
Concept explainers
(a)
Interpretation:
Enatiomers for the given compound has to be drawn using perspective formula.
Concept Introduction:
Perspective formulas show the 3D array of atoms in which the solid wedges indicate bonds projecting above the plane of the drawing and wedge shaded with parallel line indicate bonds projecting below the plane of drawing.
Example:
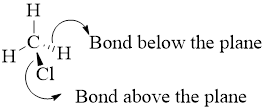
Enantiomers: These are stereoisomers that are not superimposable mirror images of each other and the configurations at all stereo genic centers are exactly opposite.
Enantiomer can be drawn by replacing the wedge with a dash from the perspective formula of a compound.
Chiral center: A chiral center is defined as the tetrahedral carbon atom in an organic molecule that is connected to four non-identical groups/substituents. It is sometimes known as a stereo genic center.
An achiral carbon is a carbon having two or more identical groups around it.
(b)
Interpretation:
Enatiomers for the given compound has to be drawn using perspective formula.
Concept Introduction:
Perspective formulas show the 3D array of atoms in which the solid wedges indicate bonds projecting above the plane of the drawing nad wedge shaded with parallel line indicate bonds projecting below the plane of drawing.
Example:
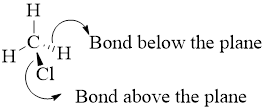
Enantiomers: These are stereoisomers that are not superimposable mirror images of each other and the configurations at all stereo genic centers are exactly opposite.
Enantiomer can be drawn by replacing the wedge with a dash from the perspective formula of a compound.
Chiral center: A chiral center is defined as the tetrahedral carbon atom in an organic molecule that is connected to four non-identical groups/substituents. It is sometimes known as a stereo genic center.
An achiral carbon is a carbon having two or more identical groups around it.
(c)
Interpretation:
Enatiomers for the given compound has to be drawn using perspective formula.
Concept Introduction:
Perspective formulas show the 3D array of atoms in which the solid wedges indicate bonds projecting above the plane of the drawing nad wedge shaded with parallel line indicate bonds projecting below the plane of drawing.
Example:
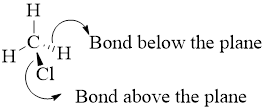
Enantiomers: These are stereoisomers that are not superimposable mirror images of each other and the configurations at all stereo genic centers are exactly opposite.
Enantiomer can be drawn by replacing the wedge with a dash from the perspective formula of a compound.
Chiral center: A chiral center is defined as the tetrahedral carbon atom in an organic molecule that is connected to four non-identical groups/substituents. It is sometimes known as a stereo genic center.
An achiral carbon is a carbon having two or more identical groups around it.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Pearson eText for Essential Organic Chemistry -- Instant Access (Pearson+)
- Draw Newman projects for each of the following molecules with 3 different rotational angles from carbon 2 to carbon 3. Rank your structures from lowest to highest energy. What causes the energy differences? Label the overlap. a. b. Br OH C. Br Brarrow_forwardDraw the stereoisomers of 3,5-diethylcylopentane. Identify the different relationships between each molecules (diasteromers, enantiomers, meso compounds, etc.)arrow_forwardPlease correct answer and don't use hand ratingarrow_forward
- Please correct answer and don't use hand rating and don't use Ai solutionarrow_forwardShow work....don't give Ai generated solutionarrow_forwardIs it possible to do the following reduction in one step? If so, add the necessary reagents and catalysts to the reaction arrow. If not, check the box under the drawing area. T G टे 13arrow_forward
- Convert the following structures into a chair representation. Then conduct a chair flip. Cl a. b. C\.... оarrow_forwardAktiv Learning App Cengage Digital Learning Part of Speech Table for Assign x o Mail-Karen Ento-Outlook * + app.aktiv.com Your Aktiv Learning trial expires on 02/06/25 at 01:15 PM Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. Problem 17 of 30 Drawing Arrows heat 4 O M B D 5x H H Und Settings H Done :0: H Jararrow_forwardConvert the following chairs into ring representations: a. Brz b.arrow_forward

 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


