
ORGANIC CHEMISTRY-W/S.G+SOLN.MANUAL
8th Edition
ISBN: 9780134595450
Author: Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 21, Problem 64P
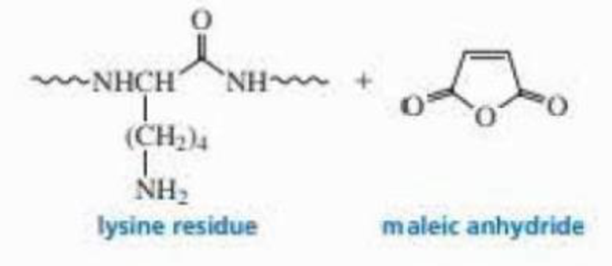
Draw the product obtained when a lysine side chain in a polypeptide reacts with maleic anhydride.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Can you please explain this problem to me and expand it so I can understand the full Lewis dot structure? Thanks!
Can you please explain this problem to me and expand it so I can understand the full Lewis dot structure? Thanks!
Please answer the questions in the photos and please revise any wrong answers. Thank you
Chapter 21 Solutions
ORGANIC CHEMISTRY-W/S.G+SOLN.MANUAL
Ch. 21.1 - a. Explain why, when the imidazole ring of...Ch. 21.2 - a. Which isomer(R)-alanine or (S)-alanineis...Ch. 21.2 - Prob. 4PCh. 21.3 - Prob. 5PCh. 21.3 - Prob. 6PCh. 21.3 - Draw the predominant form for glutamate in a...Ch. 21.3 - a. Why is the pKa of the glutamate side chain...Ch. 21.4 - Calculate the pI of each of the following amino...Ch. 21.4 - a. Which amino acid has the lowest pI value? b....Ch. 21.4 - Prob. 12P
Ch. 21.4 - Prob. 13PCh. 21.4 - Explain why the pI of lysine is the average of the...Ch. 21.5 - What aldehyde is formed when valine is treated...Ch. 21.5 - Prob. 16PCh. 21.5 - Prob. 17PCh. 21.5 - Prob. 18PCh. 21.5 - Prob. 19PCh. 21.6 - Why is excess ammonia used in the preceding...Ch. 21.6 - Prob. 21PCh. 21.6 - What amino acid is formed using the...Ch. 21.6 - Prob. 23PCh. 21.6 - What amino acid is formed when the aldehyde used...Ch. 21.7 - Esterase is an enzyme that catalyzes the...Ch. 21.8 - Draw the tetrapeptide Ala-Thr-Asp-Asn and indicate...Ch. 21.8 - Draw the resonance contributors of the peptide...Ch. 21.8 - Which bonds in the backbone of a peptide can...Ch. 21.9 - An opioid pentapeptide has the following...Ch. 21.9 - What is the configuration about each of the...Ch. 21.9 - Glutathione is a tripeptide whose function is to...Ch. 21.10 - What dipeptides would be formed by heating a...Ch. 21.10 - Suppose you are trying to synthesize the dipeptide...Ch. 21.10 - Show the steps in the synthesis of the...Ch. 21.10 - a. Calculate the overall yield of bradykinin when...Ch. 21.11 - Show the steps in the synthesis of the...Ch. 21.13 - Prob. 37PCh. 21.13 - In determining the primary structure of insulin,...Ch. 21.13 - A decapeptide undergoes partial hydrolysis to give...Ch. 21.13 - Explain why cyanogen bromide does not cleave on...Ch. 21.13 - Indicate the peptides produced from cleavage by...Ch. 21.14 - Prob. 43PCh. 21.14 - Three peptides were obtained from a trypsin...Ch. 21.14 - Prob. 45PCh. 21.15 - How would a protein that resides in the nonpolar...Ch. 21.16 - a. Which would have the greatest percentage of...Ch. 21.17 - When apples that have been cut are exposed to...Ch. 21 - Glycine has pK2 values of 2.34 and 9.60. At what...Ch. 21 - Prob. 50PCh. 21 - A titration curve is a plot of the pH of a...Ch. 21 - Prob. 52PCh. 21 - Aspartame (its structure is on page 1007) has a pl...Ch. 21 - Draw the form of aspartate that predominates at...Ch. 21 - Show how phenylalanine can be prepared by...Ch. 21 - A professor was preparing a manuscript for...Ch. 21 - What aldehydes are formed when the following amino...Ch. 21 - Prob. 58PCh. 21 - Determine the amino acid sequence of a polypeptide...Ch. 21 - Prob. 60PCh. 21 - Prob. 61PCh. 21 - Which is the more effective buffer at...Ch. 21 - Identify the location and type of charge on the...Ch. 21 - Draw the product obtained when a lysine side chain...Ch. 21 - After the polypeptide shown below was treated with...Ch. 21 - Treatment of a polypeptide with 2-mercaptoethanol...Ch. 21 - Show how aspartame can be synthesized using DCCD.Ch. 21 - -Amino acids can be prepared by treating an...Ch. 21 - Reaction of a polypeptide with carboxypeptidase A...Ch. 21 - a. How many different octapeptides can be made...Ch. 21 - Glycine has pKa values of 2.3 and 9.6. Do you...Ch. 21 - A mixture of 15 amino acids gave the fingerprint...Ch. 21 - Write the mechanism for the reaction of an amino...Ch. 21 - Prob. 74PCh. 21 - Show how valine can be prepared by a. a...Ch. 21 - The primary structure of -endorphin, a peptide...Ch. 21 - A chemist wanted to test his hypothesis that the...Ch. 21 - Propose a mechanism for the rearrangement of the...Ch. 21 - A normal polypeptide and a mutant of the...Ch. 21 - Determine the amino acid sequence of a polypeptide...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- (Please be sure that 7 carbons are available in the structure )Based on the 1H NMR, 13C NMR, DEPT 135 NMR and DEPT 90 NMR, provide a reasoning step and arrive at the final structure of an unknown organic compound containing 7 carbons. Dept 135 shows peak to be positive at 128.62 and 13.63 Dept 135 shows peak to be negative at 130.28, 64.32, 30.62 and 19.10.arrow_forward-lease help me answer the questions in the photo.arrow_forwardDefine electronegativity.arrow_forward
- Why do only the immediately adjacent H's show up in the number of peaks? Are there normally peaks for the H's that are 2-3 carbons away?arrow_forwardPlease help me understand this question. Thank you. Organic Chem 1arrow_forwardFor the reaction below, the concentrations at equilibrium are [SO₂] = 0.50 M, [0] = 0.45 M, and [SO3] = 1.7 M. What is the value of the equilibrium constant, K? 2SO2(g) + O2(g) 2SO3(g) Report your answer using two significant figures. Provide your answer below:arrow_forward
- scratch paper, and the integrated rate table provided in class. our scratch work for this test. Content attribution 3/40 FEEDBACK QUESTION 3 - 4 POINTS Complete the equation that relates the rate of consumption of H+ and the rate of formation of Br2 for the given reaction. 5Br (aq) + BrO3 (aq) + 6H (aq) →3Br2(aq) + 3H2O(l) • Your answers should be whole numbers or fractions without any decimal places. Provide your answer below: Search 尚 5 fn 40 * 00 99+ 2 9 144 a [arrow_forward(a) Write down the structure of EDTA molecule and show the complex structure with Pb2+ . (b) When do you need to perform back titration? (c) Ni2+ can be analyzed by a back titration using standard Zn2+ at pH 5.5 with xylenol orange indicator. A solution containing 25.00 mL of Ni2+ in dilute HCl is treated with 25.00 mL of 0.05283 M Na2EDTA. The solution is neutralized with NaOH, and the pH is adjusted to 5.5 with acetate buffer. The solution turns yellow when a few drops of indicator are added. Titration with 0.02299 M Zn2+ requires 17.61 mL to reach the red end point. What is the molarity of Ni2+ in the unknown?arrow_forwardA compound has the molecular formula CH40, and shows a strong IR absorption at 2850-3150 cm. The following signals appear in the 'H NMR spectrum: 1.4 ppm (triplet, 6H), 4.0 ppm (quartet, 4H), 6.8 ppm (broad singlet, 4H). Which of the following structures is consistent with these data? Select the single best answer. OCH CH₂ x OCH2CH3 CH₂OCH3 OH CH₂OCH OH CH, OCH₁ CH₂OCH, CH₂OCH HO OH ° CH₂OCH3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning
Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY