
Physics for Scientists and Engineers with Modern Physics
4th Edition
ISBN: 9780131495081
Author: Douglas C. Giancoli
Publisher: Addison-Wesley
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 19, Problem 68P
(III) Suppose the insulating qualities of the wall of a house come mainly from a 4.0-in, layer of brick and an R-19 layer of insulation, as shown in Fig. 19-35. What is the total rate of heat loss through such a wall, if its total area is 195 ft2 and the temperature difference across it is 12 F°?
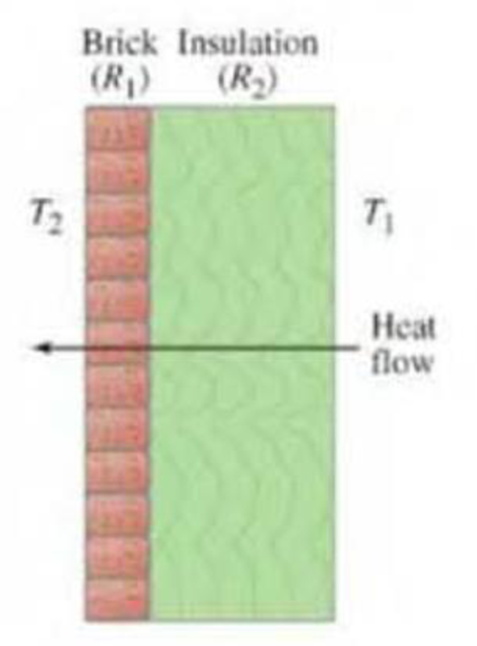
FIGURE 19-35 Problem 68.
Two layers insulating a wall.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Please solve and answer this problem correctly please. Thank you!!
Please solve and answer this problem correctly please. Thank you!!
a) Use the node-voltage method to find v1, v2, and
v3 in the circuit in Fig. P4.14.
b) How much power does the 40 V voltage source
deliver to the circuit?
Figure P4.14
302
202
w
w
+
+
+
40 V
V1
80 Ω 02
ΣΑΩ
28 A
V3 +
w
w
102
202
Chapter 19 Solutions
Physics for Scientists and Engineers with Modern Physics
Ch. 19.2 - Return to the Chapter-Opening Question, page 496,...Ch. 19.5 - Prob. 1BECh. 19.5 - How much more ice at 10C would be needed in...Ch. 19.6 - What would be the internal energy change in...Ch. 19.7 - Is the work done by the gas in process ADB of Fig....Ch. 19.7 - In Example 1910, if the heat lost from the gas in...Ch. 19.10 - Fanning yourself on a hot day cools you by (a)...Ch. 19 - What happens to the work done on a jar of orange...Ch. 19 - Prob. 2QCh. 19 - Prob. 3Q
Ch. 19 - Prob. 4QCh. 19 - Prob. 5QCh. 19 - Why does water in a canteen stay cooler if the...Ch. 19 - Explain why burns caused by steam at 100C on the...Ch. 19 - Prob. 8QCh. 19 - Will potatoes cook faster if the water is boiling...Ch. 19 - Prob. 10QCh. 19 - Prob. 11QCh. 19 - Use the conservation of energy to explain why the...Ch. 19 - In an isothermal process, 3700 J of work is done...Ch. 19 - Explorers on failed Arctic expeditions have...Ch. 19 - Why is wet sand at the beach cooler to walk on...Ch. 19 - When hot-air furnaces are used to heat a house,...Ch. 19 - Is it possible for the temperature of a system to...Ch. 19 - Discuss how the first law of thermodynamics can...Ch. 19 - Explain in words why CP is greater than CV.Ch. 19 - Prob. 20QCh. 19 - An ideal monatomic gas is allowed to expand slowly...Ch. 19 - Ceiling fans are sometimes reversible, so that...Ch. 19 - Goose down sleeping bags and parkas are often...Ch. 19 - Microprocessor chips nowadays have a heat sink...Ch. 19 - Sea breezes are often encountered on sunny days at...Ch. 19 - The Earth cools off at night much more quickly...Ch. 19 - Explain why air-temperature readings are always...Ch. 19 - A premature baby in an incubator can be...Ch. 19 - Prob. 29QCh. 19 - A 22C day is warm, while a swimming pool at 22C...Ch. 19 - Prob. 32QCh. 19 - Prob. 33QCh. 19 - Prob. 34QCh. 19 - Prob. 35QCh. 19 - An emergency blanket is a thin shiny...Ch. 19 - Explain why cities situated by the ocean tend to...Ch. 19 - (I) To what temperature will 8700 J of heat raise...Ch. 19 - Prob. 2PCh. 19 - Prob. 3PCh. 19 - (II) A British thermal unit (Btu) is a unit of...Ch. 19 - Prob. 5PCh. 19 - Prob. 6PCh. 19 - (I) An automobile cooling system holds 18 L of...Ch. 19 - Prob. 8PCh. 19 - (II) (a) How much energy is required to bring a...Ch. 19 - Prob. 10PCh. 19 - Prob. 11PCh. 19 - (II) A hot iron horseshoe (mass = 0.40kg), just...Ch. 19 - (II) A 31.5-g glass thermometer reads 23.6C before...Ch. 19 - Prob. 14PCh. 19 - (II) When a 290-g piece of iron at 180C is placed...Ch. 19 - (II) The heat capacity. C, of an object is defined...Ch. 19 - (II) The 1.20-kg head of a hammer has a speed of...Ch. 19 - (I) How much heat is needed to melt 26.50kg of...Ch. 19 - (I) During exercise, a person may give off 180...Ch. 19 - (II) A 35g ice cube at its melting point is...Ch. 19 - (II) High-altitude mountain climbers do not eat...Ch. 19 - (II) An iron boiler of mass 180 kg contains 730kg...Ch. 19 - (II) In a hot days race, a bicyclist consumes 8.0...Ch. 19 - (II) The specific heat of mercury is 138 J/kg C....Ch. 19 - Prob. 25PCh. 19 - (II) A 58-kg ice-skater moving at 7.5 m/s glides...Ch. 19 - (I) Sketch a PV diagram of the following process:...Ch. 19 - (I) A gas is enclosed in a cylinder fitted with a...Ch. 19 - (II) The pressure in an ideal gas is cut in half...Ch. 19 - (II) A 1.0-L volume of air initially at 3.5 atm of...Ch. 19 - (II) Consider the following two-step process. Heat...Ch. 19 - (II) The PV diagram in Fig. 1931 shows two...Ch. 19 - (II) Suppose 2.60 mol of an ideal gas of volume V1...Ch. 19 - (II) In an engine, an almost ideal gas is...Ch. 19 - (II) One and one-half moles of an ideal monatomic...Ch. 19 - (II) Determine (a) the work done and (b) the...Ch. 19 - (II) How much work is done by a pump to slowly...Ch. 19 - (II) When a gas is taken from a to c along the...Ch. 19 - (III) In the process of taking a gas from state a...Ch. 19 - (III) Suppose a gas is taken clockwise around the...Ch. 19 - (III) Determine the work done by 1.00 mol of a van...Ch. 19 - (I) What is the internal energy of 4.50 mol of an...Ch. 19 - Prob. 43PCh. 19 - Prob. 44PCh. 19 - Prob. 45PCh. 19 - What gas is it? (II) Show that the work done by n...Ch. 19 - (II) An audience of 1800 fills a concert hall of...Ch. 19 - Prob. 48PCh. 19 - Prob. 49PCh. 19 - (III) A 1.00-mol sample of an ideal diatomic gas...Ch. 19 - (I) A 1.00-mol sample of an ideal diatomic gas,...Ch. 19 - (II) Show, using Eqs. 196 and 1915, that the work...Ch. 19 - (III) A 3.65-mol sample of an ideal diatomic gas...Ch. 19 - (II) An ideal monatomic gas, consisting of 2.8 mol...Ch. 19 - (III) A 1.00-mol sample of an ideal monatomic gas,...Ch. 19 - (III) Consider a parcel of air moving to a...Ch. 19 - Prob. 57PCh. 19 - (I) One end of a 45-cm-long copper rod with a...Ch. 19 - (II) How long does it take the Sun to melt a block...Ch. 19 - (II) Heat conduction to skin. Suppose 150 W of...Ch. 19 - (II) A ceramic teapot ( = 0.70) and a shiny one (...Ch. 19 - (II) A copper rod and an aluminum rod of the same...Ch. 19 - Prob. 63PCh. 19 - Prob. 64PCh. 19 - (III) A house thermostat is normally set to 22C,...Ch. 19 - (III) Approximately how long should it take 9.5 kg...Ch. 19 - (III) A cylindrical pipe has inner radius R1 and...Ch. 19 - (III) Suppose the insulating qualities of the wall...Ch. 19 - Prob. 69GPCh. 19 - (a) Find the total power radiated into space by...Ch. 19 - Prob. 71GPCh. 19 - A mountain climber wears a goose-down jacket 3.5...Ch. 19 - Prob. 73GPCh. 19 - Estimate the rate at which heat can he conducted...Ch. 19 - A marathon runner has an average metabolism rate...Ch. 19 - A house has well-insulated walls 19.5 cm thick...Ch. 19 - In a typical game of squash (Fig. 19-36), two...Ch. 19 - A bicycle pump is a cylinder 22 cm long and 3.0 cm...Ch. 19 - Prob. 79GPCh. 19 - The temperature within the Earths crust increases...Ch. 19 - An ice sheet forms on a lake. The air above the...Ch. 19 - An iron meteorite melts when it enters the Earths...Ch. 19 - A scuba diver releases a 3.60-cm-diameter...Ch. 19 - A reciprocating compressor is a device that...Ch. 19 - The temperature of the glass surface of a 75-W...Ch. 19 - Suppose 3.0 mol of neon (an ideal monatomic gas)...Ch. 19 - At very low temperatures, the molar specific heat...Ch. 19 - A diesel engine accomplishes ignition without a...Ch. 19 - When 6.30 105 J of heat is added to a gas...Ch. 19 - In a cold environment, a person can lose heat by...Ch. 19 - Prob. 91GP
Additional Science Textbook Solutions
Find more solutions based on key concepts
How Would the experiments result charge if oxygen (O2) were induced in the spark chamber?
Biology: Life on Earth with Physiology (11th Edition)
Choose the best answer to each of the following. Explain your reasoning. Which lists the major steps of solar s...
Cosmic Perspective Fundamentals
The bioremediation process shown in the photograph is used to remove benzene and other hydrocarbons from soil c...
Microbiology: An Introduction
Carefully examine the common sedimentary rocks shown In Figure 2.13. Use these photos and the preceding discuss...
Applications and Investigations in Earth Science (9th Edition)
Police Captain Jeffers has suffered a myocardial infarction. a. Explain to his (nonmedically oriented) family w...
Human Physiology: An Integrated Approach (8th Edition)
What are four functions of connective tissue?
Anatomy & Physiology (6th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- Please solve and answer this problem correctly please. Thank you!!arrow_forwardYou're on an interplanetary mission, in an orbit around the Sun. Suppose you make a maneuver that brings your perihelion in closer to the Sun but leaves your aphelion unchanged. Then you must have Question 2 options: sped up at perihelion sped up at aphelion slowed down at perihelion slowed down at aphelionarrow_forwardThe force of the quadriceps (Fq) and force of the patellar tendon (Fp) is identical (i.e., 1000 N each). In the figure below angle in blue is Θ and the in green is half Θ (i.e., Θ/2). A) Calculate the patellar reaction force (i.e., R resultant vector is the sum of the horizontal component of the quadriceps and patellar tendon force) at the following joint angles: you need to provide a diagram showing the vector and its components for each part. a1) Θ = 160 degrees, a2) Θ = 90 degrees. NOTE: USE ONLY TRIGNOMETRIC FUNCTIONS (SIN/TAN/COS, NO LAW OF COSINES, NO COMPLICATED ALGEBRAIC EQUATIONS OR ANYTHING ELSE, ETC. Question A has 2 parts!arrow_forward
- The force of the quadriceps (Fq) and force of the patellar tendon (Fp) is identical (i.e., 1000 N each). In the figure below angle in blue is Θ and the in green is half Θ (i.e., Θ/2). A) Calculate the patellar reaction force (i.e., R resultant vector is the sum of the horizontal component of the quadriceps and patellar tendon force) at the following joint angles: you need to provide a diagram showing the vector and its components for each part. a1) Θ = 160 degrees, a2) Θ = 90 degrees. NOTE: USE DO NOT USE LAW OF COSINES, NO COMPLICATED ALGEBRAIC EQUATIONS OR ANYTHING ELSE, ETC. Question A has 2 parts!arrow_forwardNo chatgpt pls will upvotearrow_forwardThe force of the quadriceps (Fq) and force of the patellar tendon (Fp) is identical (i.e., 1000 N each). In the figure below angle in blue is Θ and the in green is half Θ (i.e., Θ/2). A) Calculate the patellar reaction force (i.e., R resultant vector is the sum of the horizontal component of the quadriceps and patellar tendon force) at the following joint angles: you need to provide a diagram showing the vector and its components for each part. a1) Θ = 160 degrees, a2) Θ = 90 degrees. NOTE: USE ONLY TRIGNOMETRIC FUNCTIONS (SIN/TAN/COS, NO LAW OF COSINES, NO COMPLICATED ALGEBRAIC EQUATIONS OR ANYTHING ELSE, ETC. Question A has 2 parts!arrow_forward
- ་ The position of a particle is described by r = (300e 0.5t) mm and 0 = (0.3t²) rad, where t is in seconds. Part A Determine the magnitude of the particle's velocity at the instant t = 1.5 s. Express your answer to three significant figures and include the appropriate units. v = Value Submit Request Answer Part B ? Units Determine the magnitude of the particle's acceleration at the instant t = 1.5 s. Express your answer to three significant figures and include the appropriate units. a = Value A ? Unitsarrow_forwardSolve and answer the question correctly please. Thank you!!arrow_forwardSolve and answer the question correctly please. Thank you!!arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
 Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

College Physics
Physics
ISBN:9781285737027
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning


Physics for Scientists and Engineers
Physics
ISBN:9781337553278
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers with Modern ...
Physics
ISBN:9781337553292
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

College Physics
Physics
ISBN:9781938168000
Author:Paul Peter Urone, Roger Hinrichs
Publisher:OpenStax College
Heat Transfer: Crash Course Engineering #14; Author: CrashCourse;https://www.youtube.com/watch?v=YK7G6l_K6sA;License: Standard YouTube License, CC-BY