
GENERAL,ORGANIC, & BIOLOGICAL CHEM-ACCES
4th Edition
ISBN: 9781265982959
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 24, Problem 54P
Interpretation Introduction
Interpretation:
The amount of ATP produced by the complete catabolism of (fatty acid) oleic acid (C17H33COOH), needs to be identified.
Concept Introduction:
Beta − oxidation of fatty acids involves four series of reactions. In this reaction, the long-chain fatty acid is degraded into many two-carbon unit's acetyl CoA.
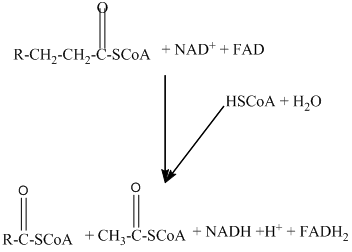
The number of acetyl CoA molecules formed, and the number of time beta-oxidation occur can be determined by using the number of carbon atoms present in the fatty acid.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
The two pKa values of oxalic acid are 1.25 and 3.81. Why are they not the same value? Show the protontransfer as part of your explanation. *see image
асть
Identify all the bonds that gauche interact with C-OMe in
the most stable conformation of the above compound.
Predict the reactants used in the formation of the following compounds using Acid-Catalyzed dehydration reaction
Chapter 24 Solutions
GENERAL,ORGANIC, & BIOLOGICAL CHEM-ACCES
Ch. 24.2 - Analyze the following reaction by considering the...Ch. 24.2 - Prob. 24.2PPCh. 24.3 - Prob. 24.1PCh. 24.3 - Prob. 24.2PCh. 24.3 - Prob. 24.3PCh. 24.3 - Prob. 24.4PCh. 24.3 - Prob. 24.5PCh. 24.3 - Prob. 24.6PCh. 24.4 - Prob. 24.7PCh. 24.4 - Prob. 24.8P
Ch. 24.4 - Prob. 24.9PCh. 24.5 - Prob. 24.10PCh. 24.5 - Prob. 24.11PCh. 24.5 - Prob. 24.12PCh. 24.6 - Prob. 24.13PCh. 24.7 - Prob. 24.14PCh. 24.7 - Prob. 24.3PPCh. 24.7 - Prob. 24.15PCh. 24.7 - Prob. 24.16PCh. 24.7 - Use the number of molecules of ATP formed from the...Ch. 24.7 - Prob. 24.18PCh. 24.8 - Prob. 24.19PCh. 24.8 - Prob. 24.20PCh. 24.8 - Prob. 24.21PCh. 24.9 - Prob. 24.4PPCh. 24.9 - What products are formed when each amino acid is...Ch. 24.9 - Prob. 24.22PCh. 24 - Analyze each reaction by considering the...Ch. 24 - Analyze each reaction by considering the...Ch. 24 - Prob. 25PCh. 24 - Prob. 26PCh. 24 - Prob. 27PCh. 24 - Prob. 28PCh. 24 - Prob. 29PCh. 24 - Prob. 30PCh. 24 - Prob. 31PCh. 24 - Prob. 32PCh. 24 - Glucose is completely metabolized to six molecules...Ch. 24 - Why is glycolysis described as an anaerobic...Ch. 24 - Write the overall equation with key coenzymes for...Ch. 24 - Prob. 36PCh. 24 - Prob. 37PCh. 24 - Prob. 38PCh. 24 - Consider the aerobic and anaerobic avenues of...Ch. 24 - Prob. 40PCh. 24 - Prob. 41PCh. 24 - Prob. 42PCh. 24 - Prob. 43PCh. 24 - Prob. 44PCh. 24 - Prob. 45PCh. 24 - Prob. 46PCh. 24 - Prob. 47PCh. 24 - Prob. 48PCh. 24 - Prob. 49PCh. 24 - Prob. 50PCh. 24 - Prob. 51PCh. 24 - Prob. 52PCh. 24 - Prob. 53PCh. 24 - Prob. 54PCh. 24 - Prob. 55PCh. 24 - Prob. 56PCh. 24 - Prob. 57PCh. 24 - Prob. 58PCh. 24 - Prob. 59PCh. 24 - How much ATP is generated by the complete...Ch. 24 - Prob. 61PCh. 24 - Fill in the boxes with the number of moles of each...Ch. 24 - Prob. 63PCh. 24 - Prob. 64PCh. 24 - Prob. 65PCh. 24 - Prob. 66PCh. 24 - Prob. 67PCh. 24 - Prob. 68PCh. 24 - Prob. 69PCh. 24 - Prob. 70PCh. 24 - What is the difference between ketogenic and...Ch. 24 - Prob. 72PCh. 24 - Prob. 73PCh. 24 - Draw the structure of the keto acid formed by the...Ch. 24 - Draw the products formed in each transamination...Ch. 24 - Prob. 76PCh. 24 - Prob. 77PCh. 24 - Prob. 78PCh. 24 - Prob. 79PCh. 24 - Prob. 80PCh. 24 - What metabolic intermediate is formed from the...Ch. 24 - What metabolic intermediate is formed from the...Ch. 24 - Prob. 83PCh. 24 - Prob. 84PCh. 24 - Prob. 85PCh. 24 - Prob. 86PCh. 24 - Prob. 87PCh. 24 - What is the cause of the pain and cramping in a...Ch. 24 - Prob. 89PCh. 24 - Prob. 90PCh. 24 - Prob. 91PCh. 24 - Prob. 92PCh. 24 - Prob. 93PCh. 24 - Prob. 94PCh. 24 - What type of enzyme would catalyze the conversion...Ch. 24 - Prob. 96PCh. 24 - Prob. 97CPCh. 24 - Prob. 98CPCh. 24 - Prob. 99CPCh. 24 - Prob. 100CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Can I please get help with this?arrow_forward.. Give the major organic product(s) for each of the following reactions or sequences of reactions. Show ll relevant stereochemistry [3 ONLY]. A H Br 1. NaCN 2 NaOH, H₂O, heat 3. H3O+ B. CH₂COOH 19000 1. LiAlH4 THF, heat 2 H₂O* C. CH Br 1. NaCN, acetone 2 H3O+, heat D. Br 1. Mg. ether 3. H₂O+ 2 CO₂ E. CN 1. (CH) CHMgBr, ether 2 H₂O+arrow_forwardAssign this COSY spectrumarrow_forward
- Can I please get help with this?arrow_forward1. Draw structures corresponding to each of the following names [3 ONLY]: A. 2,2,2-trichloroethanal (chloral). B. trans-3-isopropylcyclohexanecarbaldehyde C. What is the correct structure for 2-hydroxyacetophenone? Circle the letter of your response. a C 0 OH OH OH HO b. H3C CH 0 H d OH D. Provide IUPAC names for each structure below. 0 H C-H 0 0 CH3 H NO₂ E. The substance formed on addition of water to an aldehyde or ketone is called a hydrate or a/an: a. vicinal diol b. geminal diol C. acetal d. ketalarrow_forwardAssign this spectrumarrow_forward
- Redraw the tripeptide with or without its acidic hydrogensto demonstrate where the total charge of -2 comes from: *see imagearrow_forward2. Consider the data below to answer the following questions. Cyanohydrins are important intermediates in the synthesis of α-hydroxycarboxylic acids from ketones and aldehydes. The nitrile functional group can be hydrolyzed by aqueous acid to yield a carboxylic acid. Nitriles can also be hydrolyzed to carboxylic acids using aqueous base. Unfortunately, when a cyanohydrin is treated with aqueous base the original carbonyl compound is isolated. OH CH-COOH 0 HO CN C H30* C. H H HC N NaOH H₂O C=O 0 cyanohydrin H + NaCN + H₂Oarrow_forwardAssign all integrated peaksarrow_forward
- - Consider the data in the Table below to answer the following questions: Acidities of Substituted Benzoic and Acetic Acids pk,s at 25C Y-CH COOH Y Y - CH₂COOH meta para H 4.75 4.19 4.19 2.47 3.64 3.55 3.57 4.09 4.46 CN OCH 3 A. Draw the structure of the strongest acid in the table above. B. Explain why cyanoacetic acid and methoxyacetic acid are more acidic than their correspondingly substituted benzoic acid counterparts.arrow_forwardDraw the curved arrow mechanism for this reaction starting with 2-propanol in sulfuric acid. Show all nonzero formal charges and all nonbonded electrons in each step. Species not involved in a particular step do not need to be included in that step, and resonance forms do not need to be shown. Note that the alcohol is in much higher concentration than H₂O in this reaction. Harrow_forwardProvide reactions showing the following conversions: * see imagearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,
