
Tutorials in Introductory Physics
1st Edition
ISBN: 9780130970695
Author: Peter S. Shaffer, Lillian C. McDermott
Publisher: Addison Wesley
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 27.1, Problem 2bTH
A long pin is used to hold the piston in place as shown in the diagram. The cylinder is then placed into boiling water.
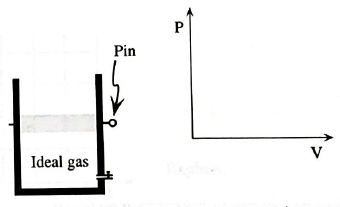
b. Sketch this process in the PV diagram at right.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
!
Required information
The radius of the Moon is 1.737 Mm and the distance between Earth and the Moon is 384.5 Mm.
The intensity of the moonlight incident on her eye is 0.0220 W/m². What is the intensity incident on her retina if the
diameter of her pupil is 6.54 mm and the diameter of her eye is 1.94 cm?
W/m²
Required information
An object is placed 20.0 cm from a converging lens with focal length 15.0 cm (see the figure, not drawn to scale). A
concave mirror with focal length 10.0 cm is located 76.5 cm to the right of the lens. Light goes through the lens, reflects
from the mirror, and passes through the lens again, forming a final image.
Converging
lens
Object
Concave
mirror
15.0 cm
-20.0 cm-
10.0 cm
d cm
d = 76.5.
What is the location of the final image?
cm to the left of the lens
!
Required information
A man requires reading glasses with +2.15-D refractive power to read a book held 40.0 cm away with a relaxed eye.
Assume the glasses are 1.90 cm from his eyes.
His uncorrected near point is 1.00 m. If one of the lenses is the one for distance vision, what should the refractive power of the other
lens (for close-up vision) in his bifocals be to give him clear vision from 25.0 cm to infinity?
2.98 D
Chapter 27 Solutions
Tutorials in Introductory Physics
Ch. 27.1 - Prob. 1aTHCh. 27.1 - In this process, which of the quantities P, V, n,...Ch. 27.1 - Consider the following incorrect student...Ch. 27.1 - Explain why it is not possible to use the ideal...Ch. 27.1 - A long pin is used to hold the piston in place as...Ch. 27.1 - A long pin is used to hold the piston in place as...Ch. 27.1 - Prob. 2cTHCh. 27.2 - Prob. 1aTHCh. 27.2 - Prob. 1bTHCh. 27.2 - Prob. 1cTH
Additional Science Textbook Solutions
Find more solutions based on key concepts
4. Three groups of nonvascular plants are _______, ______, and _______. Three groups of seedless vascular plant...
Biology: Life on Earth (11th Edition)
29. For the reaction
determine the expression for the rate of the reaction in terms of the change in concentr...
Chemistry: Structure and Properties (2nd Edition)
What is the anatomical position? Why is it important that you learn this position?
Anatomy & Physiology (6th Edition)
2. What are the primary functions of the skeletal system?
Human Anatomy & Physiology (2nd Edition)
Name the components (including muscles) of the thoracic cage. List the contents of the thorax.
Human Physiology: An Integrated Approach (8th Edition)
5.28 Neurofibromatosis is an autosomal dominant disorder inherited on human chromosome. Part of the analysis ma...
Genetic Analysis: An Integrated Approach (3rd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- ! Required information Assume that the magnifier is held close to the eye. Use the standard near point of 25.0 cm to find the angular magnification. An insect that is 4.10 mm long is placed 10.3 cm from a simple magnifier with a focal length of 13.0 cm. What is the angular magnification?arrow_forward2arrow_forward3arrow_forward
- Imagine you are out for a stroll on a sunny day when you encounter a lake. Unpolarized light from the sun is reflected off the lake into your eyes. However, you notice when you put on your vertically polarized sunglasses, the light reflected off the lake no longer reaches your eyes. What is the angle between the unpolarized light and the surface of the water, in degrees, measured from the horizontal? You may assume the index of refraction of air is nair=1 and the index of refraction of water is nwater=1.33 . Round your answer to three significant figures. Just enter the number, nothing else.arrow_forwardDeduce what overvoltage is like in reversible electrodes.arrow_forwardpls help on thesearrow_forward
- pls help on thesearrow_forward20. Two small conducting spheres are placed on top of insulating pads. The 3.7 × 10-10 C sphere is fixed whie the 3.0 × 107 C sphere, initially at rest, is free to move. The mass of each sphere is 0.09 kg. If the spheres are initially 0.10 m apart, how fast will the sphere be moving when they are 1.5 m apart?arrow_forwardpls help on allarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning


Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

College Physics
Physics
ISBN:9781938168000
Author:Paul Peter Urone, Roger Hinrichs
Publisher:OpenStax College

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers
Physics
ISBN:9781337553278
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers with Modern ...
Physics
ISBN:9781337553292
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning
Thermodynamics: Crash Course Physics #23; Author: Crash Course;https://www.youtube.com/watch?v=4i1MUWJoI0U;License: Standard YouTube License, CC-BY