
(a)
Interpretation:
The name of the Parent chain for the given compound should be identified.
Concept introduction:
Parent chain is the longest chain of a compound. If there are two longest chains, the more substituted chain is the parent chain.
Name for the
| Number of carbon atoms | Name of parent alkane |
| 1 | methane |
| 2 | ethane |
| 3 | propane |
| 4 | butane |
| 5 | Pentane |
| 6 | hexane |
| 7 | Heptane |
| 8 | octane |
| 9 | nonane |
| 10 | decane |
(a)
Answer to Problem 36PP
Octane
Explanation of Solution
The longest chain of the given compounds is identified by counting the number of carbon atoms presented in each chain of a compound. Longest chain is the carbon skeleton of a molecule that contains more number of carbons.
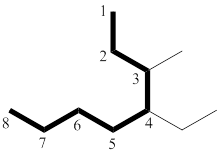
The longest chain has eight carbon atoms. By referring the above table, deduce the name as octane for parent chain.
(b)
Interpretation:
The name of the Parent chain for given compounds should be identified.
Concept introduction:
Parent chain is the longest chain of a compound. If there are two longest chains, the more substituted chain is the parent chain.
Name for the alkane parent chain depends on the number of carbon atoms present in the longest continuous carbon chain.
| Number of carbon atoms | Name of parent alkane |
| 1 | methane |
| 2 | ethane |
| 3 | propane |
| 4 | butane |
| 5 | Pentane |
| 6 | hexane |
| 7 | Heptane |
| 8 | octane |
| 9 | nonane |
| 10 | decane |
(b)
Answer to Problem 36PP
Nonane
Explanation of Solution
The longest chain of the given compounds is identified by counting the number of carbon atoms presented in each chain of a compound. Longest chain is the carbon skeleton of a molecule that contains more number of carbons.
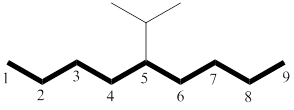
The longest chain has nine carbon atoms. By referring the above table, deduce the name as nonane for parent chain.
(c)
Interpretation:
The name of the Parent chain for given compounds should be identified.
Concept introduction:
Parent chain is the longest chain of a compound. If there are two longest chains, the more substituted chain is the parent chain.
Name for the alkane parent chain depends on the number of carbon atoms present in the longest continuous carbon chain.
| Number of carbon atoms | Name of parent alkane |
| 1 | methane |
| 2 | ethane |
| 3 | propane |
| 4 | butane |
| 5 | Pentane |
| 6 | hexane |
| 7 | Heptane |
| 8 | octane |
| 9 | nonane |
| 10 | decane |
(c)
Answer to Problem 36PP
Octane
Explanation of Solution
The longest chain of the given compounds is identified by counting the number of carbon atoms presented in each chain of a compound. Longest chain is the carbon skeleton of a molecule that contains more number of carbons.
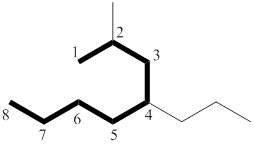
Here there are two longest chain of carbon atoms presented. But one has one substituent and other has two substituents. So the two substituted longest chain is the parent chain.
The longest chain has eight carbon atoms. By referring the above table, deduce the name as octane for parent chain.
(d)
Interpretation:
The name of the Parent chain for given compounds should be identified.
Concept introduction:
Parent chain is the longest chain of a compound. If there are two longest chains, the more substituted chain is the parent chain.
Name for the alkane parent chain depends on the number of carbon atoms present in the longest continuous carbon chain.
| Number of carbon atoms | Name of parent alkane |
| 1 | methane |
| 2 | ethane |
| 3 | propane |
| 4 | butane |
| 5 | Pentane |
| 6 | hexane |
| 7 | Heptane |
| 8 | octane |
| 9 | nonane |
| 10 | decane |
(d)
Answer to Problem 36PP
Heptane
Explanation of Solution
The longest chain of the given compounds is identified by counting the number of carbon atoms presented in each chain of a compound. Longest chain is the carbon skeleton of a molecule that contains more number of carbons.
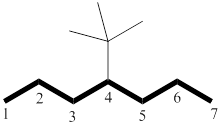
The longest chain has seven carbon atoms. By referring the above table, deduce the name as heptane for parent chain.
Want to see more full solutions like this?
Chapter 4 Solutions
EBK ORGANIC CHEMISTRY-STUD.SOLNS.MAN+SG
- 10. The most important reason why Br- is a better nucleophile than Cl-is ___. A. polarizability; B. size; C. solvation; D. basicity; E. polarity. Please include all steps. Thanks!arrow_forwardPredicting the qualitative acid-base properties of salts Consider the following data on some weak acids and weak bases: base acid Ка K₁₁ name formula name formula nitrous acid HNO2 4.5×10 4 pyridine CHEN 1.7 × 10 9 4 hydrofluoric acid HF 6.8 × 10 methylamine CH3NH2 | 4.4 × 10¯ Use this data to rank the following solutions in order of increasing pH. In other words, select a '1' next to the solution that will have the lowest pH, a '2' next to the solution that will have the next lowest pH, and so on. solution 0.1 M NaNO2 0.1 M KF pH choose one v choose one v 0.1 M C5H5NHBr 0.1 M CH3NH3CI choose one v ✓ choose one 1 (lowest) 2 ☑ 3 4 (highest) 000 18 Ararrow_forward4. The major product from treatment of 2-propanol with the Jonesreagent is ___.A. acetone; B. none of the other answers is correct C. propene; D.propanoic acid; E carbon dioxide. Please include all steps! Thank you!arrow_forward
- 7. All of the following compounds that are at the same oxidation levelare ___.u. methyl epoxide, v. propyne, w. propanal, x. propene,y. 2,2-dihydroxypropane, z. isopropanol?A. u,v,w,y; B. u,v,w; C. v,w,y,z; D. v, z; E. x,y,z Please include all steps. Thank you!arrow_forward9. Which one of the following substituents is the worst leaving group inan SN2 reaction? A. -NH2; B. -OH; C. –F; D. NH3; E. H2O Please include all steps. Thanks!arrow_forwardUsing the general properties of equilibrium constants At a certain temperature, the equilibrium constant K for the following reaction is 2.5 × 105: CO(g) + H2O(g) CO2(g) + H2(g) Use this information to complete the following table. Suppose a 7.0 L reaction vessel is filled with 1.7 mol of CO and 1.7 mol of H2O. What can you say about the composition of the mixture in the vessel at equilibrium? What is the equilibrium constant for the following reaction? Be sure your answer has the correct number of significant digits. CO2(9)+H2(g) CO(g)+H₂O(g) What is the equilibrium constant for the following reaction? Be sure your answer has the correct number of significant digits. 3 CO(g)+3H2O(g) = 3 CO2(g)+3H2(g) There will be very little CO and H2O. x10 There will be very little CO2 and H2. 000 Neither of the above is true. K = ☐ K = ☐ 18 Ararrow_forward
- 8. When ethane thiol is treated with hydrogen peroxide the product is___.A. ethane disulfide; B. diethyl sulfide; C. ethane sulfoxide; D. ethanesulfate; E. ethyl mercaptan. Please include all steps. Thanks!arrow_forward5. The major product of the three step reaction that takes place when 1-propanol is treated with strong acid is?A. dipropyl ether; B. propene; C. propanal; D. isopropyl propyl ether;E. 1-hexanol Please include all steps. Thank you!arrow_forward6. The formula of the product of the addition of HCN to benzaldehydeis ___.A. C8H7NO; B. C8H6NO; C. C14H11NO; D. C9H9NO; E. C9H8NO Please include all steps. Thank you!arrow_forward
- Predicting the qualitative acid-base properties of salts Consider the following data on some weak acids and weak bases: base acid K K a name formula name formula nitrous acid HNO2 4.5×10 hydroxylamine HONH2 1.1 × 10 8 hypochlorous acid HCIO 8 3.0 × 10 methylamine CH3NH2 | 4.4 × 10¯ 4 Use this data to rank the following solutions in order of increasing pH. In other words, select a '1' next to the solution that will have the lowest pH, a '2' next to the solution that will have the next lowest pH, and so on. 0.1 M KCIO solution PH choose one 0.1 M NaNO2 0.1 M CH3NH3Br 0.1 M NaBr choose one ✓ choose one v ✓ choose one 1 (lowest) ☑ 2 3 4 (highest)arrow_forwardFor this Orgo problem, don't worry about question 3 below it. Please explain your thought process, all your steps, and also include how you would tackle a similar problem. Thank you!arrow_forwardUsing the general properties of equilibrium constants At a certain temperature, the equilibrium constant K for the following reaction is 0.84: H2(g) + 2(g) 2 HI(g) = Use this information to complete the following table. Suppose a 34. L reaction vessel is filled with 0.79 mol of HI. What can you say about the composition of the mixture in the vessel at equilibrium? There will be very little H2 and 12. ☐ x10 There will be very little HI. Neither of the above is true. What is the equilibrium constant for the following reaction? Be sure your answer has the correct number of significant digits. 2 HI(g) H₂(9)+12(9) K = What is the equilibrium constant for the following reaction? Be sure your answer has the correct number of significant digits. 2 H2(g)+212(9) 4 HI(g) K = ☐ ☑arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





