
(a)
Interpretation:
The most likely site(s) of electrophilic
Concept introduction:
Electrophilic aromatic substitution reaction rate increases with increase in electron density around the ring. The electron donating groups activates whereas the electron withdrawing groups deactivates the aromatic ring. The electron donating groups increases the electron density at ortho-para position and thus they are ortho-para directing. The electron withdrawing groups decrease electron density at ortho/para and has comparatively greater electron density at meta position, thus they are meta directing.
Answer to Problem 23.53P
The most likely site(s) electrophilic aromatic substitution in the given molecule is:
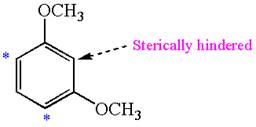
Explanation of Solution
The given molecule is:
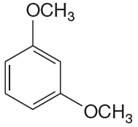
In the given molecule, benzene has two methoxy substituents. These are activating groups, thus ortho-para directors. Each methoxy group activates their two ortho and one para positions. The activated sites are same for both groups, thus overall there are three activated sites. But the position between two methoxy groups is sterically hindered. Thus, the molecules have only two possible sites of electrophilic aromatic substitution which are indicated by an asterisk
The most likely site(s) of electrophilic aromatic substitution are determined by identifying positions activated by the substituents attached.
(b)
Interpretation:
The most likely site(s) of electrophilic aromatic substitution in the given molecule are to be indicated.
Concept introduction:
Electrophilic aromatic substitution reaction rate increases with increase in electron density around the ring. The electron donating groups activate whereas the electron withdrawing groups deactivate the aromatic ring. The electron donating groups increase the electron density at ortho-para position and thus they are ortho-para directing. The electron withdrawing groups decreases electron density at ortho/para and has comparatively greater electron density at meta position, thus they are meta directing.
Answer to Problem 23.53P
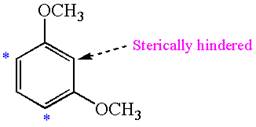
The most likely site(s) of electrophilic aromatic substitution in the given molecule are:
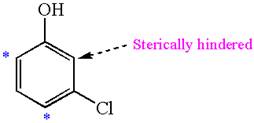
Explanation of Solution
The given molecule is:
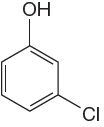
In the given molecule, benzene has one hydroxyl and one chloride substituent. Both these are activating groups, thus ortho-para directors. Each of the susbtituents activates their two ortho and one para positions. The activated sites are same for both groups thus overall there are three activated sites. But the position between these two substituents is sterically hindered. Thus, molecules have only two possible sites of electrophilic aromatic substitution which are indicated by an asterisk
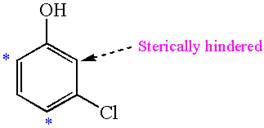
The most likely site(s) of electrophilic aromatic substitution are determined by identifying positions activated by the substituents attached.
(c)
Interpretation:
The most likely site(s) of electrophilic aromatic substitution in the given molecule are to be indicated.
Concept introduction:
The electrophilic aromatic substitution reaction rate increases with increase in electron density around the ring. The electron donating groups activates whereas the electron withdrawing groups deactivates the aromatic ring. The electron donating groups increase the electron density at ortho-para position and thus they are ortho-para directing. The electron withdrawing groups decrease electron density at ortho/para and have comparatively greater electron density at meta position, thus they are meta directing.
Answer to Problem 23.53P
The most likely site(s) of electrophilic aromatic substitution in the given molecule are:
Explanation of Solution
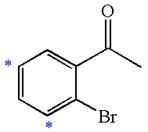
The given molecule is:
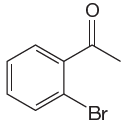
In the given molecule, benzene has one acetyl and one bromide substituent. Acetyl is carbonyl group which is deactivating thus it is meta director. The bromine atom is activating group thus it is ortho/para director. The acetyl group activates its two meta positions and bromine activates its one ortho and one para positions. The meta positions of acetyl group are same as the ortho/para position of bromine atom. Thus the molecules have only two possible sites of electrophilic aromatic substitution which are indicated by an asterisk
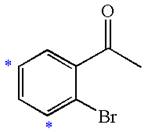
The most likely site(s) of electrophilic aromatic substitution are determined by identifying positions activated by the substituents attached.
(d)
Interpretation:
The most likely site(s) of electrophilic aromatic substitution in the given molecule are to be indicated.
Concept introduction:
The electrophilic aromatic substitution reaction rate increases with increase in electron density around the ring. The electron donating groups activates whereas the electron withdrawing groups deactivates the aromatic ring. The electron donating groups increases the electron density at ortho-para position and thus they are ortho-para directing. The electron withdrawing groups decreases electron density at ortho/para and has comparatively greater electron density at meta position, thus they are meta directing.
Answer to Problem 23.53P
The most likely site(s) of electrophilic aromatic substitution in the given molecule are:
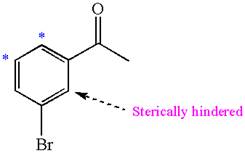
Explanation of Solution
The given molecule is:
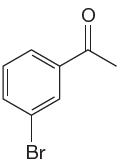
In the given molecule, benzene has one acetyl and one bromide substituent. Acetyl is carbonyl group which is deactivating thus it is meta director. The bromine atom is activating group thus it is ortho/para director.
The acetyl group activates its one meta positions and bromine activates two ortho and one para positions. The activating sites of both groups are different, thus overall there are three activated sites. But the position between these two substituents is sterically hindered. Thus, molecules have only two possible sites of electrophilic aromatic substitution which are indicated by an asterisk

The most likely site(s) of electrophilic aromatic substitution are determined by identifying positions activated by the substituents attached.
(e)
Interpretation:
The most likely site(s) of electrophilic aromatic substitution in the given molecule are to be indicated.
Concept introduction:
The electrophilic aromatic substitution reaction rate increases with increase in electron density around the ring. The electron donating groups activates whereas the electron withdrawing groups deactivates the aromatic ring. The electron donating groups increases the electron density at ortho-para position and thus they are ortho-para directing. The electron withdrawing groups decreases electron density at ortho/para and has comparatively greater electron density at meta position, thus they are meta directing.
Answer to Problem 23.53P
The most likely site(s) of electrophilic aromatic substitution in the given molecule are:
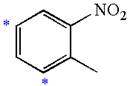
Explanation of Solution
The given molecule is:
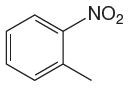
In the given molecule, benzene has one nitro and one methyl substituent. Nitro group is deactivating thus it is meta director. The methyl group is activating group thus it is ortho/para director. The nitro group activates its two meta positions and methyl group activates its one ortho and one para positions. The meta positions of nitro group are same as the ortho/para positions of methyl groups. Thus, the molecules have only two possible sites of electrophilic aromatic substitution which are indicated by an asterisk

The most likely site(s) of electrophilic aromatic substitution are determined by identifying positions activated by the substituents attached.
(f)
Interpretation:
The most likely site(s) of electrophilic aromatic substitution in the given molecule are to be indicated.
Concept introduction:
The electrophilic aromatic substitution reaction rate increases with increase in electron density around the ring. The electron donating groups activates whereas the electron withdrawing groups deactivates the aromatic ring. The electron donating groups increases the electron density at ortho-para position and thus they are ortho-para directing. The electron withdrawing groups decreases electron density at ortho/para and has comparatively greater electron density at meta position, thus they are meta directing.
Answer to Problem 23.53P
The most likely site(s) of electrophilic aromatic substitution in the given molecule are:
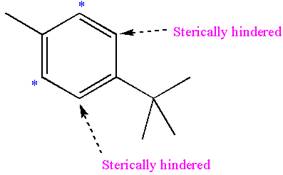
Explanation of Solution
The given molecule is:
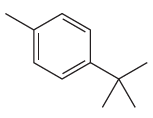
In the given molecule, benzene has one methyl and one

The most likely site(s) of electrophilic aromatic substitution are determined by identifying positions activated by the substituents attached.
(g)
Interpretation:
The most likely site(s) of electrophilic aromatic substitution in the given molecule are to be indicated.
Concept introduction:
The electrophilic aromatic substitution reaction rate increases with increase in electron density around the ring. The electron donating groups activates whereas the electron withdrawing groups deactivates the aromatic ring. The electron donating groups increases the electron density at ortho-para position and thus they are ortho-para directing. The electron withdrawing groups decreases electron density at ortho/para and has comparatively greater electron density at meta position, thus they are meta directing.
Answer to Problem 23.53P
The most likely site(s) of electrophilic aromatic substitution in the given molecule are:
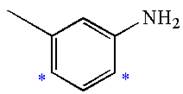
Explanation of Solution
The given molecule is:
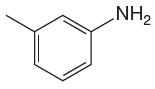
In the given molecule, benzene has one methyl and one amino substituent. They both are activating groups, thus they are ortho/para positions. Each of the susbtituents activates their two ortho and one para positions. The some activated sites are not same for both groups so, overall there are three activated sites. But the position between two groups is sterically hindered. Thus, molecules have only two possible sites of electrophilic aromatic substitution which are indicated by an asterisk

The most likely site(s) of electrophilic aromatic substitution are determined by identifying positions activated by the substituents attached.
Want to see more full solutions like this?
Chapter 23 Solutions
Organic Chemistry: Principles And Mechanisms
- Draw the product of the reaction shown below. Ignore small byproducts that would evaporate pleasearrow_forwardPoly(ethylene adipate) is a biodegradable polyester (shown below). Identify the type of polymerization process used in the production of this polymer.arrow_forwardPolymers may be composed of thousands of monomers. draw two repeat units(dimer) of the polymer formed in this reaction. assume there are hydrogen atoms on the two ends of the dimer. ignore inorganic byproducts pleasearrow_forward
- Draw the product of the reaction shown below. Use a dash or wedge bond to indicate stereochemistry of substituents on asymmetric centers, Ignore inorganic byproductsarrow_forwardDraw the product of this reaction please. Ignore inorganic byproductsarrow_forwardOne of the pi molecular orbitals of 1,3-butadiene (CH2=CHCH=CH2) is shown below. Please identify the number of nodal planes perpendicular to the bonding axisarrow_forward
- Draw the monomers required to synthesize this condensation polymer please.arrow_forwardProvide the correct systematic name for the compound shown here. Please take into account the keyboard options belowarrow_forwardcurved arrows are used to illustrate the flow of electrons. using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s)arrow_forward
- Identify the 'cartoon' drawing of the acceptor orbital in the first mechanistic step of an electrophilic addition reaction of butadiene with HBr. Pleasearrow_forwardH- H H H H H H Identify and select all structures below that represent a constitutional isomer(s) of the compound shown above. H- H H H A. H H H H-C CI H H D. H H H H H H C C -H H C C H H H H B. H CI H H- C C H H H H E. H CI H C.arrow_forwardWhy doesn't this carry on to form a ring by deprotonating the alpha carbon and the negatively-charged carbon attacking the C=O?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





