
Organic Chemistry As a Second Language: Second Semester Topics
4th Edition
ISBN: 9781119110651
Author: David R. Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2.3, Problem 2.4P
For each of the following compounds, determine whether or not you would expect its IR spectrum to exhibit a signal to the left of 3000 cm–1
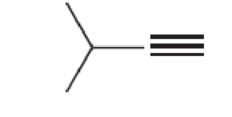
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Compound 1 has molecular formula C6H12. It shows three signals in the 1H-NMR spectrum, one at 0.96 ppm, one at 2.03 ppm, and one at 5.33 ppm. The relative integrals of these three signals are 3, 2, and 1, respectively.
Compound 2 has molecular formula C7H15Br. It shows two signals in the 1H-NMR spectrum, one at 1.08 ppm and one at 1.59 ppm. The relative integrals of these two signals are 3 and 2, respectively.
Propose structures for compounds 1 and 2, explaining how you reach your conclusion.
Compound 1 has molecular formula C7H16. It shows three signals in the 1H-NMR spectrum, one at 0.85 ppm, one at 1.02 ppm, and one at 1.62 ppm. The relative integrals of these three signals are 6, 1, and 1, respectively.
Compound 2 has molecular formula C7H14. It shows three signals in the 1H-NMR spectrum, one at 0.98 ppm, one at 1.36 ppm, and one at 1.55 ppm. The relative integrals of these three signals are 3, 2, and 2, respectively.
Propose structures for compounds 1 and 2, explaining how you reach your conclusion.
1Compound 1 has molecular formula C7H16. It shows three signals in the 1H-NMR spectrum, one at 0.85 ppm, one at 1.02 ppm, and one at 1.62 ppm. The relative integrals of these three signals are 6, 1, and 1, respectively.
Compound 2 has molecular formula C7H14. It shows three signals in the 1H-NMR spectrum, one at 0.98 ppm, one at 1.36 ppm, and one at 1.55 ppm. The relative integrals of these three signals are 3, 2, and 2, respectively.
Propose structures for compounds 1 and 2, explaining how you reach your conclusion.
Chapter 2 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Ch. 2.3 - For each of the following compounds, determine...Ch. 2.3 - For each of the following compounds, determine...Ch. 2.3 - For each of the following compounds, determine...Ch. 2.3 - For each of the following compounds, determine...Ch. 2.3 - The following compound has three carbonyl groups....Ch. 2.4 - Predict which of the following C=C bonds will...Ch. 2.4 - The C=C bond in the following compound produces an...Ch. 2.5 - For each IR spectrum below, identify whether it is...Ch. 2.5 - For each IR spectrum below, identify whether it is...Ch. 2.5 - For each IR spectrum below, identify whether it is...
Ch. 2.5 - For each IR spectrum below, identify whether it is...Ch. 2.5 - For each IR spectrum below, identify whether it is...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.6 - Prob. 2.22P
Additional Science Textbook Solutions
Find more solutions based on key concepts
Determine the de Brogue wavelength of a. an electron moving at 1/10 the speed of light. b. a 400 g Frisbee movi...
Inorganic Chemistry
27. Consider the reaction.
Express the rate of the reaction in terms of the change in concentration of each of...
Chemistry: Structure and Properties
44. Calculate the ratio of CH3NH2 to CH3NH3Cl concentration required to create a buffer with pH = 10.24.
Chemistry: A Molecular Approach
12.1 Give the IUPAC name for each of the following:
a. CH3-CH2-OH
b.
c.
d.
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Complete the spectroscopy data tables for a compound with molecular formula C6H14. Determine the structure of the compound. Any labile protons, if they exist, will not be present in this particular 1H NMR spectrum.arrow_forward6) Complete the spectroscopy data tables for a compound with molecular formula C6H12O. Determine the structure of the compound. Any labile protons, if they exist, will not be present in this particular 1H NMR spectrum.arrow_forwardWhich of the following compounds (methylene blue, methyl orange, methyl red, or carotene) does the spectrum belong to?arrow_forward
- 1) Complete the spectroscopy data tables for a compound with molecular formula C8H8O. Determine the structure of the compound. Any labile protons, if they exist, will not be present in this particular1H NMR spectrum.arrow_forwardWhich compound gives a signal in the 1H-NMR spectrum with a larger chemical shift, furan or cyclopentadiene? Explain.arrow_forwardExplain why these are the correct spectra for the molecules. Identify a specific absorption band which identifies each characteristic functional group of the molecule chosen.arrow_forward
- 3. Draw a molecule with the molecular formula C6H10O that would have the following peak(s) in its IR spectrum. (all major non-fingerprint absorbances are listed) a. 3200-3500 cm¹ (broad) 2900-3000 cm-¹ 2200 cm¹ (weak)arrow_forwardQ1: Consider the two spectra shown below. One is for 3-methyl-1-cyclopentanone and the other one is for 3-methyl-1-cyclohexanone. Identify which spectra belongs to one compound explain your reasoning. Identify the important absorption peaks correspond to a functional group. Describe the absorption peaks. am 100 4900 400 3000 d 1500 1300 REG ho DEFE 18 PES SAFE RECERES 100arrow_forwardFrom the 13C spectrum of a compound, one can assign signals to specific carbon atoms of the compound and determine the number of protons on each of those carbon atoms. Such information in conjunction with the DEPT C-13 chart will allow the appearance of the corresponding DEPT spectrum of the compound to be predicted. There are five signals (I – V) in the 13C spectrum of compound A as shown below. At the exactly same location of each of these five signals, "a peak", "no peak", or "an inverted peak" may appear in the corresponding DEPT-135 spectrum. Predict the DEPT-135 spectrum of compound A by selecting the expected signal appearance at each of these five locations in the DEPT-135 spectrum. - II II IV V in HO A 100 PPM 180 160 140 120 80 60 40 20 v At the location of signal I A. no peak v At the location of signal IIl В. а рeak v At the location of signal IIl C. an inverted peak v At the location of signal IV v A the location of signal Varrow_forward
- 4000 For the following compound identify what peaks would be found in an IR spectrum. Draw the expected IR spectrum on the graph below. Label each peak with the location and portion of the molecule it represents. 3500 3000 IR spectrum 2500 2000 1500 1000 500 0arrow_forwardWithin what region of the electromagnetic spectrum do you expect this compound to absorb? CIRCLE one. < 400 nm 400 – 500 nm above 500 nm but lower than 700 nmarrow_forward15) Which one of the compounds corresponds to this IR spectrum. NH2 1000 1503 2000 3000arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning


EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY