
Organic Chemistry, Books a la Carte Edition (8th Edition)
8th Edition
ISBN: 9780134074580
Author: Bruice, Paula Yurkanis
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 26, Problem 30P
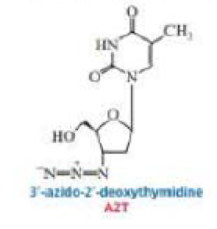
Human immunodeficiency virus (HIV) is the retrovirus that causes AIDS. AZT was one of the first drugs designed to interfere with retroviral DNA synthesis. When cells take up AZT, they convert it to AZT-triphosphate. Explain how AZT interferes with DNA synthesis.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
How do I calculate the amount of quarks in magnesium?
Please provide the mechanism for the reaction attached. Please include all arrows, intermediates, and formalcharges. If a Sigma complex, please draw all major resonance forms.
Predict the product or products for the following reactions. Please include both ortho and para substitutions, if it applies, and indicate the major product, if it applies.
Chapter 26 Solutions
Organic Chemistry, Books a la Carte Edition (8th Edition)
Ch. 26.1 - In acidic solutions, nucleosides are hydrolyzed to...Ch. 26.1 - Draw the structure for each of the following: a....Ch. 26.3 - Indicate whether each functional group of the five...Ch. 26.3 - Prob. 4PCh. 26.3 - If one of the strands of DNA has the following...Ch. 26.4 - The 2',3-cyclic phosphodiester that is formed...Ch. 26.5 - Using a dark line for the original parental DNA...Ch. 26.7 - Why do both thymine and uracil specify the...Ch. 26.9 - If methionine is always the first amino acid...Ch. 26.9 - Four Cs occur in a row in the segment of mRNA...
Ch. 26.9 - UAA is a stop codon. Why does the UAA sequence in...Ch. 26.9 - Prob. 12PCh. 26.9 - Write the sequences of bases in the sense strand...Ch. 26.9 - List the possible codons on mRNA that speciy each...Ch. 26.10 - Adenine can be deaminated to hypoxanthine, and...Ch. 26.10 - Explain why thymine cannot be deaminated.Ch. 26.12 - Which of the following base sequences is most...Ch. 26 - What nonapeptide is coded for by the following...Ch. 26 - What is the sequence of bases in the template...Ch. 26 - Prob. 20PCh. 26 - A segment of DNA has 18 base pairs. It has 7...Ch. 26 - Name the following:Ch. 26 - Prob. 23PCh. 26 - Propose a mechanism for the following reaction:Ch. 26 - The first amino acid incorporated into a...Ch. 26 - Match the codon with the anticodon:Ch. 26 - a. Using the single-letter abbreviations for the...Ch. 26 - Which of the following pairs of dinucleotides are...Ch. 26 - Human immunodeficiency virus (HIV) is the...Ch. 26 - If a mRNA contained only U and G in random...Ch. 26 - Why is the codon a triplet rather than a doublet...Ch. 26 - RNAase, the enzyme that catalyzes the hydrolysis...Ch. 26 - The amino acid sequences of peptide fragments...Ch. 26 - Which cytosine in the following sense strand of...Ch. 26 - Prob. 36PCh. 26 - Sodium nitrite, a common food preservative (page...Ch. 26 - Why does DNA not unravel completely before...Ch. 26 - Staphylococcus nuclease is an enzyme that...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The bromination of naphthalene via electrophilic aromatic substitution. Please draw out all of the resonance structures created from addition at the C1 carbon. Please also draw out all of the resonance structures created from addition at the C2 carbon. Which carbon (C1 or C2) is more favored?arrow_forwardPredict the product or products for the following reactions. Please include both ortho and para substitutions, if it applies, and indicate the major product, if it applies.arrow_forwardChapter 1 In-Class-Problem set ☺ Study The C3-C4 carbon-carbon bond in the following molecule results from the overlap of which orbitals (in the order C3-C4)? о a) sp-sp² b) sp-sp³ c) sp²-sp² d) sp²-sp³ e) sp³-sp² 7 6 M 4 3 2arrow_forward
- Count how many pi and sigma bonds are in the following molecule, caffeine. Circle all sp3 hybridized atoms, box sp₂ hybridized atoms, and draw a triangle around all sp hybridized atoms. N N N Z - - N -arrow_forwardFill in the blanks of the following chart Characteristic Atomic Number Symbol amul Description Protons + Neutrons Use the following words to label the diagrams: Atomic Number, Valence State, Chemical Symbol, Number of Atoms, Molecule, Atomic Mass 40Ca 74W H₂O Na+¹ CI-1arrow_forwardin aqueous solution, bromine oxidises sulphur dioxide so2 to sulphate ions so42- i) deduce the oxidation state of sulphur in so2 and so4 2- ii) deduce a half equation for the reduction of bromine in an aqueous solution iii) deduce a hald equation for the oxidatio of so2 in aquoes solution forming so42- and h+ ionsarrow_forward
- Please correct answer and don't used hand raiting and don't used Ai solutionarrow_forwardDraw orbitals for a and c and identify all of the molecules/functional groups' electron geometry, molecular shape and bond angles: a. H₂O b. Ketone c. Alkyne d. Etherarrow_forwardIdentify the functional groups in the following molecule, naxalone (aka narcan): HO OH Narrow_forward
- Benzene-toluene equilibrium is often approximated as αBT = 2.34. Generate the y-x diagram for this relative volatility. Also, generate the equilibrium data using Raoult’s law, and compare your results to these. post excel spreadsheet w values used to generate both graphsarrow_forwardConvert the following structures to skeletal structures: a. CH3CH2CH2CH2COOH b. CH3CH2OCH2 Br C. CH2CHCHCHCCCHCH2arrow_forwardConvert the following structures to condensed structures: OH a. b. Cl C. Harrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning
Nucleic acids - DNA and RNA structure; Author: MEDSimplified;https://www.youtube.com/watch?v=0lZRAShqft0;License: Standard YouTube License, CC-BY