
An interesting pair of consecutive reactions involve the absorption of ethyl alcohol by the body, which is a first-order process, and the consequent oxidation of alcohol to acetaldehyde by liver alcohol dehydrogenase (LADH), which is a zeroth order process. The differential changes in the three states of ethanol can therefore be described as
which are slightly modified from equations
(a)
Interpretation:
The species
Concept introduction:
In the consecutive reactions the product of first reaction acts as the reactant for the second reaction and so on. The radioactive decays are one of the examples of the consecutive reactions. The simple two step consecutive reaction is shown below.
Answer to Problem 20.57E
The species
Explanation of Solution
The given consecutive reaction involves the absorption of ethyl alcohol by the body which is a first order reaction followed by the oxidation of ethyl alcohol to acetaldehyde in the presence of liver alcohol dehydrogenase (LADH).
The rate of change of concentration of the three species is given below.
From the above consecutive reaction it is concluded that
The species
(b)
Interpretation:
The integrated form for
Concept introduction:
In the consecutive reactions, the product of first reaction acts as the reactant for the second reaction and so on. The radioactive decays are one of the examples of the consecutive reactions. The simple two step consecutive reaction is shown below.
Answer to Problem 20.57E
The integrated form for
Explanation of Solution
The differential change in the concentration of
The integrated rate law for the first order reaction of
Substitute equation (2) in equation (1).
Integrate the above equation.
The integrated form for
(c)
Interpretation:
The integrated form for
Concept introduction:
In the consecutive reactions the product of first reaction acts as the reactant for the second reaction and so on. The radioactive decays are one of the examples of the consecutive reactions. The simple two step consecutive reaction is shown below.
Answer to Problem 20.57E
The integrated form for
Explanation of Solution
The differential change in the concentration of
The integrated form for
Substitute equation (4) in equation (3).
Integrate the above equation.
The above exponential function is integrated using the identity shown below.
On applying this identity on the exponential function the expression for the integrated form for
The integrated form for
(d)
Interpretation:
The graph for
Concept introduction:
In the consecutive reactions the product of first reaction acts as the reactant for the second reaction and so on. The radioactive decays are one of the examples of the consecutive reactions. The simple two step consecutive reaction is shown below.
Answer to Problem 20.57E
The graph for
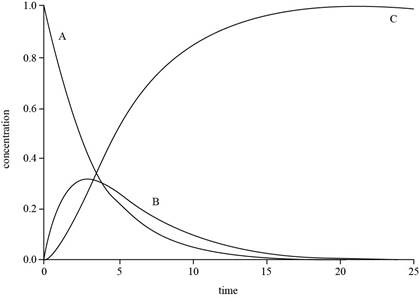
Explanation of Solution
The expressions for
The value of
The plot for
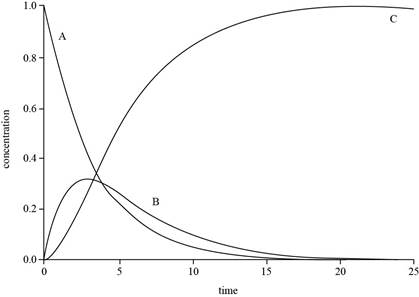
Figure 1
The concentration of
The graph for
Want to see more full solutions like this?
Chapter 20 Solutions
Student Solutions Manual for Ball's Physical Chemistry, 2nd
- For the following compound: HO -H Draw a mechanism for the tautomerization process under BASIC conditions: Mechanism A: H-O: H-OH H-O HH H-OO Mechanism B: H-Q Mechanism C: Θ OH H-O: Mechanism D: H-O H- H-OO C H-OO H- H- H-OO HH OH -H - HON H :OH H-Harrow_forwardidentify the product (or multiple products) for each of the following reactions: CI 1) NaNH2 (excess) ठ Cl 2) H₂O Hz H₂SO₂, H₂O HgSO Lindlar's catalyst 1) n-BuLi 2) 1)9-BBN 2) H₂O, NaOH ? Br H A B C afó gó H OA B O c OD E OF D E F H H Na, NHarrow_forwardIdentify the product (or multiple products) for each of the following reactions: ? or CI CI 1) NaNHz (excess) 2) H₂O OA OB O C OD OE OF H₂SO₂, H₂O Hq50. 1) n-BuLi 2) Br 1) 9-BBN 2) H₂O₂, NaOH A B H H متته D E H H H H C H H F H H H₂ Lindlar's catalyst Na NHarrow_forward
- Identify the product (or multiple products) for each of the following reactions: O A OB Oc OD OE OF CI CI 1) NaNH2 (excess) 2) H₂O H₂ H₂SO2, H₂O HgSO Lindlar's catalyst 1) n-BuLi 2) Br 1)9-BBN 2) H₂O₂, NaOH ? Na, NH3 C H A H H مننه مننه منن مننه H F H H E مند H D H Harrow_forwardFor the following compound: HO H Draw a mechanism for the tautomerization process under BASIC conditions: Mechanism A: + H-O: H-OH₂ H Mechanism B: H-Ö: HO-H H-OO -H H HH H H HH H-O: H-OO H-OO -H H e -H : OH Θ Mechanism C: Θ A : OH H-O: H H H-O-H 0. Mechanism D: e.. : OH :0 H H-O-H H-O: H-OO :O H -H H H сём H 0 :0 + H Θ H H H-arrow_forwardFor the following compound: H OH Draw a mechanism for the tautomerization process under ACIDIC conditions: Mechanism A: Θ :OH O O-H HO 0: Mechanism B: :O-H e.. Θ :OH Mechanism C: H HO-H :0: Θ 0: H H e.. : OH 0: "Θ HH O. :OH :OH O-H O-H Mechanism D: :OH H-OH₂ :OH HO-H 0: © O-H H HH 0: HHarrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





