
EP ORGANIC CHEMISTRY-OWL V2 ACCESS
8th Edition
ISBN: 9781305582453
Author: Brown
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 20, Problem 20.54P
Interpretation Introduction
Interpretation: The mechanism for the given reaction has to be proposed:
Concept Introduction:
Pericyclic reaction:
The pericyclic reaction involves the redistribution of six electrons through the formation of a cyclic transition state from which a diene compound gets converted into its isomeric form.
Example with mechanism of a pericyclic reaction:
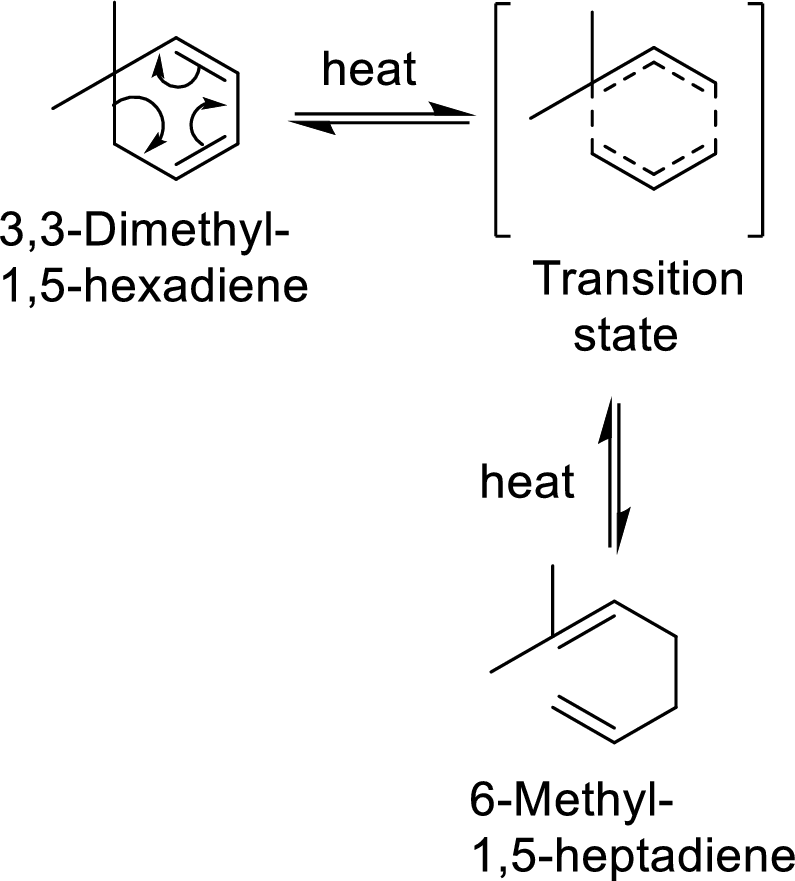
In this mechanism, two pi-bonds and one sigma bond of the reactant molecule has been broken and formed two new pi-bonds through a cyclic transition state.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What is the pH of a 1.0 L buffer made with 0.300 mol of HF (Ka = 6.8 × 10⁻⁴) and 0.200 mol of NaF to which 0.160 mol of NaOH were added?
Can I please get help with this.
Determine if the following salt is neutral, acidic or basic. If acidic or basic, write the appropriate equilibrium equation for the acid or base that exists when the salt is dissolved in aqueous solution. If neutral, simply write only NR. Be sure to include the proper phases for all species within the reaction. N₂H₅ClO₄
Chapter 20 Solutions
EP ORGANIC CHEMISTRY-OWL V2 ACCESS
Ch. 20.1 - Prob. 20.1PCh. 20.1 - Estimate the stabilization gained as a result of...Ch. 20.2 - Predict the product(s) formed by addition of one...Ch. 20.3 - Prob. 20.4PCh. 20.3 - Prob. 20.5PCh. 20.4 - Prob. 20.6PCh. 20.5 - Prob. 20.7PCh. 20.5 - Prob. 20.8PCh. 20.5 - Prob. 20.9PCh. 20.6 - Prob. 20.10P
Ch. 20.6 - Prob. 20.11PCh. 20.6 - Prob. 20.12PCh. 20 - If an electron is added to 1,3-butadiene, into...Ch. 20 - Prob. 20.15PCh. 20 - Predict the structure of the major product formed...Ch. 20 - Predict the major product formed by 1,4-addition...Ch. 20 - Predict the structure of the major 1,2-addition...Ch. 20 - Prob. 20.19PCh. 20 - Prob. 20.20PCh. 20 - Prob. 20.21PCh. 20 - Prob. 20.22PCh. 20 - Prob. 20.23PCh. 20 - Pyridine exhibits a UV transition of the type n at...Ch. 20 - Prob. 20.25PCh. 20 - Prob. 20.26PCh. 20 - Prob. 20.27PCh. 20 - Write the frontier molecular orbital analysis for...Ch. 20 - Prob. 20.29PCh. 20 - Draw structural formulas for the products of...Ch. 20 - Propose structural formulas for compounds A and B...Ch. 20 - Under certain conditions, 1,3-butadiene can...Ch. 20 - Prob. 20.33PCh. 20 - Prob. 20.34PCh. 20 - The following triene undergoes an intramolecular...Ch. 20 - Prob. 20.36PCh. 20 - Prob. 20.37PCh. 20 - Prob. 20.38PCh. 20 - Prob. 20.39PCh. 20 - The Diels-Alder reaction is not limited to making...Ch. 20 - The first step in a synthesis of dodecahedrane...Ch. 20 - Bicyclo-2,5-heptadiene can be prepared in two...Ch. 20 - Prob. 20.43PCh. 20 - Prob. 20.44PCh. 20 - Following is a retrosynthetic scheme for the...Ch. 20 - Prob. 20.46PCh. 20 - Prob. 20.47PCh. 20 - Prob. 20.48PCh. 20 - Prob. 20.49PCh. 20 - Prob. 20.50PCh. 20 - What reaction presented in this chapter is...Ch. 20 - Claisen rearrangement of an allyl phenyl ether...Ch. 20 - Prob. 20.53PCh. 20 - Prob. 20.54PCh. 20 - We now continue the use of organic chemistry...Ch. 20 - Write the products of the following sequences of...
Knowledge Booster
Similar questions
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning