
Concept explainers
(a)
Interpretation:
The name, condensed structural formula and molecular mass of the given molecule should be drawn.
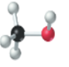
Concept introduction:
Name of the compound can be assigned according to below rule:
First, identify the number of carbon atoms present in chain, and then identify the function group if present in the given compound.
Condensed structural formula is defined as the structural formula which shows the spatial arrangement of bonds in an organic compound. In this structural formula, the bonds between the atoms are shown with lines and omit the bond between carbon and hydrogen atoms.
Molecular mass is defined as the sum of mass of each atom present in the compound.
(b)
Interpretation:
The name, condensed structural formula and molecular mass of the given molecule should be drawn.
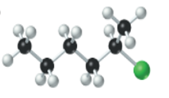
Concept introduction:
Name of the compound can be assigned according to below rule:
First, identify the number of carbon atoms present in chain, and then identify the function group if present in the given compound.
Condensed structural formula is defined as the structural formula which shows the spatial arrangement of bonds in an organic compound. In this structural formula, the bonds between the atoms are shown with lines and omit the bond between carbon and hydrogen atoms.
Molecular mass is defined as the sum of mass of each atom present in the compound.
(c)
Interpretation:
The name, condensed structural formula and molecular mass of the given molecule should be drawn.
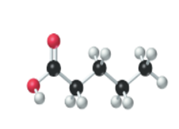
Concept introduction:
Name of the compound can be assigned according to below rule:
First, identify the number of carbon atoms present in chain, and then identify the function group if present in the given compound.
Condensed structural formula is defined as the structural formula which shows the spatial arrangement of bonds in an organic compound. In this structural formula, the bonds between the atoms are shown with lines and omit the bond between carbon and hydrogen atoms.
Molecular mass is defined as the sum of mass of each atom present in the compound.
(d)
Interpretation:
The name, condensed structural formula and molecular mass of the given molecule should be drawn.
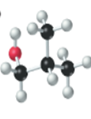
Concept introduction:
Name of the compound can be assigned according to below rule:
First, identify the number of carbon atoms present in chain, and then identify the function group if present in the given compound.
Condensed structural formula is defined as the structural formula which shows the spatial arrangement of bonds in an organic compound. In this structural formula, the bonds between the atoms are shown with lines and omit the bond between carbon and hydrogen atoms.
Molecular mass is defined as the sum of mass of each atom present in the compound.
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
General Chemistry: Principles and Modern Applications (11th Edition)
- Identify the compound with the longest carbon - nitrogen bond. O CH3CH2CH=NH O CH3CH2NH2 CH3CH2C=N CH3CH=NCH 3 The length of all the carbon-nitrogen bonds are the samearrow_forwardIdentify any polar covalent bonds in epichlorohydrin with S+ and 8- symbols in the appropriate locations. Choose the correct answer below. Η H's+ 6Η Η Η Η Η Ηδ Η Ο Ο HH +Η Η +Η Η Η -8+ CIarrow_forwardH H:O::::H H H HH H::O:D:D:H HH HH H:O:D:D:H .. HH H:O:D:D:H H H Select the correct Lewis dot structure for the following compound: CH3CH2OHarrow_forward
- Rank the following compounds in order of decreasing boiling point. ннннн -С-С-Н . н-с- ННННН H ΗΤΗ НННН TTTĪ н-с-с-с-с-о-н НННН НН C' Н н-с-с-с-с-н НН || Ш НННН H-C-C-C-C-N-H ННННН IVarrow_forwardRank the following compounds in order of decreasing dipole moment. |>||>||| ||>|||>| |>|||>|| |||>||>| O ||>>||| H F H F H c=c || H c=c F F IIIarrow_forwardchoose the description that best describes the geometry for the following charged species ch3-arrow_forward
- Why isn't the ketone in this compound converted to an acetal or hemiacetal by the alcohol and acid?arrow_forwardWhat is the approximate bond angle around the nitrogen atom? HNH H Harrow_forwardOH 1. NaOCH2CH3 Q 2. CH3CH2Br (1 equiv) H3O+ Select to Draw 1. NaOCH2 CH3 2. CH3Br (1 equiv) heat Select to Edit Select to Drawarrow_forward
- Complete and balance the following half-reaction in acidic solution. Be sure to include the proper phases for all species within the reaction. S₂O₃²⁻(aq) → S₄O₆²⁻(aq)arrow_forwardQ Select to Edit NH3 (CH3)2CHCI (1 equiv) AICI 3 Select to Draw cat. H2SO4 SO3 (1 equiv) HO SOCl2 pyridine Select to Edit >arrow_forwardComplete and balance the following half-reaction in basic solution. Be sure to include the proper phases for all species within the reaction. Zn(s) → Zn(OH)₄²⁻(aq)arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning  Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning





