
Concept explainers
(a)
Interpretation: The given compound is to be classified as identical to
Concept introduction: The stereochemistry of the compound is determined by prioritizing the groups attached to its stereogenic center. The groups are prioritized on the basis of
Answer to Problem 28.39P
The given compound is classified as identical to
Explanation of Solution
The structure of compound
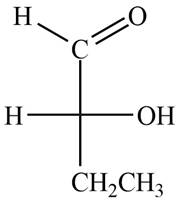
Figure 1
In the compound
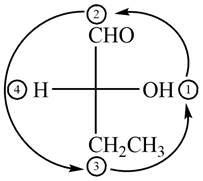
Figure 2
The circle rotates in the anticlockwise direction and its configuration will be
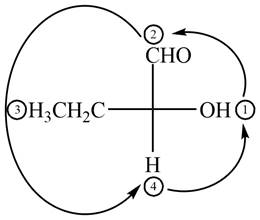
Figure 3
Thus, the configuration will reverse. Therefore, the compound
The given compound is,
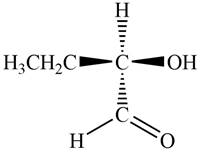
Figure 4
In the given compound,
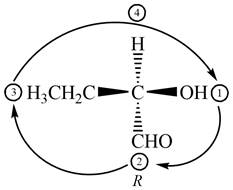
Figure 5
The circle rotates in the clockwise direction. Thus, the given compound is labeled as
Hence, the given compound is classified as identical to
The given compound is classified as identical to
(b)
Interpretation: The given compound is to be classified as identical to
Concept introduction: The stereochemistry of the compound is determined by prioritizing the groups attached to its stereogenic center. The groups are prioritized on the basis of atomic number of their atoms. The group that contain atom with higher atomic number is given higher priority. Complete the circle in decreasing order of priority from
Answer to Problem 28.39P
The given compound is classified as identical to
Explanation of Solution
The compound
The given compound is,
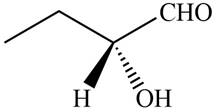
Figure 6
In the given compound,
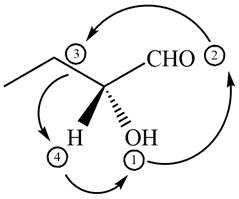
Figure 7
The circle rotates in the anticlockwise direction hence its configuration will be
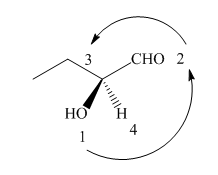
Figure 8
Thus, the configuration will reverse. Therefore, the compound is labeled as
Hence, the given compound is classified as identical to
The given compound is classified as identical to
(c)
Interpretation: The given compound is to be classified as identical to
Concept introduction: The stereochemistry of the compound is determined by prioritizing the groups attached to its stereogenic center. The groups are prioritized on the basis of atomic number of their atoms. The group that contain atom with higher atomic number is given higher priority. Complete the circle in decreasing order of priority from
Answer to Problem 28.39P
The given compound is classified as enantiomer to
Explanation of Solution
The compound
The given compound is,
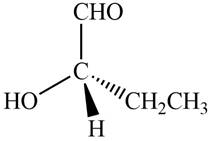
Figure 9
In the given compound,
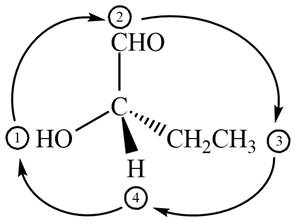
Figure 10
The circle rotates in the clockwise direction hence its configuration will be
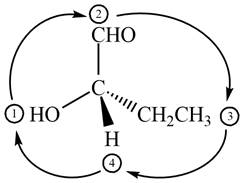
Figure 11
Thus, the configuration will reverse. Therefore, the compound is labeled as
The given compound and
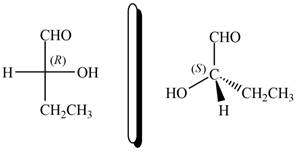
Figure 12
Hence, the given compound is classified as enantiomer to
The given compound is classified as enantiomer to
Want to see more full solutions like this?
Chapter 28 Solutions
Package: Loose Leaf for Organic Chemistry with Biological Topics with Connect Access Card
- Q6: Predict the major product(s) for the following reactions. Note the mechanism (SN1, SN2, E1 or E2) the reaction proceeds through. If no reaction takes place, indicate why. Pay attention to stereochemistry. NaCN DMF Br σ Ilm... Br H Br H H NaCN CH3OH KOtBu tBuOH NaBr H₂O LDA Et2O (CH3)2CHOH KCN DMSO NaOH H₂O, A LDA LDA Systemarrow_forwardQ7: For the following reactions, indicate the reaction conditions that would provide the indicated product in a high yield. Note the major reaction pathway that would take place (SN1, SN2, E1, or E2) Note: There may be other products that are not shown. There maybe more than one plausible pathway. Br H3C OH H3C CI ... H3C SCH2CH3 CI i SCH2CH3 ཨ་ Br System Settarrow_forwardQ2: Rank the compounds in each of the following groups in order of decreasing rate of solvolysis in aqueous acetone. OSO2CF3 OSO2CH3 OH a. b. CI Brarrow_forward
- ох 4-tert-butyl oxy cyclohex-1-ene Incorrect, 1 attempt remaining The systematic name of this compound classifies the -OR group as a substituent of the hydrocarbon, which is considered the principal functional group. The ether substituent is named with the suffix 'oxy'. The general format for the systematic name of a hydrocarbon is: [prefix/substituent] + [parent] + [functional group suffix] Substituents are listed in alphabetical order. Molecules with a chiral center will indicate the absolute configuration at the beginning of its name with the R and S notation.arrow_forward5. Compressibility (6 points total). The isothermal compressibility is a measure of how hard/easy it is to compress an object (how squishy is it?) at constant temperature. It is др defined as Br=-()=-(200²)T' (a) You might wonder why there is a negative sign in this formula. What does it mean when this quantity is positive and what does it mean when this quantity is negative? (b) Derive the formula for the isothermal compressibility of an ideal gas (it is very simple!) (c) Explain under what conditions for the ideal gas the compressibility is higher or lower, and why that makes sense.arrow_forward19. (3 pts) in Chapter 7 we will see a reaction of halocyclohexanes that requires that the halogen occupy an axial position with this in mind, would you expect cis-1-bromo-3-methylcyclohexane or trans-1-bromo-3-methylcyclohexane to be more reactive in this reaction? Briefly explain your choice using structures to support your answer. Mere-eries-cecleone) The tran-i-browse-3-methylcyclohexionearrow_forward
- Please help me calculate the undiluted samples ppm concentration. My calculations were 280.11 ppm. Please see if I did my math correctly using the following standard curve. Link: https://mnscu-my.sharepoint.com/:x:/g/personal/vi2163ss_go_minnstate_edu/EVSJL_W0qrxMkUjK2J3xMUEBHDu0UM1vPKQ-bc9HTcYXDQ?e=hVuPC4arrow_forwardProvide an IUPAC name for each of the compounds shown. (Specify (E)/(Z) stereochemistry, if relevant, for straight chain alkenes only. Pay attention to commas, dashes, etc.) H₁₂C C(CH3)3 C=C H3C CH3 CH3CH2CH CI CH3 Submit Answer Retry Entire Group 2 more group attempts remaining Previous Nextarrow_forwardArrange the following compounds / ions in increasing nucleophilicity (least to most nucleophilic) CH3NH2 CH3C=C: CH3COO 1 2 3 5 Multiple Choice 1 point 1, 2, 3 2, 1, 3 3, 1, 2 2, 3, 1 The other answers are not correct 0000arrow_forward
- curved arrows are used to illustrate the flow of electrons. using the provided starting and product structures, draw the cured electron-pushing arrows for thw following reaction or mechanistic steps. be sure to account for all bond-breaking and bond making stepsarrow_forwardUsing the graphs could you help me explain the answers. I assumed that both graphs are proportional to the inverse of time, I think. Could you please help me.arrow_forwardSynthesis of Dibenzalacetone [References] Draw structures for the carbonyl electrophile and enolate nucleophile that react to give the enone below. Question 1 1 pt Question 2 1 pt Question 3 1 pt H Question 4 1 pt Question 5 1 pt Question 6 1 pt Question 7 1pt Question 8 1 pt Progress: 7/8 items Que Feb 24 at You do not have to consider stereochemistry. . Draw the enolate ion in its carbanion form. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. ⚫ Separate multiple reactants using the + sign from the drop-down menu. ? 4arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
