
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 19, Problem 19.58SP
A compound of formula C11H16N2 gives the IR, 1HNMR, and 13CNMR spectra shown. The proton NMR peak at δ2.0 disappears on shaking with D2O. Propose a structure for this compound, and show how your structure accounts for the observed absorptions.
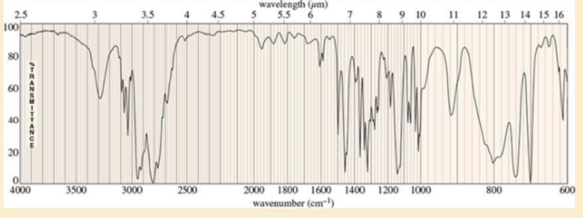
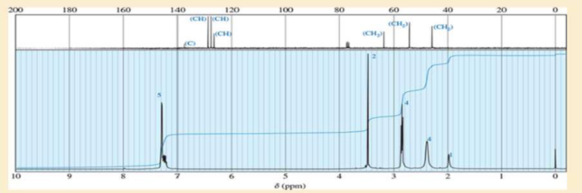
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Write the equilibrium constant expression for the following system at equilibrium:
I2 (g) ⇌ 2 I (g)
Don't used hand raiting and don't used Ai solution
We learned four factors (ARIO) for comparing the relative acidity of compounds. When two of these factors are in
competition, the order of priority is the order in which these factors were covered ("atom" being the most important factor
and "orbital" being the least important). However, we also mentioned that there are exceptions to this order of priority.
Compare the two compounds and identify the exception.
OH
PK-4.75
SH
PK-10.6
5.
"Resonance" is more important than "atom" because the conjugate base of first compound is more stable than the
second.
"Atom" is more important than "resonance" because the conjugate base of first compound is more stable than the
second.
"Resonance" is more important than "atom" because the conjugate base of second compound is more stable than
the first.
"Atom" is more important than "resonance" because the conjugate base of second compound is more stable than
the first.
Chapter 19 Solutions
Organic Chemistry (9th Edition)
Ch. 19.2A - Prob. 19.1PCh. 19.2B - Prob. 19.2PCh. 19.2B - Give correct names for the following amines:Ch. 19.3 - Prob. 19.4PCh. 19.4 - Prob. 19.5PCh. 19.6 - Rank each set of compounds in order of increasing...Ch. 19.8A - Prob. 19.7PCh. 19.8C - Prob. 19.8PCh. 19.8C - Prob. 19.9PCh. 19.8D - a. Show how fragmentation occurs to give the base...
Ch. 19.10B - Propose a mechanism for nitration of pyridine at...Ch. 19.10B - Prob. 19.12PCh. 19.10C - Prob. 19.13PCh. 19.10C - Prob. 19.14PCh. 19.11 - Propose a mechanism to show the individual...Ch. 19.11 - Prob. 19.16PCh. 19.12 - Give the products expected from the following...Ch. 19.13 - Prob. 19.18PCh. 19.13 - Prob. 19.19PCh. 19.14 - Prob. 19.20PCh. 19.15 - Prob. 19.21PCh. 19.15 - Prob. 19.22PCh. 19.16 - Prob. 19.23PCh. 19.17 - Prob. 19.24PCh. 19.17 - Prob. 19.25PCh. 19.18 - Prob. 19.26PCh. 19.19 - Prob. 19.27PCh. 19.20A - Addition of one equivalent of ammonia to...Ch. 19.20A - Prob. 19.29PCh. 19.20B - Show how you would accomplish the following...Ch. 19.20C - Prob. 19.31PCh. 19 - For each compound, 1. classify the...Ch. 19 - Prob. 19.33SPCh. 19 - Within each structure, rank the indicated...Ch. 19 - In each pair of compounds, select the stronger...Ch. 19 - Which of the following compounds are capable of...Ch. 19 - Complete the following proposed acid-base...Ch. 19 - Predict the products of the following reactions:...Ch. 19 - Prob. 19.39SPCh. 19 - Show how m-toluidine can be converted to the...Ch. 19 - The mass spectrum of tert-butylamine follows shows...Ch. 19 - Prob. 19.42SPCh. 19 - The following drugs are synthesized using the...Ch. 19 - Prob. 19.44SPCh. 19 - Synthesize from benzene. (Hint: All of these...Ch. 19 - Propose mechanisms for the following reactions.Ch. 19 - Prob. 19.47SPCh. 19 - Prob. 19.48SPCh. 19 - Prob. 19.49SPCh. 19 - Show how you can synthesize the following...Ch. 19 - Prob. 19.51SPCh. 19 - The alkaloid coniine has been isolated from...Ch. 19 - A chemist is summoned to an abandoned...Ch. 19 - Pyrrole undergoes electrophilic aromatic...Ch. 19 - Prob. 19.55SPCh. 19 - Prob. 19.56SPCh. 19 - An unknown compound shows a weak molecular ion at...Ch. 19 - A compound of formula C11H16N2 gives the IR,...Ch. 19 - (A true story.) A drug user responded to an ad...Ch. 19 - Prob. 19.60SPCh. 19 - Prob. 19.61SPCh. 19 - Prob. 19.62SPCh. 19 - Prob. 19.63SPCh. 19 - Prob. 19.64SPCh. 19 - Prob. 19.65SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The relative fitnesses of three genotypes are WA/A= 1.0, WA/a = 0.7, and Wa/a = 0.3. If the population starts at the allele frequency p = 0.5, what is the value of p in the next generation? (3 pts) 12pt v Paragraph V BIU ALarrow_forwardIdentify the most acidic proton in the compound: a d b Оа Ob Ос ○ darrow_forwardA Standard Reference Material is certified to contain 94.6 ppm of an organic contaminant in soil. Your analysis gives values of 98.6, 98.4, 97.2, 94.6, and 96.2. Do your results differ from the expected results at the 95% confidence interval?arrow_forward
- The percentage of an additive in gasoline was measured six times with the following results: 0.13, 0.12, 0.16, 0.17, 0.20, and 0.11%. Find the 95% confidence interval for the percentage of additive.arrow_forwardExplain why this data led Rayleigh to look for and to discover Ar.arrow_forward5) Confidence interval. Berglund and Wichardt investigated the quantitative determination of Cr in high-alloy steels using a potentiometric titration of Cr(VI). Before the titration, samples of the steel were dissolved in acid and the chromium oxidized to Cr(VI) using peroxydisulfate. Shown here are the results (as %w/w Cr) for the analysis of a reference steel. 16.968, 16.922, 16.840, 16.883, 16.887, 16.977, 16.857, 16.728 Calculate the mean, the standard deviation, and the 95% confidence interval about the mean. What does this confidence interval mean?arrow_forward
- In the Nitrous Acid Test for Amines, what is the observable result for primary amines? Group of answer choices nitrogen gas bubbles form a soluble nitrite salt yellow oily layer of nitrosoaminearrow_forward3. a. Use the MS to propose at least two possible molecular formulas. For an unknown compound: 101. 27.0 29.0 41.0 50.0 52.0 55.0 57.0 100 57.5 58.0 58.5 62.0 63.0 64.0 65.0 74.0 40 75.0 76.0 20 20 40 60 80 100 120 140 160 180 200 220 m/z 99.5 68564810898409581251883040 115.0 116.0 77404799 17417M 117.0 12.9 118.0 33.5 119.0 36 133 0 1.2 157.0 2.1 159.0 16 169.0 219 170.0 17 171.0 21.6 172.0 17 181.0 1.3 183.0 197.0 100.0 198.0 200. 784 Relative Intensity 2 2 8 ō (ppm) 6 2arrow_forwardSolve the structure and assign each of the following spectra (IR and C-NMR)arrow_forward
- 1. For an unknown compound with a molecular formula of C8H100: a. What is the DU? (show your work) b. Solve the structure and assign each of the following spectra. 8 6 2 ō (ppm) 4 2 0 200 150 100 50 ō (ppm) LOD D 4000 3000 2000 1500 1000 500 HAVENUMBERI -11arrow_forward16. The proton NMR spectral information shown in this problem is for a compound with formula CioH,N. Expansions are shown for the region from 8.7 to 7.0 ppm. The normal carbon-13 spec- tral results, including DEPT-135 and DEPT-90 results, are tabulated: 7 J Normal Carbon DEPT-135 DEPT-90 19 ppm Positive No peak 122 Positive Positive cus и 124 Positive Positive 126 Positive Positive 128 No peak No peak 4° 129 Positive Positive 130 Positive Positive (144 No peak No peak 148 No peak No peak 150 Positive Positive してしarrow_forward3. Propose a synthesis for the following transformation. Do not draw an arrow-pushing mechanism below, but make sure to draw the product of each proposed step (3 points). + En CN CNarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY