
Organic Chemistry
7th Edition
ISBN: 9780321803221
Author: Paula Y. Bruice
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 24.3, Problem 9P
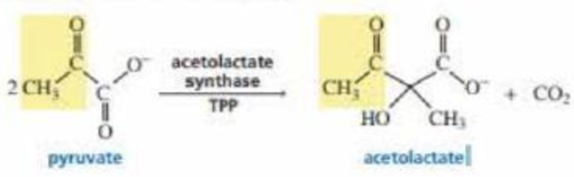
Acetolactate synthase is another TPP-requiring enzyme. It transfers the acyl group to another molecule of pyruvate, forming acetolactate. This is the first step in the biosynthesis of the amino acids valine and leucine. Propose a mechanism for this reaction.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
You have started a patient on a new drug. Each dose introduces 40 pg/mL of drug after redistribution and prior to elimination. This drug is administered at 24 h intervals and has a half life of 24 h. What will the concentration of drug be after each of the first six doses? Show your work
a. What is the concentration after the first dose? in pg/mL
b. What is the concentration after the second dose? in pg/mL
c. What is the concentration after the third dose? in pg/mL
How many different molecules are drawn below?
Only 100% sure experts solve it correct complete solutions need to get full marks it's my quiz okkkk.take your time but solve full accurate okkk chemistry expert solve it.qno4
Chapter 24 Solutions
Organic Chemistry
Ch. 24.1 - Prob. 2PCh. 24.1 - Prob. 3PCh. 24.2 - How many conjugated double bonds are there in a....Ch. 24.2 - Instead of adding to the 4a position and...Ch. 24.2 - Prob. 7PCh. 24.3 - Prob. 8PCh. 24.3 - Acetolactate synthase is another TPP-requiring...Ch. 24.3 - Acetolactate synthase transfers the acyl group of...Ch. 24.5 - Prob. 12PCh. 24.5 - The enzyme that catalyzes the C C bond cleavage...
Ch. 24.5 - Propose a mechanism for the ,-elimination reaction...Ch. 24.5 - Which compound is more easily decarboxylated?Ch. 24.5 - Explain why the ability of PLP to catalyze an...Ch. 24.5 - Explain why the ability of PLP to catalyze an...Ch. 24.6 - Ethanolamine ammonia lyase, a coenzyme...Ch. 24.6 - Prob. 19PCh. 24.7 - How do the structure of tetrahydrofolate and...Ch. 24.7 - What is the source of the methyl group in...Ch. 24.8 - Thiols such as ethanethiol and propanethiol can be...Ch. 24 - How does the metal ion in carboxypeptidase A...Ch. 24 - Prob. 23PCh. 24 - Prob. 24PCh. 24 - Prob. 25PCh. 24 - S-Adenosylmethionine (SAM) is formed from the...Ch. 24 - Prob. 27PCh. 24 - What acyl groups have we seen transferred by...Ch. 24 - Propose a mechanism for the following reaction:Ch. 24 - When transaminated, the three branched-chain amino...Ch. 24 - Draw the products of the following reaction, where...Ch. 24 - When UMP is dissolved in T2O, exchange of T for H...Ch. 24 - Dehydratase is a PLP-requiring enzyme that...Ch. 24 - In addition to the reaction mentioned in Section...Ch. 24 - PLP can catalyze both ,-elimination reactions...Ch. 24 - The glycine cleavage system is a group of four...Ch. 24 - Prob. 37PCh. 24 - FADH2 reduces , -unsaturated thioesters to...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Nonearrow_forwardA complete tensile test was performed on a magnesium specimen of 12 mm diameter and 30 mm length, until breaking. The specimen is assumed to maintain a constant volume. Calculate the approximate value of the actual stress at breaking. TABLE. The tensile force F and the length of the specimen are represented for each L until breaking. F/N L/mm 0 30,0000 30,0296 5000 10000 30,0592 15000 30,0888 20000 30,15 25000 30,51 26500 30,90 27000 31,50 26500 32,10 25000 32,79arrow_forwardNonearrow_forward
- Differentiate between plastic deformation, elastic deformation, viscoelastic deformation and viscoplastic deformation.arrow_forward1.57 Draw all reasonable resonance structures for the following cation. Then draw the resonance hybrid.arrow_forwardFor the two questions below, draw the mechanism and form the major product.arrow_forward
- Indicate similarities and differences between natural, exchanged and pillared clays.arrow_forwardShow work. don't give Ai generated solutionarrow_forwardIn intercalation compounds, their sheets can be neutral or have a negative or positive charge, depending on the nature of the incorporated species and its structure. Is this statement correct?arrow_forward
- This thermodynamic cycle describes the formation of an ionic compound MX2 from a metal element M and nonmetal element X in their standard states. What is the lattice enthalpy of MX2 ? What is the enthalpy formation of MX2 ? Suppose both the heat of sublimation of M and the ionization enthalpy of M were smaller. Would MX2 be more stable? Or less? or impossible to tell without more information?arrow_forward7. Draw the mechanism to describe the following transformation: Note: This is a base catalyzed reaction. So, the last steps must make [OH]- OH [OH]¯ OH Heat Oarrow_forwardShow work with explanation...don't give Ai generated solutionarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,


Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
DIGESTER-35 | VITAMINS AND THEIR RELATED COENZYMES| GPAT | NIPER | PHARMACIST| DI; Author: GPAT DISCUSSION CENTER;https://www.youtube.com/watch?v=CGrdNYmho0s;License: Standard YouTube License, CC-BY