
a)
Interpretation:
The steps involved in preparing the compound represented by the model, using either malonic ester synthesis or an acetoacetic ester synthesis, are to be given.
Concept introduction:
Acetoacetic ester synthesis converts an
Both reactions involve the same steps such as i) enolate ion formation ii) SN2 attack of the enolate anion on the alkyl halide iii) hydrolysis and decarboxylation.
Answer to Problem 17VC
The steps involved in preparing the compound represented by the model using acetoacetic ester synthesis are given below.
Explanation of Solution
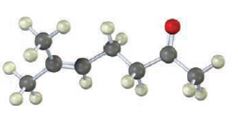
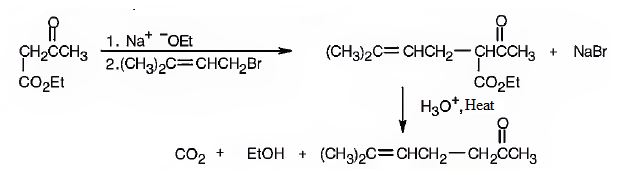
The compound represented by the model is 6-methylhept-5-ene-2-one. It is a methyl ketone and hence it can be prepared using acetoacetic ester synthesis. The ethoxide ion abstracts a proton from the active methylene group of the ester to form the enolate ion. The enolate ion then attacks 1-bromo-3-methy-2-butene and displaces the bromine as bromide ion. The product obtained upon hydrolysis with dilute acids and decarboxylation by heating yields the product.
The steps involved in preparing the compound represented by the model using acetoacetic ester synthesis are given below.

b)
Interpretation:
The steps involved in preparing the compound represented by the model using either malonic ester synthesis or an acetoacetic ester synthesis are to be given.
Concept introduction:
Acetoacetic ester synthesis converts an alkyl halide in to a methyl ketone having three more carbons. The methyl ketone part comes from acetoacetic eater while the remaining carbon comes from the primary alkyl halide. Malonic ester synthesis converts an alkyl halide to a carboxylic acid having two more carbon atoms.
Both reactions involve the same steps such as i) enolate ion formation ii) SN2 attack of the enolate anion on the alkyl halide iii) hydrolysis and decarboxylation.
Answer to Problem 17VC
The steps involved in preparing the compound represented by the model using malonic ester synthesis are given below.
Explanation of Solution
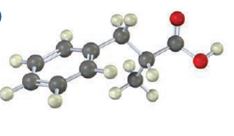
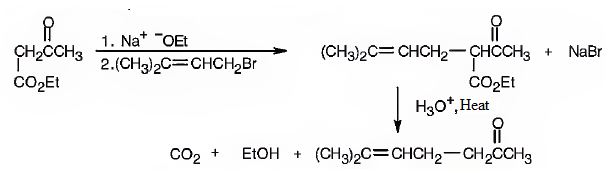
The compound represented by the model is 2-methyl-3-phenylpropanoic acid and hence it can be prepared using malonic ester synthesis. The ethoxide ion abstracts a proton from the active methylene group to form the enolate ion. The enolate ion then attacks benzyl bromide and displaces the bromine as bromide ion. The abstraction of another acidic hydrogen in the product by the base and the nucleophilic displacement of bromine from methyl bromide by enolate ion introduces a methyl group at α- position of the diester. The alkylated diester obtained upon hydrolysis with aqueous acids and decarboxylation by heating yields the product.
The steps involved in preparing the compound represented by the model using malonic ester synthesis are given below.

Want to see more full solutions like this?
Chapter 22 Solutions
Bundle: Organic Chemistry, 9th, Loose-Leaf + OWLv2, 4 terms (24 months) Printed Access Card
- An organic chemistry Teaching Assistant (TA) suggested in your last discussion section that there is only one major organic product of the following reaction and that this reaction builds a ring. If the TA is right, draw the product in the drawing area below. If the TA is wrong, just check the box below the drawing area. 1. NaOMe CH3O N. OCH3 ? 2. H3O+arrow_forwardComplete the reaction in the drawing area below by adding the major products to the right-hand side. If there won't be any products, because nothing will happen under these reaction conditions, check the box under the drawing area instead. Note: if the products contain one or more pairs of enantiomers, don't worry about drawing each enantiomer with dash and wedge bonds. Just draw one molecule to represent each pair of enantiomers, using line bonds at the chiral center. + More... ☐ ☐ : ☐ + G 1. NaOMe Click and drag to start drawing a structure. 2. H +arrow_forward6. Ammonia reacts with nitrogen monoxide and oxygen to form nitrogen and water vapor. If the rate of consumption of NO is 4.5 mollitermin) (a) Find the rate of reaction (b) Find the rate of formations of N; and HO (c) Find the rate of consumption of NH, and O 4NH: 4NO 0:4: +60arrow_forward
- 34. Give the expected major product of each of the following reactions. Conc. HI a. CH3CH2CH2OH b. (CH3)2CHCH2CH2OH Conc. HBr H Conc. HI C. OH Conc.HCI d. (CH3CH2)3COHarrow_forward42. Which of the following halogenated compounds can be used successfully to prepare a Grignard reagent for alcohol synthesis by subsequent reaction with an aldehyde or ketone? Which ones cannot and why? H3C CH3 a. Br H OH b. Cl C. I H H d. Cl e. H OCH3 Br Harrow_forwardFor each reaction below, decide if the first stable organic product that forms in solution will create a new CC bond, and check the appropriate box. Next, for each reaction to which you answered "Yes" to in the table, draw this product in the drawing area below. Note for advanced students: for this problem, don't worry if you think this product will continue to react under the current conditions - just focus on the first stable product you expect to form in solution. ? Will the first MgBr product that forms in this reaction create a new CC bond? olo ? OH جمله O Yes Ⓒ No MgCl ? Will the first product that forms in this reaction create a new CC bond? Click and drag to start drawing a structure. Yes No X ☐ : ☐ टे PHarrow_forward
- Assign all the carbonsarrow_forward9 7 8 C 9 8 200 190 B 5 A -197.72 9 8 7 15 4 3 0: ང་ 200 190 180 147.52 134.98 170 160 150 140 130 120 110 100 90 90 OH 10 4 3 1 2 -143.04 140. 180 170 160 150 140 130 120 110 100 90 CI 3 5 1 2 141.89 140.07 200 190 180 170 160 150 140 130 120 110 100 ៖- 90 129. 126.25 80 70 60 -60 50 40 10 125.19 -129.21 80 70 3.0 20 20 -8 60 50 10 ppm -20 40 128.31 80 80 70 60 50 40 40 -70.27 3.0 20 10 ppm 00˚0-- 77.17 30 20 20 -45.36 10 ppm -0.00 26.48 22.32 ―30.10 ―-0.00arrow_forwardAssign all the carbonsarrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

