
Concept explainers
(a)
Interpretation:
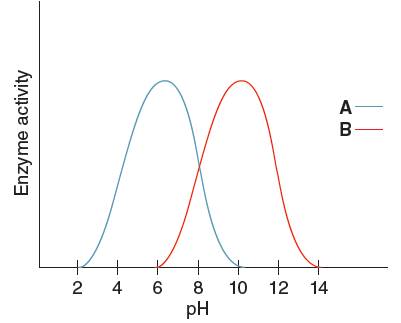
The optimum pH for enzyme 'A' needs to be determined, using the given pH versus enzyme activity graph for enzyme A and B.
Concept Introduction:
Enzymes are defined as a biological catalyst for various reactions in the living organisms. Enzymes are like all catalyst as it also increases the rate reaction but they themselves do not changed permanently during the process. Enzyme does not alter the position of equilibrium and the relative energy of both the reactant and the products. Reaction when catalyzed by an enzyme increases the
Answer to Problem 77P
Optimum pH for enzyme 'A' is
Explanation of Solution
The activity of enzyme is affected by both the temperature and pH. By increasing temperature, rate of reaction also increases until its maximum activity occurs that is at
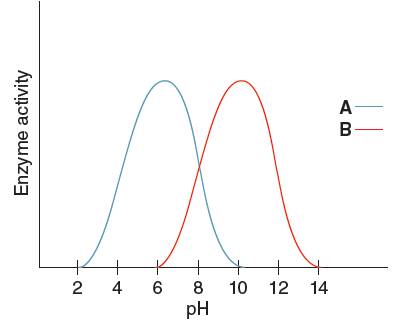
Optimum pH is the pH at which the activity of an enzyme is maximum. So, from the given graph, the activity of enzyme A is maximum at pH 6. Hence the optimum pH of enzyme A is 6.
(b)
Interpretation:
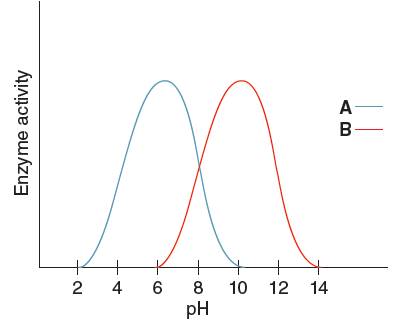
The optimum pH for enzyme 'B' needs to be determined, using the given pH versus enzyme activity graph for enzyme A and B.
Concept Introduction:
Enzymes are defined as a biological catalyst for various reactions in the living organisms. Enzymes are like all catalyst as it also increases the rate reaction but they themselves do not changed permanently during the process. Enzyme does not alter the position of equilibrium and the relative energy of both the reactant and the products. Reaction when catalyzed by an enzyme increases the rate of reaction by
Answer to Problem 77P
Optimum pH for enzyme 'B' is
Explanation of Solution
The activity of enzyme is affected by both the temperature and pH. By increasing temperature, rate of reaction also increases until its maximum activity occurs that is at
Optimum pH is the pH at which the activity of an enzyme is maximum. So, from the given graph, the activity of enzyme B is maximum at pH 10.
Hence, the optimum pH of enzyme B is 10.
(c)
Interpretation:
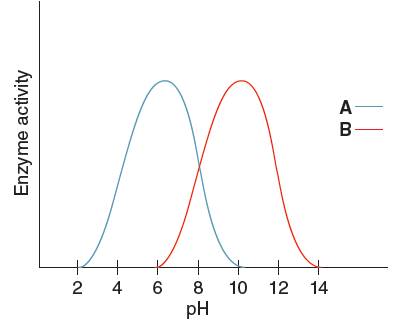
The enzyme having the higher activity when pH is
Concept Introduction:
Enzymes are defined as a biological catalyst for various reactions in the living organisms. Enzymes are like all catalyst as it also increases the rate reaction but they themselves do not changed permanently during the process. Enzyme does not alter the position of equilibrium and the relative energy of both the reactant and the products. Reaction when catalyzed by an enzyme increases the rate of reaction by
Answer to Problem 77P
Enzyme 'A' will have higher pH at
Explanation of Solution
The activity of enzyme is affected by both the temperature and pH. Optimum pH is the pH value at which the enzyme is most active. The rate of enzyme catalyzed reaction is maximum at optimum pH
The graph shown below clearly explains the activity of an enzyme with change in pH values:
From the given graph, it can be seen that the activity of enzyme B starts from pH 6 onwards and activity of enzyme A starts from pH 2 onwards. So, before pH 2 enzyme A is inactive and before pH 6 enzyme B is inactive. Therefore, at pH 4 the activity of enzyme A is higher than that of enzyme B.
(d)
Interpretation:
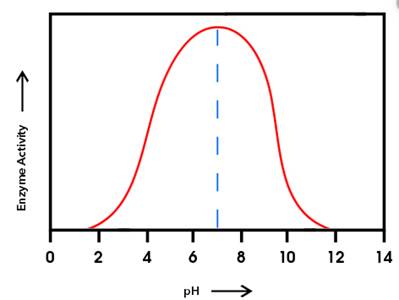
The enzyme having the higher activity at pH
Concept Introduction:
Enzymes are defined as a biological catalyst for various reactions in the living organisms. Enzymes are like all catalyst as it also increases the rate reaction but they themselves do not changed permanently during the process. Enzyme does not alter the position of equilibrium and the relative energy of both the reactant and the products. Reaction when catalyzed by an enzyme increases the rate of reaction by
Answer to Problem 77P
Enzyme 'B' will have the higher activity at pH
Explanation of Solution
The activity of enzyme is affected by both the temperature and pH. Optimum pH is the pH value at which the enzyme is most active. The rate of enzyme catalyzed reaction is maximum at optimum pH
The graph shown below clearly explains the activity of an enzyme with change in pH values:
From the given graph, it can be seen that the activity of enzyme B starts from pH 6 onwards and becomes maximum at pH 10and then activity of enzyme starts decreasing. And activity of enzyme A starts from pH 2 onwards and becomes maximum at pH 6 and then starts decreasing.
At pH 9, activity of enzyme A is decreasing and that of enzyme B is increasing. Hence, at pH 9 activity of enzyme B is higher than that of enzyme A.
Want to see more full solutions like this?
Chapter 21 Solutions
Connect One Semester Access Card for General, Organic, & Biological Chemistry
- Draw the Zaitsev product of the dehydration of this alcohol. + I X 5 OH ざ~ TSOH Click and drag to start drawing a structure.arrow_forwardPlease help with identifying these.arrow_forwardFor the reaction: CO2(g) + H2(g) --> CO (g) + H2O (g) Kc= 0.64 at 900 degrees celcius. if initially you start with 1.00 atmoshpere of carbon dioxide and 1 atmoshpere of hydrogen gas, what are the equilibrium partial pressuses of all species.arrow_forward
- Can I please get this answered? With the correct number of significant digits.arrow_forwardDraw the Hofmann product of the dehydroiodination of this alkyl iodide. ☐ : + Explanation Check esc F1 2 3 I 88 % 5 F5 I. X © tBuOK Click and drag to sta drawing a structure. © 2025 McGraw Hill LLC. All Rights Reserved. Te BI BB F6 W E R Y S H Karrow_forwardCan I please get help with this graph, if you could show exactly where it needs to pass through please.arrow_forward
- Draw the condensed structure of 1,3-dihydroxy-2-pentanone. Explanation Check Click anywhere to draw the first atom of your structure. Х C © 2025 McGraw Hill LLC. All Rights Reserved. Terms of use +arrow_forward0.500 moles of NOCl are placed into a 1.00 L vessesl at 700K and after the system comes to equilibrium, the consentration of NOCl is 0.440 M. Calculate the equilibrium constant Kc for the reaction: 2NOCL (g) --> 2NO (g) + Cl2 (g)arrow_forwardWhat is the hydronium ion concentration in a solution of water that has a hydroxide ion concentrationof 1.0 x 10-2 M?arrow_forward
- Identify conjugate acid-base pairs in the following reactions:HBr (aq) + H2O (l) ⇌ H3O+ (aq) + Br- (aq) - OH (aq) + CH3COOH (aq) ⇌ H2O (l) + CH3COO- (aq)arrow_forward4:45 PM Tue Apr 1 K 77% Problem 9 of 10 Submit Curved arrows are used to illustrate the flow of electrons. Using the provided starting structure, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. Then draw any missing organic intermediates or products for this reaction. Include all lone pairs in the structures. Ignore inorganic byproducts, counterions, and solvents. :0: H Select to Add Arrows HI CH3OH H+ ·HO CH3OH, H+ 0:0 H H Select to Add Arrows tion Versirate CH3OH, H* Select to Draw Productarrow_forwardCan I please get help with this graph? If you can show exactly where it needs to pass through.arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning





