. 2. Give a full arrow pushing mechanism of the spontaneous redox reaction between NAD+/NADH and oxaloacetate/malate.arrow_forward
 Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning





