
Package: Loose Leaf for Organic Chemistry with Biological Topics with Connect Access Card
5th Edition
ISBN: 9781260170405
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 28, Problem 28.58P
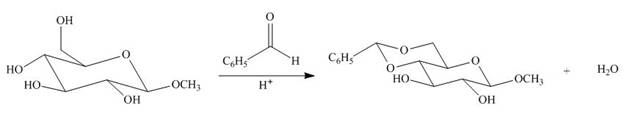
Draw a stepwise mechanism for the following reaction.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
9. Write Me product as well as the reaction
Mechanism For each of the Following Veritious
+H2504
4.50+
+ 1/₂ Felly
◎+
7
b. Praw he potential energy Diagrams For each
OF Mese Ronctions and account for any
differences that appeak in the two potential
Puergy Diagrams
Draw the major product of this reaction. Ignore inorganic
byproducts.
Incorrect, 3 attempts remaining
1. excess Br2, NaOH
2. neutralizing workup
Q
Given the electrode Pt | Ag | Ag+ (aq), describe it.
Chapter 28 Solutions
Package: Loose Leaf for Organic Chemistry with Biological Topics with Connect Access Card
Ch. 28 - Prob. 28.1PCh. 28 - Prob. 28.2PCh. 28 - Label each stereogenic center as R or S. a. b. c....Ch. 28 - Convert the ball-and-stick model to a Fischer...Ch. 28 - Prob. 28.5PCh. 28 - Prob. 28.6PCh. 28 - Prob. 28.7PCh. 28 - Prob. 28.8PCh. 28 - Prob. 28.9PCh. 28 - Prob. 28.10P
Ch. 28 - Prob. 28.11PCh. 28 - Prob. 28.12PCh. 28 - Prob. 28.13PCh. 28 - Prob. 28.14PCh. 28 - Prob. 28.15PCh. 28 - Prob. 28.16PCh. 28 - Prob. 28.17PCh. 28 - Draw a stepwise mechanism for the following...Ch. 28 - Prob. 28.19PCh. 28 - Prob. 28.20PCh. 28 - Prob. 28.21PCh. 28 - Prob. 28.22PCh. 28 - Draw the products formed when D-arabinose is...Ch. 28 - Prob. 28.24PCh. 28 - Prob. 28.25PCh. 28 - Prob. 28.26PCh. 28 - Prob. 28.27PCh. 28 - Prob. 28.28PCh. 28 - Prob. 28.29PCh. 28 - Prob. 28.30PCh. 28 - Prob. 28.31PCh. 28 - Prob. 28.32PCh. 28 - Prob. 28.33PCh. 28 - Prob. 28.34PCh. 28 - Problem-28.35
Draw the structures of the...Ch. 28 - Prob. 28.36PCh. 28 - 28.37 Convert each ball-and-stick model to a...Ch. 28 - Prob. 28.38PCh. 28 - Prob. 28.39PCh. 28 - 28.40 Convert each compound to a Fischer...Ch. 28 - Prob. 28.41PCh. 28 - Prob. 28.42PCh. 28 - 28.43 Draw a Haworth projection for each compound...Ch. 28 - Prob. 28.44PCh. 28 - 28.45 Draw both pyranose anomers of each...Ch. 28 - Prob. 28.46PCh. 28 - Prob. 28.47PCh. 28 - Prob. 28.48PCh. 28 - Prob. 28.49PCh. 28 - 28.50 Draw the products formed when D-altrose is...Ch. 28 - Prob. 28.51PCh. 28 - Prob. 28.52PCh. 28 - Prob. 28.53PCh. 28 - Prob. 28.54PCh. 28 - Prob. 28.55PCh. 28 - Prob. 28.56PCh. 28 - Prob. 28.57PCh. 28 - 28.58 Draw a stepwise mechanism for the following...Ch. 28 - Prob. 28.59PCh. 28 - Prob. 28.60PCh. 28 - Prob. 28.61PCh. 28 - Prob. 28.62PCh. 28 - Prob. 28.63PCh. 28 - Prob. 28.64PCh. 28 - Prob. 28.65PCh. 28 - Prob. 28.66PCh. 28 - Prob. 28.67PCh. 28 - Prob. 28.68PCh. 28 - Prob. 28.69PCh. 28 - Prob. 28.70PCh. 28 - 28.71 Draw a stepwise mechanism for the following...
Additional Science Textbook Solutions
Find more solutions based on key concepts
More than one choice may apply. Using the terms listed below, fill in the blank with the proper term. anterior ...
Essentials of Human Anatomy & Physiology (12th Edition)
To test your knowledge, discuss the following topics with a study partner or in writing ideally from memory. Th...
HUMAN ANATOMY
What were the major microbiological interests of Martinus Beijerinck and Sergei Winogradsky? It can be said tha...
Brock Biology of Microorganisms (15th Edition)
2. Which of the following is the best example of the use of a referent? _
a. A red bicycle
b. Big as a dump tru...
Physical Science
Label each statement about the polynucleotide ATGGCG as true or false. The polynucleotide has six nucleotides. ...
General, Organic, and Biological Chemistry - 4th edition
11. In the early 1800s, French naturalist Jean Baptiste Lamarck suggested that the best explanation for the rel...
Campbell Biology: Concepts & Connections (9th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- At 25°C, the reaction Zn2+ + 2e ⇄ Zn has a normal equilibrium potential versus the saturated calomel electrode of -1.0048 V. Determine the normal equilibrium potential of Zn versus the hydrogen electrode.Data: The calomel electrode potential is E° = 0.2420 V versus the normal hydrogen electrode.arrow_forwardElectrochemistry. State the difference between E and E0.arrow_forwardIn an electrolytic cell, the positive pole is always assumed to be on the right side of the battery notation. Is that correct?arrow_forward
- In an electrolytic cell, the positive pole is always assumed to be on the right side of the battery. Is that correct?arrow_forwardCalculate the free energy of formation of 1 mol of Cu in cells where the electrolyte is 1 mol dm-3 Cu2+ in sulfate solution, pH 0. E° for the Cu2+/Cu pair in this medium is +142 mV versus ENH.Assume the anodic reaction is oxygen evolution.Data: EH2 = -0.059 pH (V) and EO2 = 1.230 - 0.059 pH (V); 2.3RT/F = 0.059 Varrow_forwardIf the normal potential for the Fe(III)/Fe(II) pair in acid at zero pH is 524 mV Hg/Hg2Cl2 . The potential of the saturated calomel reference electrode is +246 mV versus the NHE. Calculate E0 vs NHE.arrow_forward
- Given the galvanic cell whose scheme is: (-) Zn/Zn2+ ⋮⋮ Ag+/Ag (+). If we know the normal potentials E°(Zn2+/Zn) = -0.76V and E°(Ag+/Ag) = 0.799 V. Indicate the electrodes that are the anode and the cathode and calculate the E0battery.arrow_forwardIndicate the functions that salt bridges have in batteries.arrow_forwardIn the battery:Pt | H2 (g) | H+ (aq) | Fe2+ (aq) | FeIndicate the cathode and anode.arrow_forward
- Write the equations that occur when the electrode Pb (s) | PbI2 (s) | KI (ac) in a galvanic cell. a) It functions as a positive electrode b) It functions as a negative electrode c) What is the ion with respect to which this electrode is reversible?arrow_forwardState the formula to find the electromotive force of a battery as a function of the potential of the anode and the cathode.arrow_forwardWhy are normal electrode potentials also called relative electrode potentials?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY