
Concept explainers
Locate the isoprene units in each compound.
a. 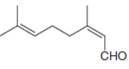 e.
e. 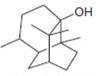 g.
g. 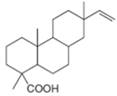
neral patchouli alcohol dextropimaric acid
b. 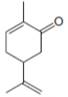 f.
f. 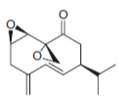 h.
h.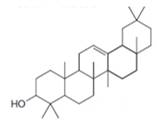
carvone periplanone B
c. 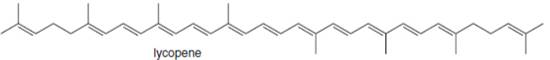
d. 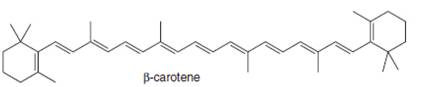
(a)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: Terpenes are naturally occurring compounds that are present in plants and animals. Terpenes contain one or more isoprene units. Terpenoids are derivatives of terpenes with oxygen containing functional group such as carbonyl groups.
Answer to Problem 31.26P
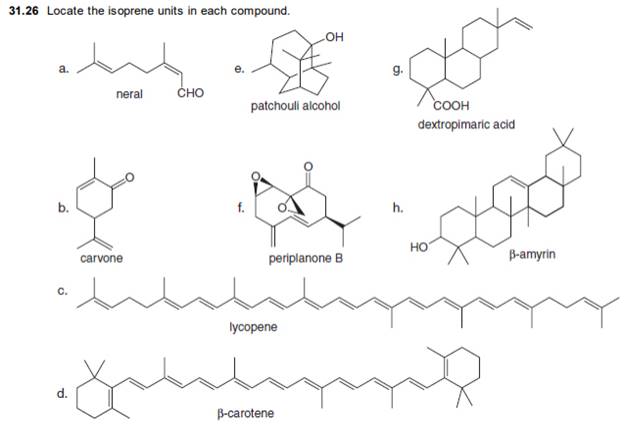
The isoprene units in the neral are highlighted by blue color as shown below.
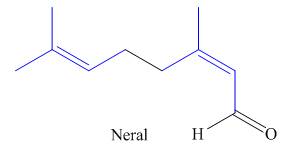
Figure 1
Explanation of Solution
The structure of isoprene is,
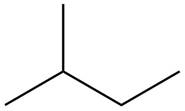
Figure 2
The isoprene unit contains four carbon atoms in the long chain and one carbon atom in branch. Isoprene unit in a terpene consists of carbon-carbon sigma or pi bonds.
The isoprene units in the given compound are highlighted by blue color as shown below.

Figure 1
The highlighted bonds represent the isoprene unit. There are two isoprene units present in the given compound.
The isoprene units in the neral are shown in Figure 1.
(b)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: Terpenes are naturally occurring compounds that are present in plants and animals. Terpenes contain one or more isoprene units. Terpenoids are derivatives of terpenes with oxygen containing functional group such as carbonyl groups.
Answer to Problem 31.26P
The isoprene units in the carvone are highlighted by blue color as shown below.
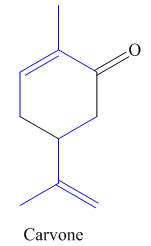
Figure 3
Explanation of Solution
The structure of isoprene is,
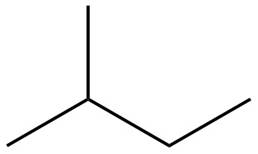
Figure 2
The isoprene unit contains four carbon atoms in the long chain and one carbon atom in branch. Isoprene unit in a terpene consists of carbon-carbon sigma or pi bonds.
The isoprene units in the given compound are highlighted by blue color as shown below.

Figure 3
The highlighted bonds represent the isoprene unit. There are two isoprene units present in the given compound.
The isoprene units in the carvone are shown in Figure 3.
(c)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: Terpenes are naturally occurring compounds that are present in plants and animals. Terpenes contain one or more isoprene units. Terpenoids are derivatives of terpenes with oxygen containing functional group such as carbonyl groups.
Answer to Problem 31.26P
The isoprene units in the lycopene are highlighted by blue color as shown below.
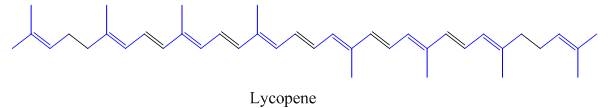
Figure 4
Explanation of Solution
The structure of isoprene is,
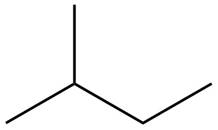
Figure 2
The isoprene unit contains four carbon atoms in the long chain and one carbon atom in branch. Isoprene unit in a terpene consists of carbon-carbon sigma or pi bonds.
The isoprene units in the given compound are are highlighted by blue color as shown below.

Figure 4
The highlighted bonds represent the isoprene unit. There are eight isoprene units present in the given compound.
The isoprene units in the lycopene are shown in Figure 4.
(d)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: Terpenes are naturally occurring compounds that are present in plants and animals. Terpenes contain one or more isoprene units. Terpenoids are derivatives of terpenes with oxygen containing functional group such as carbonyl groups.
Answer to Problem 31.26P
The isoprene units in the
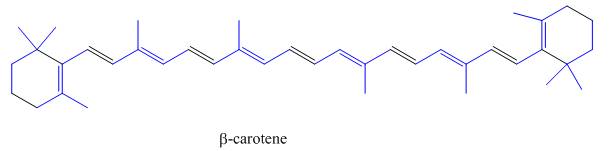
Figure 5
Explanation of Solution
The structure of isoprene is,
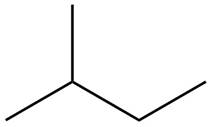
Figure 2
The isoprene unit contains four carbon atoms in the long chain and one carbon atom in branch. Isoprene unit in a terpene consists of carbon-carbon sigma or pi bonds.
The isoprene units in the given compound are highlighted by blue color as shown below.

Figure 5
The highlighted bonds represent the isoprene unit. There are eight isoprene units present in the given compound.
The isoprene units in the
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: Terpenes are naturally occurring compounds that are present in plants and animals. Terpenes contain one or more isoprene units. Terpenoids are derivatives of terpenes with oxygen containing functional group such as carbonyl groups.
Answer to Problem 31.26P
The isoprene units in the patchouli alcohol are highlighted by blue color as shown below.
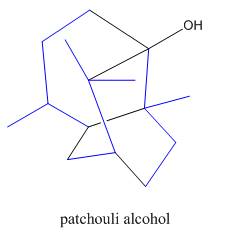
Figure 6
Explanation of Solution
The structure of isoprene is,
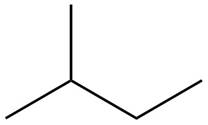
Figure 2
The isoprene unit contains four carbon atoms in the long chain and one carbon atom in branch. Isoprene unit in a terpene consists of carbon-carbon sigma or pi bonds.
The isoprene units in the given compound are are highlighted by blue color as shown below.

Figure 6
The highlighted bonds represent the isoprene unit. There are three isoprene units present in the given compound.
The isoprene units in the patchouli alcohol are shown in Figure 6.
(f)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: Terpenes are naturally occurring compounds that are present in plants and animals. Terpenes contain one or more isoprene units. Terpenoids are derivatives of terpenes with oxygen containing functional group such as carbonyl groups.
Answer to Problem 31.26P
The isoprene units in the periplanone B are highlighted by blue color as shown below.
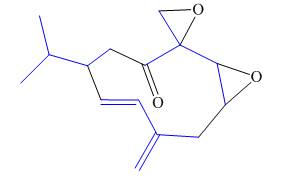
Figure 7
Explanation of Solution
The structure of isoprene is,

Figure 2
The isoprene unit contains four carbon atoms in the long chain and one carbon atom in branch. Isoprene unit in a terpene consists of carbon-carbon sigma or pi bonds.
The isoprene units in the given compound are highlighted by blue color as shown below.
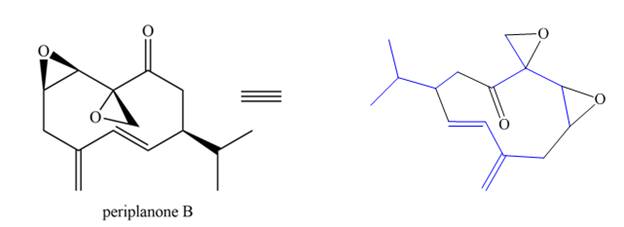
Figure 7
The highlighted bonds represent the isoprene unit. There are three isoprene units present in the given compound.
The isoprene units in the periplanone B are shown in Figure 7.
(g)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: Terpenes are naturally occurring compounds that are present in plants and animals. Terpenes contain one or more isoprene units. Terpenoids are derivatives of terpenes with oxygen containing functional group such as carbonyl groups.
Answer to Problem 31.26P
The isoprene units in the dextropimaric acid are highlighted by blue color as shown below.
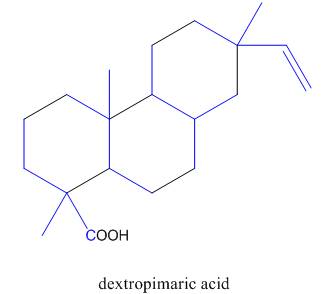
Figure 8
Explanation of Solution
The structure of isoprene is,

Figure 2
The isoprene unit contains four carbon atoms in the long chain and one carbon atom in branch. Isoprene unit in a terpene consists of carbon-carbon sigma or pi bonds.
The isoprene units in the given compound are shown below.

Figure 8
The highlighted bonds represent the isoprene unit. There are four isoprene units present in the given compound.
The isoprene units in the dextropimaric acid are shown in Figure 8.
(h)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: Terpenes are naturally occurring compounds that are present in plants and animals. Terpenes contain one or more isoprene units. Terpenoids are derivatives of terpenes with oxygen containing functional group such as carbonyl groups.
Answer to Problem 31.26P
The isoprene units in the
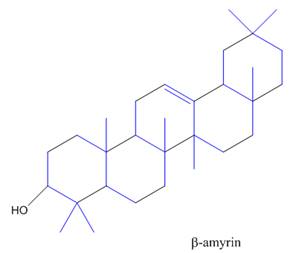
Figure 9
Explanation of Solution
The structure of isoprene is,

Figure 2
The isoprene unit contains four carbon atoms in the long chain and one carbon atom in branch. Isoprene unit in a terpene consists of carbon-carbon sigma or pi bonds.
The isoprene units in the given compound are shown below.
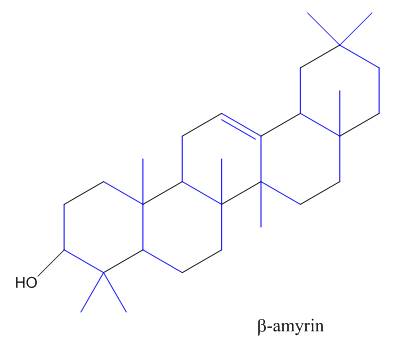
Figure 9
The highlighted bonds represent the isoprene unit. There are four isoprene units present in the given compound.
The isoprene units in the
Want to see more full solutions like this?
Chapter 31 Solutions
Organic Chemistry
- What is the final product when hexanedioic acid reacts with 1º PCl5 and 2º NH3.arrow_forwardWhat is the final product when D-galactose reacts with hydroxylamine?arrow_forwardIndicate the formula of the product obtained by reacting methyl 5-chloro-5-oxopentanoate with 1 mole of 4-penten-1-ylmagnesium bromide.arrow_forward
- The temperature on a sample of pure X held at 1.25 atm and -54. °C is increased until the sample boils. The temperature is then held constant and the pressure is decreased by 0.42 atm. On the phase diagram below draw a path that shows this set of changes. pressure (atm) 2 0 0 200 400 temperature (K) Xarrow_forwardQUESTION: Answer Question 5: 'Calculating standard error of regression' STEP 1 by filling in all the empty green boxes *The values are all provided in the photo attached*arrow_forwardpressure (atm) 3 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. 0 0 200 temperature (K) 400 аarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
