
GENERAL,ORGANIC, & BIOLOGICAL CHEM-ACCES
4th Edition
ISBN: 9781265982959
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4, Problem 72P
Label the polar bonds and then decide if each molecule is polar or nonpolar.
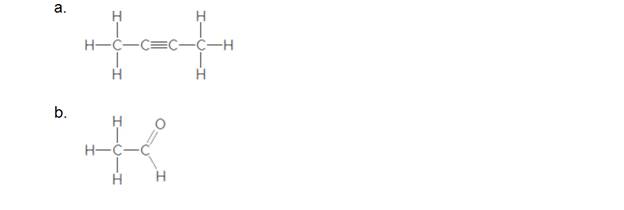
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
BA
HBr
of the fol
1)=MgCI
2) H₂O
major
NaOEt
Ts Cl
Py (pyridine)
1) 03
2) Me2S
1
4. Provide a clear arrow-pushing mechanism for the following reactions. Do not skip proton
transfers, do not combine steps, and make sure your arrows are clear enough to be interpreted
without ambiguity.
a)
NHBoc
⚫OBn
HO.
H3C
CO2CH3
-OBn
H3C
H3C.
H3C.
NHBOC
CI
CO2CH3
Draw structures of the following compounds and identify their role:
mCPBA
(MCPBA)
DMS
Py
9-BBN
LAH
Sia₂BH
TsCI
PCC
t-BuOK
LDA
MeLi
n-BuLi
DMSO
DMF
Sodium Borohydride
Lithium DiisopropylAmide
2
Chapter 4 Solutions
GENERAL,ORGANIC, & BIOLOGICAL CHEM-ACCES
Ch. 4.1 - Use electron-dot symbols to show how a hydrogen...Ch. 4.1 - Use electron-dot symbols to show how two chlorine...Ch. 4.1 - How many covalent bonds are predicted for each...Ch. 4.1 - Fill in the lone pairs on each atom to give every...Ch. 4.1 - Prob. 4.4PCh. 4.2 - Draw a Lewis structure for each covalent molecule....Ch. 4.2 - Draw a Lewis structure for dimethyl ether (C2H6O)...Ch. 4.2 - Prob. 4.4PPCh. 4.2 - Prob. 4.5PCh. 4.2 - Prob. 4.6P
Ch. 4.3 - Prob. 4.7PCh. 4.3 - Prob. 4.8PCh. 4.4 - Prob. 4.5PPCh. 4.4 - Draw resonance structures for each polyatomic...Ch. 4.4 - Nitrous oxide, N2O, is a sweet-smelling gas...Ch. 4.5 - Name each compound: (a) CS2; (b) SO2; (c) PCl5;...Ch. 4.5 - Prob. 4.6PPCh. 4.6 - What is the shape around the indicated atom in...Ch. 4.6 - NaNH2, sodium amid, is a salt that contains a...Ch. 4.6 - Prob. 4.13PCh. 4.7 - Using the trends in the periodic table, rank the...Ch. 4.7 - Use electronegativity values to classify the...Ch. 4.8 - Label the polar bonds in each molecule, and then...Ch. 4.9 - Label the polar bonds in each molecule, and then...Ch. 4.9 - Use the ball-and-stick model of dihydrocapsaicin...Ch. 4 - For each pair of compounds, classify the bonding...Ch. 4 - For each pair of compounds, classify the bonding...Ch. 4 - Prob. 17PCh. 4 - How many bonds and lone pairs are typically...Ch. 4 - Prob. 19PCh. 4 - Fill in the lone pairs needed to give the main...Ch. 4 - Prob. 21PCh. 4 - Convert the 3-D model of the general anesthetic...Ch. 4 - Draw a valid Lewis structure for each molecule. Hl...Ch. 4 - Draw a valid Lewis structure for each molecule....Ch. 4 - Prob. 25PCh. 4 - Prob. 26PCh. 4 - Draw a valid Lewis structure for...Ch. 4 - Draw a valid Lewis structure for phosgene, CCl2O ,...Ch. 4 - Draw a valid Lewis structure for each ion: (a)...Ch. 4 - Draw a valid Lewis structure for each ion: (a)...Ch. 4 - Prob. 31PCh. 4 - Keeping in mind that some elements violate the...Ch. 4 - Prob. 33PCh. 4 - Prob. 34PCh. 4 - Prob. 35PCh. 4 - Prob. 36PCh. 4 - Prob. 37PCh. 4 - Label each pair of compounds are resonance...Ch. 4 - Prob. 39PCh. 4 - Draw three resonance structures for the nitrate...Ch. 4 - Name each covalent compound. PBr3 SO3 NCl3 P2S5Ch. 4 - Name each covalent compound. SF6 CBr4 N2O P4O10Ch. 4 - Prob. 43PCh. 4 - Prob. 44PCh. 4 - Add lone pairs where needed to give octets and...Ch. 4 - Add lone pairs where needed to give octets and...Ch. 4 - Prob. 47PCh. 4 - Match each compound with one of the molecular...Ch. 4 - Prob. 49PCh. 4 - Add lone pairs where needed to give octets and...Ch. 4 - Prob. 51PCh. 4 - Considering each of the given ball-and stick...Ch. 4 - Prob. 53PCh. 4 - Prob. 54PCh. 4 - Prob. 55PCh. 4 - Predict the bond angles around the indicated atoms...Ch. 4 - Prob. 57PCh. 4 - Prob. 58PCh. 4 - Rank the atoms in each group in order of...Ch. 4 - Prob. 60PCh. 4 - Prob. 61PCh. 4 - Identify elements D, E, and F and rank them in...Ch. 4 - Prob. 63PCh. 4 - Using electronegativity values, classify the bond...Ch. 4 - Label the bond formed between carbon and each of...Ch. 4 - Label the bond formed between fluroine and each of...Ch. 4 - Which bond in each pair is more polar-that is, has...Ch. 4 - Which bond in each pair is more polar-that is, has...Ch. 4 - Prob. 69PCh. 4 - Prob. 70PCh. 4 - Label the polar bonds and then decide if each...Ch. 4 - Label the polar bonds and then decide if each...Ch. 4 - Prob. 73PCh. 4 - Explain why H2O is a polar molecule but H2S is...Ch. 4 - Convert each ball-and-stick model to a Lewis...Ch. 4 - Convert each ball-and-stick model to a Lewis...Ch. 4 - Answer the following questions about the molecule...Ch. 4 - Answer the following question about the molecule...Ch. 4 - Prob. 79PCh. 4 - Lactic acid gives sour milk its distinctive taste....Ch. 4 - Use the ball-and-stick model of zingerone, a...Ch. 4 - Prob. 82PCh. 4 - Prob. 83PCh. 4 - Prob. 84PCh. 4 - Isobutyl cyanoacrylate is used in medical glues to...Ch. 4 - Prob. 86PCh. 4 - Cyclopropane is a stable compound that contains...Ch. 4 - Prob. 88CPCh. 4 - Prob. 89CPCh. 4 - Prob. 90CP
Additional Science Textbook Solutions
Find more solutions based on key concepts
How could you separate a mixture of the following compounds? The reagents available to you are water, either, 1...
Organic Chemistry (8th Edition)
The validity of a scientific law.
Physical Universe
To test your knowledge, discuss the following topics with a study partner or in writing ideally from memory. Th...
HUMAN ANATOMY
Single penny tossed 20 times and counting heads and tails: Probability (prediction): _______/20 heads ________/...
Laboratory Manual For Human Anatomy & Physiology
Some people compare DNA to a blueprint stored in the office of a construction company. Explain how this analogy...
Biology: Concepts and Investigations
Why are mutants used as test organisms in the Ames test?
Laboratory Experiments in Microbiology (12th Edition) (What's New in Microbiology)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Using Luther's rule, calculate the reference potential of the Hg2+/Hg redox electrode. DATA: Electrode potentials E° = 0,854 V y E 0,788 V Hg2+/Hg 2+ Hg2/Hgarrow_forward1) NaNH2 (excess) 1) NaNH2 CI CI 2) H₂O 2) Mel 1) 03 2) (CH3)2S Na NH3 (liquid) 1arrow_forwardCI 1) n-BuLi 2) 1) 03 HH T&Cl 2) H₂O 2arrow_forward
- Help with a!arrow_forwardFor the following compound: HO -H Draw a mechanism for the tautomerization process under BASIC conditions: Mechanism A: H-O: H-OH H-O HH H-OO Mechanism B: H-Q Mechanism C: Θ OH H-O: Mechanism D: H-O H- H-OO C H-OO H- H- H-OO HH OH -H - HON H :OH H-Harrow_forwardidentify the product (or multiple products) for each of the following reactions: CI 1) NaNH2 (excess) ठ Cl 2) H₂O Hz H₂SO₂, H₂O HgSO Lindlar's catalyst 1) n-BuLi 2) 1)9-BBN 2) H₂O, NaOH ? Br H A B C afó gó H OA B O c OD E OF D E F H H Na, NHarrow_forward
- Identify the product (or multiple products) for each of the following reactions: ? or CI CI 1) NaNHz (excess) 2) H₂O OA OB O C OD OE OF H₂SO₂, H₂O Hq50. 1) n-BuLi 2) Br 1) 9-BBN 2) H₂O₂, NaOH A B H H متته D E H H H H C H H F H H H₂ Lindlar's catalyst Na NHarrow_forwardIdentify the product (or multiple products) for each of the following reactions: O A OB Oc OD OE OF CI CI 1) NaNH2 (excess) 2) H₂O H₂ H₂SO2, H₂O HgSO Lindlar's catalyst 1) n-BuLi 2) Br 1)9-BBN 2) H₂O₂, NaOH ? Na, NH3 C H A H H مننه مننه منن مننه H F H H E مند H D H Harrow_forwardFor the following compound: HO H Draw a mechanism for the tautomerization process under BASIC conditions: Mechanism A: + H-O: H-OH₂ H Mechanism B: H-Ö: HO-H H-OO -H H HH H H HH H-O: H-OO H-OO -H H e -H : OH Θ Mechanism C: Θ A : OH H-O: H H H-O-H 0. Mechanism D: e.. : OH :0 H H-O-H H-O: H-OO :O H -H H H сём H 0 :0 + H Θ H H H-arrow_forward
- For the following compound: H OH Draw a mechanism for the tautomerization process under ACIDIC conditions: Mechanism A: Θ :OH O O-H HO 0: Mechanism B: :O-H e.. Θ :OH Mechanism C: H HO-H :0: Θ 0: H H e.. : OH 0: "Θ HH O. :OH :OH O-H O-H Mechanism D: :OH H-OH₂ :OH HO-H 0: © O-H H HH 0: HHarrow_forwardHelp w c!arrow_forwardCan someone help me understand this?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning  World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:9781559539418
Author:Angelica Stacy
Publisher:MAC HIGHER

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY