
(a)
Interpretation:
The structure of the two isomeric propyl radicals that are formed on hydrogen abstraction from propane by
Concept introduction:
Radicals are species containing an unpaired electron, i.e., they are electron-poor species. Being electron-poor, they are highly unstable, and therefore, highly reactive species. They react with any other species present.
In the case of
Answer to Problem 25.42P
The two alkyl radicals produced when
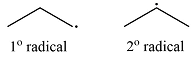
Explanation of Solution
Propane has two types of hydrogens, one attached to a primary carbon and one to a secondary carbon. The abstraction of the two types of hydrogens will produce two radicals – a primary propyl radical and a secondary propyl radical.
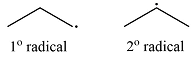
The type of radical produced when
(b)
Interpretation:
The curved arrow notation for the formation of the two isomeric propyl radicals that are formed on hydrogen abstraction from propane by
Concept introduction:
In the case of alkanes, a radical like
Curved arrow notation represents the movement of electron(s) in a reaction or resonance. Movement of a single electron is represented by a curved arrow with a half arrowhead. The arrow starts at the electron and ends on the atom or the region where it moves.
Answer to Problem 25.42P
The curved arrow notation for the formation of the two alkyl radicals produced when
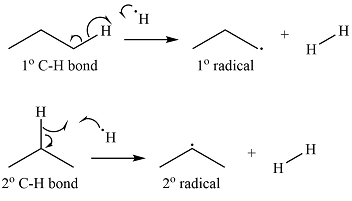
Explanation of Solution
Propane has two types of hydrogens, one attached to a primary carbon and one to a secondary carbon. The abstraction of the two types of hydrogens will produce two radicals – a primary propyl radical and a secondary propyl radical.
The
Thus, the curved arrow representation of the formation of the two radicals can be shown as

The curved arrow representation of the formation of the two radicals is drawn using half arrows on the basis of the type of hydrogen abstracted.
(c)
Interpretation:
The percentage of each propyl radical formed is to be calculated taking into account the different numbers of each type of hydrogen in propane.
Concept introduction:
The ease with which a hydrogen is abstracted from an alkane in its reaction with a radical depends on the relative stability of the type of alkyl radical that is produced. The relative stability of alkyl radicals changes as
The likelihood of abstraction of a particular type of hydrogen from an alkane also increases with the number of hydrogens of that type present in the molecule.
The percentage of formation of a particular type of alkyl radical is calculated from the product of the number of that type of hydrogens present in the molecule and its reactivity.
Answer to Problem 25.42P
The percentages of the
Explanation of Solution
The percentage of formation of each type of alkyl radical is calculated from the product of the number of that type of hydrogens present in the molecule and its reactivity.
There are six primary hydrogens and two secondary hydrogens in propane, and the reactivities of the two types are 1 and 5 respectively.
Thus, the percentages of the
The percentage of each type of radical produced is calculated from the number of hydrogens of each type and their relative reactivities.
Want to see more full solutions like this?
Chapter 25 Solutions
ORG CHEM W/ EBOOK & SW5 + STUDY GUIDE
- Would the following organic synthesis occur in one step? Add any missing products, required catalysts, inorganic reagents, and other important conditions. Please include a detailed explanation and drawings showing how the reaction may occur in one step.arrow_forward(a) Sketch the 'H NMR of the following chemical including the approximate chemical shifts, the multiplicity (splitting) of all signals and the integration (b) How many signals would you expect in the 13C NMR? CH3arrow_forwardDraw the Show the major and minor product(s) for the following reaction mechanisms for both reactions and show all resonance structures for any Explain why the major product is favoured? intermediates H-Brarrow_forward
- 3. Draw ALL THE POSSBILE PRODUCTS AND THE MECHANISMS WITH ALL RESONANCE STRUCTURES. Explain using the resonance structures why the major product(s) are formed over the minor product(s). H₂SO4, HONO CHarrow_forward7. Provide the product(s), starting material(s) and/or condition(s) required for the No mechanisms required. below reaction HO + H-I CI FO Br2, FeBr3 O I-Oarrow_forward6. Design the most efficient synthesis of the following product starting from phenot Provide the reaction conditions for each step (more than one step is required) and explain the selectivity of each reaction. NO MECHANISMS ARE REQUIRED. OH step(s) CIarrow_forward
- What is the skeletal structure of the product of the following organic reaction?arrow_forwardIf a reaction occurs, what would be the major products? Please include a detailed explanation as well as a drawing showing how the reaction occurs and what the final product is.arrow_forwardWhat is the major organic product of the following nucleophilic acyl substitution reaction of an acid chloride below?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





