
FUNDAMENTALS OF PHYSICS (LLF)+WILEYPLUS
11th Edition
ISBN: 9781119459132
Author: Halliday
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 19, Problem 7Q
(a) Rank the four paths of Fig. 19-16 according to the work done by the gas, greatest first, (b) Rank paths 1, 2, and 3 according to the change in the internal energy of the gas, most positive first and most negative last.
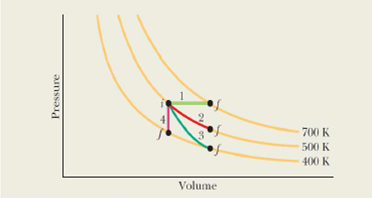
Figure 19-16 A p-V diagram representing four special process for an ideal monatomic gas.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Please don't use Chatgpt will upvote and give handwritten
5. An object moves in a horizontal plane with
constant speed on the path shown. At which
marked point is the magnitude of its
acceleration greatest?
A
B
Ꭰ
E
C
No chatgpt pls will upvote
Chapter 19 Solutions
FUNDAMENTALS OF PHYSICS (LLF)+WILEYPLUS
Ch. 19 - For four situations for an ideal gas, the table...Ch. 19 - In the p-V diagram of Fig. 19-17, the gas does 5 J...Ch. 19 - For a temperature increase of T1, a certain amount...Ch. 19 - The dot in Fig, 19-18a represents the initial...Ch. 19 - A certain amount of energy is to be transferred as...Ch. 19 - The dot in Fig. 19-18b represents the initial...Ch. 19 - a Rank the four paths of Fig. 19-16 according to...Ch. 19 - The dot in Fig. 19-18c represents the initial...Ch. 19 - Prob. 9QCh. 19 - Does the temperature of an ideal gas increase,...
Ch. 19 - Prob. 1PCh. 19 - Gold has a molar mass of 197 g/mol. a How many...Ch. 19 - SSM Oxygen gas having a volume of 1000 cm3 at...Ch. 19 - A quantity of ideal gas at: 10.0C and 100 kPa...Ch. 19 - The best laboratory vacuum has a pressure of about...Ch. 19 - Water bottle in a hot car. In the American...Ch. 19 - Suppose 1.80 mol of an ideal gas is taken from a...Ch. 19 - Compute a the number of moles and b the number of...Ch. 19 - An automobile tire has a volume of 1.64 102 m3...Ch. 19 - A container encloses 2 mol of an ideal gas that...Ch. 19 - SSM ILW WWW Air that initially occupies 0.140 m3...Ch. 19 - GO Submarine rescue. When the U.S. submarine...Ch. 19 - Prob. 13PCh. 19 - In the temperature range 310 K to 330 K, the...Ch. 19 - Suppose 0.825 mol of an ideal gas undergoes an...Ch. 19 - An air bubble of volume 20 cm3 is at the bottom of...Ch. 19 - GO Container A in Fig. 19-22 holds an ideal gas at...Ch. 19 - The temperature and pressure in the Suns...Ch. 19 - a Compute the rms speed of a nitrogen molecule at...Ch. 19 - Calculate the rms speed of helium atoms at 1000 K....Ch. 19 - SSM The lowest possible temperature in outer space...Ch. 19 - Find the rms speed of argon atoms at 313 K. See...Ch. 19 - A beam of hydrogen molecules H2 is directed toward...Ch. 19 - At 273 K and 1.00 102 atm, the density of a gas...Ch. 19 - Prob. 25PCh. 19 - Prob. 26PCh. 19 - Water standing in the open at 32.0C evaporates...Ch. 19 - At what frequency would the wavelength of sound in...Ch. 19 - SSM The atmospheric density at an altitude of 2500...Ch. 19 - Prob. 30PCh. 19 - In a certain particle accelerator, protons travel...Ch. 19 - Prob. 32PCh. 19 - Prob. 33PCh. 19 - Prob. 34PCh. 19 - Prob. 35PCh. 19 - The most probable speed of the molecules in a gas...Ch. 19 - Prob. 37PCh. 19 - Figure 19-24 gives the probability distribution...Ch. 19 - At what temperature does the rms speed of a...Ch. 19 - Two containers are at the same temperature. The...Ch. 19 - Prob. 41PCh. 19 - What is the internal energy of 1.0 mol of an ideal...Ch. 19 - Prob. 43PCh. 19 - GO One mole of ail ideal diatomic gas goes from a...Ch. 19 - ILW The mass of a gas molecule can be computed...Ch. 19 - Under constant pressure, the temperature of 2.00...Ch. 19 - The temperature of 2.00 mol of an ideal monatomic...Ch. 19 - GO When 20.9 J was added as heat to a particular...Ch. 19 - SSM A container holds a mixture of three...Ch. 19 - We give 70 J as heat to a diatomic gas, which then...Ch. 19 - Prob. 51PCh. 19 - GO Suppose 12.0 g of oxygen O2 gas is heated at...Ch. 19 - SSM WWW Suppose 4.00 mol of an ideal diatomic gas...Ch. 19 - We know that for an adiabatic process pV = a...Ch. 19 - A certain gas occupies a volume of 4.3 L at a...Ch. 19 - Suppose 1.00 L of a gas with = 1.30, initially at...Ch. 19 - The volume of an ideal gas is adiabatically...Ch. 19 - GO Opening champagne. In a bottle of champagne,...Ch. 19 - GO Figure 19-26 shows two paths that may be taken...Ch. 19 - Prob. 60PCh. 19 - GO A gas is to be expanded from initial state i to...Ch. 19 - GO An ideal diatomic gas, with rotation but no...Ch. 19 - Figure 19-27 shows a cycle undergone by 1.00 mol...Ch. 19 - Calculate the work done by an external agent...Ch. 19 - An ideal gas undergoes an adiabatic compression...Ch. 19 - Prob. 66PCh. 19 - An ideal monatomic gas initially has a temperature...Ch. 19 - Prob. 68PCh. 19 - SSM The envelope and basket of a hot-air balloon...Ch. 19 - An ideal gas, at initial temperature T1 and...Ch. 19 - Prob. 71PCh. 19 - At what temperature do atoms of helium gas have...Ch. 19 - Prob. 73PCh. 19 - Prob. 74PCh. 19 - The temperature of 3.00 mol of a gas with CV =...Ch. 19 - During a compression at a constant pressure of 250...Ch. 19 - SSM Figure 19-28 shows a hypothetical speed...Ch. 19 - Prob. 78PCh. 19 - SSM An ideal gas undergoes isothermal compression...Ch. 19 - Oxygen O2 gas at 273 K and 1.0 atm is confined to...Ch. 19 - An ideal pas is taken through a complete cycle in...Ch. 19 - Prob. 82PCh. 19 - SSM A sample of ideal gas expands from an initial...Ch. 19 - An ideal gas with 3.00 mol is initially in state 1...Ch. 19 - A steel lank contains 300 g of ammonia gas NH3 at...Ch. 19 - In an industrial process the volume of 25.0 mol of...Ch. 19 - Figure 19-29 shows a cycle consisting of five...Ch. 19 - An ideal gas initially at 300 K is compressed at a...Ch. 19 - A pipe of length L = 25.0 m that is open at one...Ch. 19 - In a motorcycle engine, a piston is forced down...Ch. 19 - For adiabatic processes in an ideal gas, show that...Ch. 19 - Air at 0.000C and 1.00 atm pressure has a density...Ch. 19 - Prob. 93PCh. 19 - Prob. 94PCh. 19 - Prob. 95PCh. 19 - For air near 0C, by how much does the speed of...Ch. 19 - Prob. 97P
Additional Science Textbook Solutions
Find more solutions based on key concepts
How do food chains and food webs differ? Which is the more accurate representation of feeding relationships in ...
Biology: Life on Earth (11th Edition)
Name the components (including muscles) of the thoracic cage. List the contents of the thorax.
Human Physiology: An Integrated Approach (8th Edition)
What is the anatomical position? Why is it important that you learn this position?
Anatomy & Physiology (6th Edition)
How can the freezing of water crack boulders?
Campbell Biology in Focus (2nd Edition)
12. Take a spring and cut it in half to make two springs. Is the spring constant of these smaller springs large...
College Physics: A Strategic Approach (3rd Edition)
8. A human maintaining a vegan diet (containing no animal products) would be a:
a. producer
b. primary consume...
Human Biology: Concepts and Current Issues (8th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- No chatgpt pls will upvotearrow_forwardUniform Circular motion. 1. Mini Lecture 2. Let the position of a particle be given by: (t) = Rcos (wt)i + Rsin (wt)j 3. Calculate the expression for the velocity vector and show that the velocity vector is tangential to the circumference of the circle. 4. Calculate the expression for the acceleration vector and show that the acceleration vector points radially inward. 5. Calculate the magnitude of the velocity and magnitude of the acceleration, and therefore show that v2 a = Rarrow_forward4. A ball is thrown vertically up, its speed. slowing under the influence of gravity. Suppose (A) we film this motion and play the tape backward (so the tape begins with the ball at its highest point and ends with it reaching the point from which it was released), and (B) we observe the motion of the ball from a frame of reference moving up at the initial speed of the ball. The ball has a downward acceleration g in: a. A and B b. Only A c. Only B d. Neither A nor Barrow_forward
- 2. Consider a 2.4 m long propeller that operated at a constant 350 rpm. Find the acceleration of a particle at the tip of the propeller.arrow_forward2. A football is kicked at an angle 37.0° above the horizontal with a velocity of 20.0 m/s, as Calculate (a) the maximum height, (b) the time of travel before the football hits the ground, and (c) how far away it hits the ground. Assume the ball leaves the foot at ground level, and ignore air resistance, wind, and rotation of the ball.arrow_forwardPlease don't use Chatgpt will upvote and give handwritten solutionarrow_forward
- Cam mechanisms are used in many machines. For example, cams open and close the valves in your car engine to admit gasoline vapor to each cylinder and to allow the escape of exhaust. The principle is illustrated in the figure below, showing a follower rod (also called a pushrod) of mass m resting on a wedge of mass M. The sliding wedge duplicates the function of a rotating eccentric disk on a camshaft in your car. Assume that there is no friction between the wedge and the base, between the pushrod and the wedge, or between the rod and the guide through which it slides. When the wedge is pushed to the left by the force F, the rod moves upward and does something such as opening a valve. By varying the shape of the wedge, the motion of the follower rod could be made quite complex, but assume that the wedge makes a constant angle of 0 = 15.0°. Suppose you want the wedge and the rod to start from rest and move with constant acceleration, with the rod moving upward 1.00 mm in 8.00 ms. Take m…arrow_forwardNo chatgpt pls will upvotearrow_forwardNo chatgpt pls will upvotearrow_forward
- No chatgpt plsarrow_forwardA rectangular current loop (a = 15.0 cm, b = 34.0 cm) is located a distance d = 10.0 cm near a long, straight wire that carries a current (Iw) of 17.0 A (see the drawing). The current in the loop is IL = 21.0 A. Determine the magnitude of the net magnetic force that acts on the loop. Solve in N. a b IL Iwarrow_forwardTwo long, straight wires are separated by distance, d = 22.0 cm. The wires carry currents of I1 = 7.50 A and I2 = 5.50 A in opposite directions, as shown in the figure. Find the magnitude of the net magnetic field at point (B). Let r₁ = 12.0 cm, r2 = 7.00 cm, and r3 = 13.0 cm. Solve in T. 12 d A √3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning


Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers, Technology ...
Physics
ISBN:9781305116399
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers with Modern ...
Physics
ISBN:9781337553292
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

College Physics
Physics
ISBN:9781285737027
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning
Thermodynamics: Crash Course Physics #23; Author: Crash Course;https://www.youtube.com/watch?v=4i1MUWJoI0U;License: Standard YouTube License, CC-BY