
Owlv2 With Ebook, 1 Term (6 Months) Printed Access Card For Kotz/treichel/townsend/treichel's Chemistry & Chemical Reactivity, 10th
10th Edition
ISBN: 9781337791182
Author: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 23, Problem 103IL
Interpretation Introduction
Interpretation:
The compound has to be identified when it gives a acetic acid on reacting with
Concept introduction:
Oxidation reaction:
Alcohol undergoes oxidation reaction using oxidising agent like
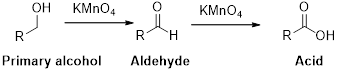
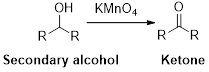
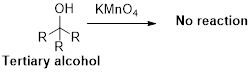
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Identify the glycosidic linkage.
Draw the mechanism for the substitution reaction converting an alcohol into an alkyl halide. If chirality is important to the reaction include it.
Write, in words three different reactions we can use to make an alcohol.
Chapter 23 Solutions
Owlv2 With Ebook, 1 Term (6 Months) Printed Access Card For Kotz/treichel/townsend/treichel's Chemistry & Chemical Reactivity, 10th
Ch. 23.2 - (a) Draw the nine isomers having the formula...Ch. 23.2 - Prob. 23.2CYUCh. 23.2 - There are 17 possible alkene isomers with the...Ch. 23.2 - Prob. 23.4CYUCh. 23.2 - Aniline, C6H5NH2, is the common name for...Ch. 23.3 - Draw the structure of 1-butanol and alcohols that...Ch. 23.4 - (a) Name each of the following compounds and its...Ch. 23.5 - Kevlar is a well-known polymer that is now used to...Ch. 23.5 - Prob. 1.1ACPCh. 23.5 - Prob. 1.2ACP
Ch. 23.5 - Prob. 1.3ACPCh. 23.5 - Prob. 2.1ACPCh. 23.5 - Prob. 2.2ACPCh. 23.5 - Prob. 2.3ACPCh. 23.5 - What is the atom economy for the reaction of...Ch. 23.5 - Prob. 3.2ACPCh. 23.5 - If drinking from a polycarbonate bottle, does a 15...Ch. 23.5 - Assume you weigh 156 lb. How much BPA do you...Ch. 23.5 - Prob. 3.5ACPCh. 23 - Prob. 1PSCh. 23 - Prob. 2PSCh. 23 - Is violet light (with a wavelength of 400 nm)...Ch. 23 - Prob. 4PSCh. 23 - Prob. 5PSCh. 23 - Prob. 6PSCh. 23 - Prob. 7PSCh. 23 - Prob. 8PSCh. 23 - Prob. 9PSCh. 23 - What is the molecular formula for an alkane with...Ch. 23 - Prob. 11PSCh. 23 - Prob. 12PSCh. 23 - One of the structural isomers with the formula...Ch. 23 - Prob. 14PSCh. 23 - Prob. 15PSCh. 23 - Give the systematic name for the following alkane....Ch. 23 - Draw the structure of each of the following...Ch. 23 - Draw structures for the following compounds. (a)...Ch. 23 - Prob. 19PSCh. 23 - Prob. 20PSCh. 23 - Draw the structure of the chair form of...Ch. 23 - Prob. 22PSCh. 23 - Prob. 23PSCh. 23 - Prob. 24PSCh. 23 - Prob. 25PSCh. 23 - Prob. 26PSCh. 23 - Prob. 27PSCh. 23 - What structural requirement is necessary for an...Ch. 23 - A hydrocarbon with the formula C5H10, can be...Ch. 23 - Prob. 30PSCh. 23 - Prob. 31PSCh. 23 - Prob. 32PSCh. 23 - The compound 2-bromobutane is a product of...Ch. 23 - The compound 2,3-dibromo-2-methylhexane is formed...Ch. 23 - Prob. 35PSCh. 23 - Prob. 36PSCh. 23 - Prob. 37PSCh. 23 - Prob. 38PSCh. 23 - Prob. 39PSCh. 23 - Give the systematic name for each of the following...Ch. 23 - Prob. 41PSCh. 23 - Write an equation for the preparation of...Ch. 23 - Prob. 43PSCh. 23 - Prob. 44PSCh. 23 - Prob. 45PSCh. 23 - Prob. 46PSCh. 23 - Prob. 47PSCh. 23 - Name the following amines: (a) CH3CH2CH2NH2 (b)...Ch. 23 - Draw structural formulas for the four possible...Ch. 23 - Prob. 50PSCh. 23 - Prob. 51PSCh. 23 - Prob. 52PSCh. 23 - Prob. 53PSCh. 23 - Prob. 54PSCh. 23 - Prob. 55PSCh. 23 - Prob. 56PSCh. 23 - Prob. 57PSCh. 23 - Prob. 58PSCh. 23 - Give the structural formula and systematic name...Ch. 23 - Prob. 60PSCh. 23 - Prob. 61PSCh. 23 - Prob. 62PSCh. 23 - Prob. 63PSCh. 23 - Prob. 64PSCh. 23 - Prob. 65PSCh. 23 - Prob. 66PSCh. 23 - Prob. 67PSCh. 23 - Prob. 68PSCh. 23 - Identify the functional groups in the following...Ch. 23 - Prob. 70PSCh. 23 - Prob. 71PSCh. 23 - Prob. 72PSCh. 23 - Prob. 73PSCh. 23 - Prob. 74PSCh. 23 - Prob. 75GQCh. 23 - Prob. 76GQCh. 23 - Prob. 77GQCh. 23 - Prob. 78GQCh. 23 - Prob. 79GQCh. 23 - Prob. 80GQCh. 23 - Prob. 81GQCh. 23 - Write equations for the following reactions,...Ch. 23 - Prob. 83GQCh. 23 - Prob. 84GQCh. 23 - Draw the structure of each of the following...Ch. 23 - Prob. 86GQCh. 23 - Prob. 87GQCh. 23 - Draw structural formulas for possible isomers with...Ch. 23 - Prob. 89GQCh. 23 - Prob. 90GQCh. 23 - Prob. 91GQCh. 23 - Prob. 92GQCh. 23 - Prob. 93GQCh. 23 - Prob. 94GQCh. 23 - Draw the structure of glyceryl trilaurate, a fat....Ch. 23 - Prob. 96GQCh. 23 - Prob. 97GQCh. 23 - Prob. 98GQCh. 23 - Prob. 99GQCh. 23 - There are three ethers with the formula C4H10O....Ch. 23 - Review the opening photograph about chocolate...Ch. 23 - Prob. 102GQCh. 23 - Prob. 103ILCh. 23 - Prob. 104ILCh. 23 - Prob. 105ILCh. 23 - Prob. 106ILCh. 23 - Prob. 107ILCh. 23 - Prob. 108ILCh. 23 - Prob. 109ILCh. 23 - Prob. 110ILCh. 23 - Prob. 111ILCh. 23 - Prob. 112ILCh. 23 - Prob. 113ILCh. 23 - Prob. 114ILCh. 23 - Prob. 115SCQCh. 23 - Prob. 116SCQCh. 23 - Prob. 117SCQCh. 23 - Prob. 118SCQCh. 23 - Prob. 119SCQCh. 23 - Prob. 120SCQCh. 23 - Prob. 121SCQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Name the following carbohydrates give both the systematic and common names. Don't forget to identify the Isomer.arrow_forwardWhat is the product of the reaction of XeF4 with H2O? Group of answer choices H2XeF2 H2XeF4 XeO3 H2XeOarrow_forwardWhile noble gas exerts the strongest London (dispersion) forces on neighboring atoms? Group of answer choices Xe Ar Kr Nearrow_forward
- Which of the following elements is corrosive to your skin due to that element breaking down C=C bonds? Group of answer choices fluorine iodine bromine chlorinearrow_forwardWhat the best source of sulfide to use on a small scale in the lab? Group of answer choices thiourea H2S NaHS Na2Sarrow_forwardWhich of the following statements about sulfur is FALSE? Group of answer choices H2S is the product of an oxygen-depleted ecosystem. In the acid mine drainage reaction, FeS2 is a product. One allotrope of sulfur has the formula S20. In the environment, bacterial oxidation can convert S2− to elemental S or SO42−.arrow_forward
- Of the following choices, which is the best reason that most materials DON'T spontaneously combust even though our atmosphere is about 21% oxygen? Group of answer choices The reduction of O2 in the gas phase (O2 + e− → O2−) is spontaneous. The reduction of O2 in acid solution (O2 + H+ + e− → HO2(aq)) is spontaneous. O2 is not a reactant in combustion. The O2 bond dissociation energy is 494 kJ/mol, leading to a high activation energy for combustion.arrow_forwardplease answer in the scope of the SCH4U course, I am having a hard time understanding, may you show all steps please and thank you! can you also put the final answers in the table so its understandablearrow_forwardPlan the synthesis of the following compound using the starting material provided and any other reagents needed as long as carbon based reagents have 3 carbons or less. Either the retrosynthesis or the forward synthesis (mechanisms are not required but will be graded if provided) will be accepted if all necessary reagents and intermediates are shown (solvents and temperature requirements are not needed unless specifically involved in the reaction, i.e. DMSO in the Swem oxidation or heat in the KMnO4 oxidation). There may be more than one correct answer, and chemically correct steps will be accepted. Extra points will be given if correct names are provided. The points earned here will be applied to your lowest exam score! H Harrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning