
Organic Chemistry As a Second Language: Second Semester Topics
4th Edition
ISBN: 9781119110651
Author: David R. Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2.5, Problem 2.16P
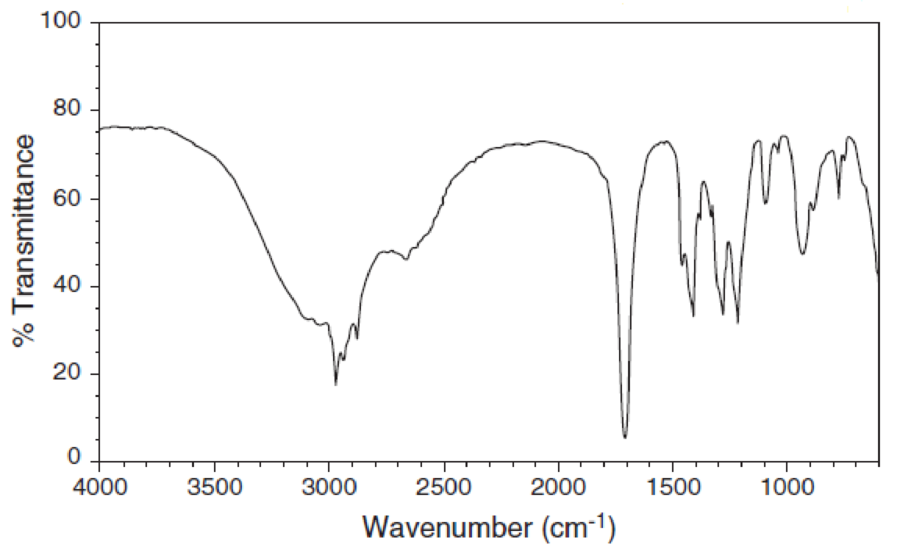
For each IR spectrum below, determine whether it is consistent with the structure of a
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
6. Draw the molecular orbital diagram shown to determine which of the following is paramagnetic.
B₂2+
B22+, B2, C22, B22- and N22+
Molecular Orbital Diagram
B2
C22-
B22-
N22+
Which molecule is paramagnetic?
Don't used hand raiting
EXERCISES: Complete the following exercises. You must show all work to receive full credit.
1. How many molecular orbitals can be built from the valence shell orbitals in O2?
2. Give the ground state electron configuration (e.g., 02s² 0*2s² П 2p²) for these molecules and deduce
its bond order.
Ground State Configuration
Bond Order
H2+
02
N2
Chapter 2 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Ch. 2.3 - For each of the following compounds, determine...Ch. 2.3 - For each of the following compounds, determine...Ch. 2.3 - For each of the following compounds, determine...Ch. 2.3 - For each of the following compounds, determine...Ch. 2.3 - The following compound has three carbonyl groups....Ch. 2.4 - Predict which of the following C=C bonds will...Ch. 2.4 - The C=C bond in the following compound produces an...Ch. 2.5 - For each IR spectrum below, identify whether it is...Ch. 2.5 - For each IR spectrum below, identify whether it is...Ch. 2.5 - For each IR spectrum below, identify whether it is...
Ch. 2.5 - For each IR spectrum below, identify whether it is...Ch. 2.5 - For each IR spectrum below, identify whether it is...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.6 - Prob. 2.22P
Additional Science Textbook Solutions
Find more solutions based on key concepts
Distinguish between microevolution, speciation, and macroevolution.
Campbell Essential Biology (7th Edition)
The electron configuration of aluminum should be written followed by noble gas shorthand configuration. Concept...
Living By Chemistry: First Edition Textbook
41. A reaction in which A, B, and C react to form products is first order in A, second order in B, and zero ord...
Chemistry: Structure and Properties (2nd Edition)
8.63 Two flasks of equal volume and at the same temperature contain different gases. One flask contains 10.0 g ...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Flask A contains yeast cells in glucose-minimal salts broth incubated at 30C with aeration. Flask B contains ye...
Microbiology: An Introduction
51. I A tennis player hits a ball 2.0 m above the ground. The ball leaves his racquet with a speed of 20.0 m/s ...
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 7. Draw the Lewis structures and molecular orbital diagrams for CO and NO. What are their bond orders? Are the molecular orbital diagrams similar to their Lewis structures? Explain. CO Lewis Structure NO Lewis Structure CO Bond Order NO Bond Order CO Molecular Orbital Diagram NO Molecular Orbital Diagramarrow_forwardDon't used hand raiting and don't used Ai solutionarrow_forwardDraw the condensed structure of 4-ethyl-1,2,4-trifluoro-2-methyloctane.arrow_forward
- 5. The existence of compounds of the noble gases was once a great surprise and stimulated a great deal of theoretical work. Label the molecular orbital diagram for XeF (include atom chemical symbol, atomic orbitals, and molecular orbitals) and deduce its ground state electron configuration. Is XeF likely to have a shorter bond length than XeF+? XeF XeF+ Bond Orderarrow_forwardDon't used hand raitingarrow_forward4. The superoxide ion, Oz, plays an important role in the ageing processes that take place in organisms. Judge whether O2 is likely to have larger or smaller dissociation energy than O2. Molecular Orbital Diagram 8 02 02 Does O2 have larger or smaller dissociation energy?: Bond Orderarrow_forward
- Will a weak base with a pKa of 8.4 be best absorbed in the stomach or the intestine? Explain your reasoning behind your answer. Use a chemical equilibrium equation in your answer.arrow_forwardYou have started a patient on a new drug. Each dose introduces 40 pg/mL of drug after redistribution and prior to elimination. This drug is administered at 24 h intervals and has a half life of 24 h. What will the concentration of drug be after each of the first six doses? Show your work a. What is the concentration after the first dose? in pg/mL b. What is the concentration after the second dose? in pg/mL c. What is the concentration after the third dose? in pg/mLarrow_forwardHow many different molecules are drawn below?arrow_forward
- Only 100% sure experts solve it correct complete solutions need to get full marks it's my quiz okkkk.take your time but solve full accurate okkk chemistry expert solve it.qno4arrow_forwardNonearrow_forwardA complete tensile test was performed on a magnesium specimen of 12 mm diameter and 30 mm length, until breaking. The specimen is assumed to maintain a constant volume. Calculate the approximate value of the actual stress at breaking. TABLE. The tensile force F and the length of the specimen are represented for each L until breaking. F/N L/mm 0 30,0000 30,0296 5000 10000 30,0592 15000 30,0888 20000 30,15 25000 30,51 26500 30,90 27000 31,50 26500 32,10 25000 32,79arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Mass Spectrometry; Author: Professor Dave Explains;https://www.youtube.com/watch?v=hSirWciIvSg;License: Standard YouTube License, CC-BY