
Chemistry
3rd Edition
ISBN: 9780073402734
Author: Julia Burdge
Publisher: MCGRAW-HILL HIGHER EDUCATION
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 3.3, Problem 1PPC
Practice Problem CONCEPTUALIZE
CONCEPTUALIZE
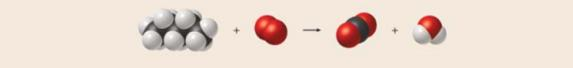
Write a balanced equation for the reaction shown here.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Naming and drawing secondary
Write the systematic (IUPAC) name for each of the following organic molecules:
CH3
Z
structure
CH3
CH2
CH2
N-CH3
CH3-CH2-CH2-CH-CH3
NH
CH3-CH-CH2-CH2-CH2-CH2-CH2-CH3
Explanation
Check
☐
name
☐
2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy C
G
C
This question shows how molecular orbital (MO) theory can be used to understand the chemical
properties of elemental oxygen O₂ and its anionic derivative superoxide Oz.
a)
Draw the MO energy diagram for both O2 and O2. Clearly label your diagram with atomic orbital names
and molecular orbital symmetry labels and include electrons.
Draw the Lewis structure of O2. How does the MO description of O2 differ from the Lewis structure, and
how does this difference relate to the high reactivity and magnetic properties of oxygen?
) Use the MO diagram in (a) to explain the difference in bond length and bond energy between superoxide
ion (Oz, 135 pm, 360 kJ/mol) and oxygen (O2, 120.8 pm, 494 kJ/mol).
Please draw
Chapter 3 Solutions
Chemistry
Ch. 3.1 - Practice ProblemATTEMPT Calculate the molecular or...Ch. 3.1 - Practice Problem BUILD
Calculate the molecular or...Ch. 3.1 - Practice ProblemCONCEPTUALIZE Some...Ch. 3.1 - What is the molecular mass of citric acid ( H 3 C...Ch. 3.1 - 3.1.2 What is the formula mass of calcium citrate...Ch. 3.2 - Practice Problem ATTEMPT
Determine the percent...Ch. 3.2 - Practice Problem BUILD
Determine the simplest...Ch. 3.2 - Practice Problem CONCEPTUALIZE
Determine the...Ch. 3.2 - 3.2.1 What is the percent composition by mass of...Ch. 3.2 - What is the percent composition by mass of sodium...
Ch. 3.3 - Prob. 1PPACh. 3.3 - Practice Problem BUILD
Write and balance the...Ch. 3.3 - Practice ProblemCONCEPTUALIZE Write a balanced...Ch. 3.3 - What are the stoichiometric coefficients in the...Ch. 3.3 - 3.3.2 Which chemical equation represents the...Ch. 3.3 - 3.3.3 Which is the correctly balanced form of the...Ch. 3.3 - 3.3.4 Carbon monoxide reacts with oxygen to...Ch. 3.4 - Prob. 1PPACh. 3.4 - Practice Problem BUILD
Write and balance the...Ch. 3.4 - Prob. 1PPCCh. 3.4 - How many molecules are in 30 .1 g of sulfur...Ch. 3.4 - How many moles of hydrogen are there in 6 .50 g of...Ch. 3.4 - 3.4.3 Determine the empirical formula of a...Ch. 3.4 - Determine the empirical formula of a compound that...Ch. 3.5 - Practice ProblemATTEMPT Potassium is the second...Ch. 3.5 - Practice Problem BUILD
Calculate (a) the number...Ch. 3.5 - Practice Problem CONCEPTUALIZE
These diagrams show...Ch. 3.5 - What is the empirical formula of a compound...Ch. 3.5 - 3.5.2 What are the empirical and molecular...Ch. 3.5 - 3.5.3 Determine the masses of and produced by...Ch. 3.5 - How is it possible for the combined masses of CO 2...Ch. 3.6 - Prob. 1PPACh. 3.6 - Practice Problem BUILD
(a) Determine the mass of...Ch. 3.6 - Practice ProblemCONCEPTUALIZE Plain doughnuts from...Ch. 3.6 - 3.6.1 How many moles of will be produced if Li...Ch. 3.6 - 3.6.2 Determine the stoichiometric amount (in...Ch. 3.7 - Prob. 1PPACh. 3.7 - Practice ProblemBUILD (a) Determine the number of...Ch. 3.7 - Practice ProblemCONCEPTUALIZE A particular...Ch. 3.7 - 3.7.1 What mass of is produced according to the...Ch. 3.7 - 3.7.2 What is the percent yield for a process in...Ch. 3.7 - How many moles of NH 3 can be produced by the...Ch. 3.7 - What mass of water is produced by the reaction of...Ch. 3.7 - Reactants A (red) and B (blue) combine to form a...Ch. 3.7 - Which of the following represents the contents of...Ch. 3.8 - Practice ProblemATTEMPT Determine the empirical...Ch. 3.8 - Practice ProblemBUILD Determine the empirical...Ch. 3.8 - Practice ProblemCONCEPTUALIZE What is the smallest...Ch. 3.9 - Practice ProblemATTEMPT The combustion of a 28...Ch. 3.9 - Practice ProblemBUILD Determine the mass of CO 2...Ch. 3.9 - Prob. 1PPCCh. 3.10 - Prob. 1PPACh. 3.10 - Prob. 1PPBCh. 3.10 - Prob. 1PPCCh. 3.11 - Practice ProblemATTEMPT Calculate the mass of...Ch. 3.11 - Practice Problem BUILD
What mass of glucose must...Ch. 3.11 - Prob. 1PPCCh. 3.12 - Prob. 1PPACh. 3.12 - Prob. 1PPBCh. 3.12 - Practice ProblemCONCEPTUALIZE The diagrams show a...Ch. 3.13 - Prob. 1PPACh. 3.13 - Practice Problem BUILD
What mass of ether will be...Ch. 3.13 - Prob. 1PPCCh. 3.14 - Prob. 1PPACh. 3.14 - Practice Problem BUILD
Using the chemical species...Ch. 3.14 - Prob. 1PPCCh. 3 - 3.1
Calculate the mass of water produced in the...Ch. 3 - 3.2
How much can be produced?
(a)
350.0...Ch. 3 - 3.3
How much can be produced?
(a)
91.51...Ch. 3 - 3.4
How much of the excess reactant remains when...Ch. 3 - What is meant by the term molecular mass, and why...Ch. 3 - Explain the difference between the terms molecular...Ch. 3 - Calculate the molecular mass (in amu) of each of...Ch. 3 - Calculate the molecular mass (in amu) of each of...Ch. 3 - Calculate the molecular mass or formula mass (in...Ch. 3 - 3.6 Calculate the molecular mass or formula mass...Ch. 3 - 3.7 Use ammonia to explain what is meant by the...Ch. 3 - 3.8 Describe how the knowledge of the percent...Ch. 3 - Tin (Sn) exists in Earth's crust as SnO 2 ....Ch. 3 - 3.10 For many years, chloroform was used as an...Ch. 3 - All the substances listed here are fertilizers...Ch. 3 - Limonene. shown here, is a by-product of the...Ch. 3 - 3.13 Tooth enamel is . Calculate the percent...Ch. 3 - A four-pack of Red Bull Energy Drink consists of...Ch. 3 - 3.15 A “variety pack" of ramen noodles consists of...Ch. 3 - Prob. 16QPCh. 3 - Prob. 17QPCh. 3 - 3.18 Why must a chemical equation be balanced?...Ch. 3 - Prob. 19QPCh. 3 - Write an unbalanced equation to represent each of...Ch. 3 - Write an unbalanced equation to represent each of...Ch. 3 - Prob. 22QPCh. 3 - For each of the following unbalanced chemical...Ch. 3 - Prob. 24QPCh. 3 - Prob. 25QPCh. 3 - Which of the following equations best represents...Ch. 3 - Prob. 27QPCh. 3 - Define the term mole. What is the unit for mole in...Ch. 3 - Prob. 29QPCh. 3 - Prob. 30QPCh. 3 - If we know the empirical formula of a compound,...Ch. 3 - Prob. 32QPCh. 3 - The thickness of a piece of paper is 0.0036 in....Ch. 3 - Prob. 34QPCh. 3 - Prob. 35QPCh. 3 - Prob. 36QPCh. 3 - How many grams of gold ( Au ) are there in 15.3...Ch. 3 - Prob. 38QPCh. 3 - Prob. 39QPCh. 3 - 3.40 What is the mass in grams of lead atoms?
Ch. 3 - Prob. 41QPCh. 3 - Prob. 42QPCh. 3 - Which of the following has a greater mass: two...Ch. 3 - Prob. 44QPCh. 3 - Prob. 45QPCh. 3 - Prob. 46QPCh. 3 - Prob. 47QPCh. 3 - Prob. 48QPCh. 3 - Prob. 49QPCh. 3 - Prob. 50QPCh. 3 - Prob. 51QPCh. 3 - Prob. 52QPCh. 3 - Prob. 53QPCh. 3 - Prob. 54QPCh. 3 - Prob. 55QPCh. 3 - Prob. 56QPCh. 3 - Prob. 57QPCh. 3 - Prob. 58QPCh. 3 - In response to invasion by a microorganism, the...Ch. 3 - Researchers recently reported that the compound in...Ch. 3 - Prob. 61QPCh. 3 - Prob. 62QPCh. 3 - 3.63 Menthol is a flavoring agent extracted from...Ch. 3 - 3.64 Ascorbic acid (vitamin C) contains C, H, and...Ch. 3 - The amino acid cysteine plays an important role in...Ch. 3 - 3.66 The diagram shows the products of a...Ch. 3 - 3.67 Which of the following diagrams could...Ch. 3 - 3.68 On what law is stoichiometry based? Why is it...Ch. 3 - Prob. 69QPCh. 3 - Prob. 70QPCh. 3 - Prob. 71QPCh. 3 - Prob. 72QPCh. 3 - Prob. 73QPCh. 3 - Prob. 74QPCh. 3 - 3.75 When baking soda (sodium bicarbonate or...Ch. 3 - Prob. 76QPCh. 3 - Prob. 77QPCh. 3 - Prob. 78QPCh. 3 - Prob. 79QPCh. 3 - Prob. 80QPCh. 3 - Prob. 81QPCh. 3 - Prob. 82QPCh. 3 - Prob. 83QPCh. 3 - Prob. 84QPCh. 3 - Prob. 85QPCh. 3 - Prob. 86QPCh. 3 - Prob. 87QPCh. 3 - Prob. 88QPCh. 3 - Prob. 89QPCh. 3 - Prob. 90QPCh. 3 - Nitroglycerin ( C 3 H 5 N 3 O 9 ) is a powerful...Ch. 3 - Prob. 92QPCh. 3 - Prob. 93QPCh. 3 - Prob. 94QPCh. 3 - Prob. 95QPCh. 3 - Prob. 96QPCh. 3 - Prob. 97QPCh. 3 - Prob. 98QPCh. 3 - Consider the reaction N 2 +3H 2 → 2NH 3 Assuming...Ch. 3 - 3.104 Determine whether each of the following...Ch. 3 - Prob. 101QPCh. 3 - Prob. 102APCh. 3 - Prob. 103APCh. 3 - Prob. 104APCh. 3 - Prob. 105APCh. 3 - Prob. 106APCh. 3 - Prob. 107APCh. 3 - 3.112 The carat is the unit of mass used by...Ch. 3 - An iron bar weighed 664 g. After the bar had been...Ch. 3 - Prob. 110APCh. 3 - Suppose you are given a cube made of magnesium...Ch. 3 - Prob. 112APCh. 3 - Prob. 113APCh. 3 - Prob. 114APCh. 3 - Calculate the number of cations and anions in each...Ch. 3 - Prob. 116APCh. 3 - 3.121 Avogadro’s number has sometimes been...Ch. 3 - Prob. 118APCh. 3 - In the formation of carbon monoxide. CO, it is...Ch. 3 - Prob. 120APCh. 3 - Prob. 121APCh. 3 - A compound made up of C, H, and Cl contains 55.0...Ch. 3 - Prob. 123APCh. 3 - Prob. 124APCh. 3 - Prob. 125APCh. 3 - Prob. 126APCh. 3 - Prob. 127APCh. 3 - 3.132 A mixture of methane and ethane of mass...Ch. 3 - Prob. 129APCh. 3 - A die has an edge length of 1.5 cm. (a) What is...Ch. 3 - Prob. 131APCh. 3 - Prob. 132APCh. 3 - A sample containing NaCl, Na 2 SO 4 , and NaNO 3...Ch. 3 - Prob. 134APCh. 3 - Prob. 135APCh. 3 - Prob. 136APCh. 3 - An impure sample of zinc (Zn) is treated with an...Ch. 3 - One of the reactions that occurs in a blast...Ch. 3 - Prob. 139APCh. 3 - Prob. 140APCh. 3 - Prob. 141APCh. 3 - 3.146 Aspirin or acetylsalicylic acid is...Ch. 3 - Prob. 143APCh. 3 - Prob. 144APCh. 3 - Lysine, an essential amino acid in the human body,...Ch. 3 - Prob. 146APCh. 3 - Prob. 147APCh. 3 - Prob. 148APCh. 3 - Prob. 149APCh. 3 - Cysteine, shown here, is one of the 20 amino acids...Ch. 3 - Prob. 151APCh. 3 - Carbon dioxide ( CO 2 ) is the gas that is mainly...Ch. 3 - Prob. 153APCh. 3 - Prob. 154APCh. 3 - Prob. 155APCh. 3 - Prob. 156APCh. 3 - 3.161 Potash is any potassium mineral that is used...Ch. 3 - Prob. 158APCh. 3 - Prob. 159APCh. 3 - Prob. 160APCh. 3 - Prob. 1SEPPCh. 3 - Prob. 2SEPPCh. 3 - Prob. 3SEPPCh. 3 - Prob. 4SEPP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- -Page: 8 nsition metal ions have high-spin aqua complexes except one: [Co(HO)₁]". What is the d-configuration, oxidation state of the metal in [Co(H:O))"? Name and draw the geometry of [Co(H2O)]? b) Draw energy diagrams showing the splitting of the five d orbitals of Co for the two possible electron configurations of [Co(H2O)]: Knowing that A = 16 750 cm and Пl. = 21 000 cm, calculate the configuration energy (.e., balance or ligand-field stabilization energy and pairing energy) for both low spin and high spin configurations of [Co(H2O)]. Which configuration seems more stable at this point of the analysis? (Note that 349.76 cm = 1 kJ/mol) Exchange energy (IT) was not taken into account in part (d), but it plays a role. Assuming exchange an occur within t29 and within eg (but not between tz, and ea), how many exchanges are possible in the low in configuration vs in the high spin configuration? What can you say about the importance of exchange energy 07arrow_forwardDraw everything please on a piece of paper explaining each steparrow_forwardDefine crystalline, polycrystalline and amorphous materials What crystal system and Bravais lattices are shown in the figure immediately below? What do a, b, C, a, ẞ and y represent and what are their values? You can label the Bravais lattices directly above or under the figure. C aarrow_forward
- 32. The diagrams below show the band structure of an intrinsic semiconductor at absolute zero and room temperature. Room Temperature EF E OK Ep- a) In the space below, sketch a similar pair of diagrams for an n-type semiconductor. D) Give the definition and an example of (i) an intrinsic semiconductor and (ii) an n-type semiconductor.arrow_forward29. a) i Which energy diagram best represents the d-electrons in tetrahedral [Co(NH3)4]²+? b) ii c) iii d) iv 11 ་ ↑↓ ↑t t ↑↓ ↑↓ e) none of these ii In1 According to Slater's rules, what is the effective nuclear charge experienced by a 3d electron in 30. Ge? a) 32.00 b) 21.15 c) 16.05 d) 14.00 e) 10.85arrow_forwardRegarding Lowis structuros and geometrios, Draw Lewis structures for the following: SOF4, SO, ICI, XeO2F4, SeF and XeO3. For each one, indicate the observed molecular geometry it adopts.arrow_forward
- Explain the following statements with equations: - The fusion product of an organic compund with sodium metal is an alkaline solution - The test for elements should be done before the solubility tests. - Using less sodium than the organic compound in the ignition test might cause a problem to detect the presence of the nitrogen and sulfur - Formation of colored product when adding ferric acid chloride to phenol solutionarrow_forward31 Indicate the symbol, mass number and the atomic number of the missing product in each of the following nuclear reactions. a) 13/3 N 41 b) 11 Ca 20 c) 90 38 Sr → 133 C + ? + - 6 0 e →? 90 Y + ? 39 11 d) 22 Na → ? + + 1 B +1 β Toarrow_forwardPlease drawarrow_forward
- 9. compore the Following two Venctions IN termy Of Ronction Rate and explan in detail the reasoning that led to your conclusion +He p₁₂ 11- ㅐ 15 .. +He H #H H / H b. Compare the Following too reactions 14 terms of reaction Rate and explain in detail the reasoning that led to your conclusion Н d-C- tłu Na +2446 е -ll +2n "Harrow_forwarda. •Write all of the possible products For the Following ronction А ----- H - H H + H₂0 H+ Н b. in Rite the complete reaction Mechaniszn For the Formation of each product. ·C. Suggest what Reaction conditions could Result in each product being the major Product of the veaction:arrow_forwarda. Write the product For each of the Following reactions H 6-836-6 레 +H₂ N A H A-C-C=C-C-CH + 2 Na +2 NH3 - H H b. Write the reaction Mechanism For. reaction eacharrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning
GCSE Chemistry - Differences Between Compounds, Molecules & Mixtures #3; Author: Cognito;https://www.youtube.com/watch?v=jBDr0mHyc5M;License: Standard YouTube License, CC-BY