
University Physics with Modern Physics (14th Edition)
14th Edition
ISBN: 9780321973610
Author: Hugh D. Young, Roger A. Freedman
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 19, Problem 19.44P
(a) Onc-third of a mole of He gas is taken along the path abc shown in Fig. P19.44. Assume that the gas may be treated as ideal. How much
Figure P19.44
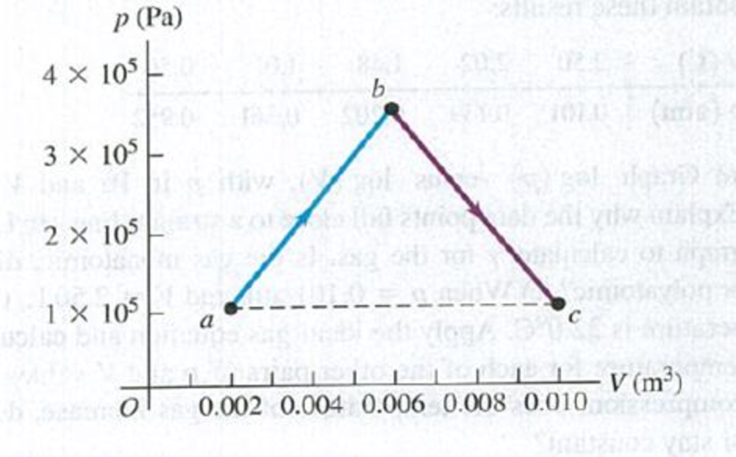
along the horizontal dashed line in Fig. P19.44, how much heat would be transferred into or out of the gas? (c) How does Q in part (b) compare with Q in part (a)? Explain.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
A hollow container is filled with an ideal gas. The container is designed to maintain a constant pressure by allowing gas to enter or leave as needed. At all times the gas &
container are in thermal equilibrium. Initially the temperature is 2350 K. Then the temperature decreases to 500 K and the volume decreases to 0.81 times the initial volume.
Determine the coefficient of volume expansion for the container and the ratio of the final number of moles to the initial number of moles.
Bcontainer
nf
ni
=
=
K-1
How do I answer this question? Also, I don’t really understand the graph
20. The following figure shows a pV-diagram for 0.0040
mol of ideal H, gas. The temperature of the gas does
not change during segment bc.
(a) What volume does this gas occupy at point c'?
(b) Find the temperature of the gas at points a, b, and
с.
(c) How much heat went into or out of the gas during
segments ab, ca, and bc? For (c), indicate whether the
heat has gone into or out of the gas.
p (atm)
b.
2.0
0.50
a
V (L)
0.20
Chapter 19 Solutions
University Physics with Modern Physics (14th Edition)
Ch. 19.1 - In Example 17.7 (Section 17.6), what is the sign...Ch. 19.2 - A quantity of ideal gas undergoes an expansion...Ch. 19.3 - The system described in Fig. 19.7a undergoes four...Ch. 19.4 - Rank the following thermodynamic processes...Ch. 19.5 - Which of the processes in Fig. 19.7 are isochoric?...Ch. 19.6 - Prob. 19.6TYUCh. 19.7 - You want to cool a storage cylinder containing 10...Ch. 19.8 - You have four samples of ideal gas, each of which...Ch. 19 - For the following processes, is the work done by...Ch. 19 - Prob. 19.2DQ
Ch. 19 - In which situation must you do more work:...Ch. 19 - Prob. 19.4DQCh. 19 - Discuss the application of the first law of...Ch. 19 - When ice melts at 0C, its volume decreases. Is the...Ch. 19 - You hold an inflated balloon over a hot-air vent...Ch. 19 - You bake chocolate chip cookies and put them,...Ch. 19 - Imagine a gas made up entirely of negatively...Ch. 19 - In an adiabatic process for an ideal gas, the...Ch. 19 - When you blow on the back of your hand with your...Ch. 19 - An ideal gas expands while the pressure is kept...Ch. 19 - A liquid is irregularly stirred in a...Ch. 19 - When you use a hand pump to inflate the tires of...Ch. 19 - In the carburetor of an aircraft or automobile...Ch. 19 - On a sunny day, large bubbles of air form on the...Ch. 19 - The prevailing winds on the Hawaiian island of...Ch. 19 - Prob. 19.18DQCh. 19 - In a constant-volume process, dU = nCV dT. But in...Ch. 19 - When a gas surrounded by air is compressed...Ch. 19 - When a gas expands adiabatically, it does work on...Ch. 19 - Prob. 19.22DQCh. 19 - A system is taken from state a to state b along...Ch. 19 - A thermodynamic system undergoes a cyclic process...Ch. 19 - Two moles of an ideal gas are heated at constant...Ch. 19 - Six moles of an ideal gas are in a cylinder fitted...Ch. 19 - Prob. 19.3ECh. 19 - BIO Work Done by the Lungs. The graph in Fig....Ch. 19 - CALC During the time 0.305 mol of an ideal gas...Ch. 19 - A gas undergoes two processes. In the first, the...Ch. 19 - Work Done in a Cyclic Process. (a) In Fig. 19.7a,...Ch. 19 - Figure E19.8 shows a pV-diagram for an ideal gas...Ch. 19 - A gas in a cylinder expands from a volume of 0.110...Ch. 19 - Five moles of an ideal monatomic gas with an...Ch. 19 - The process abc shown in the pV-diagram in Fig....Ch. 19 - A gas in a cylinder is held at a constant pressure...Ch. 19 - The pV-diagram in Fig. E19.13 shows a process abc...Ch. 19 - Boiling Water at High Pressure. When water is...Ch. 19 - An ideal gas is taken from a to b on the...Ch. 19 - During an isothermal compression of an ideal gas,...Ch. 19 - A cylinder contains 0.250 mol of carbon dioxide...Ch. 19 - A cylinder contains 0.0100 mol of helium at T =...Ch. 19 - In an experiment to simulate conditions inside an...Ch. 19 - When a quantity of monatomic ideal gas expands at...Ch. 19 - Heat Q flows into a monatomic ideal gas, and the...Ch. 19 - Three moles of an ideal monatomic gas expands at a...Ch. 19 - An experimenter adds 970 J of heat to 1.75 mol of...Ch. 19 - Propane gas (C3Hg) behaves like an ideal gas with ...Ch. 19 - CALC The temperature of 0.150 mol of an ideal gas...Ch. 19 - Five moles of monatomic ideal gas have initial...Ch. 19 - A monatomic ideal gas that is initially at 1.50 ...Ch. 19 - The engine of a Ferrari F355 F1 sports car takes...Ch. 19 - During an adiabatic expansion the temperature of...Ch. 19 - A player bounces a basketball on the floor,...Ch. 19 - On a warm summer day, a large mass of air...Ch. 19 - A cylinder contains 0.100 mol of an ideal...Ch. 19 - A quantity of air is taken from state a to state b...Ch. 19 - One-half mole of an ideal gas is taken from state...Ch. 19 - Figure P19.35 shows the pV-diagram for a process...Ch. 19 - The graph in Fig. P19.36 shows a pV-diagram for...Ch. 19 - When a system is taken from state a to state b in...Ch. 19 - A thermodynamic system is taken from state a to...Ch. 19 - A volume of air (assumed to be an ideal gas) is...Ch. 19 - Three moles of argon gas (assumed to be an ideal...Ch. 19 - Two moles of an ideal monatomic gas go through the...Ch. 19 - Three moles of an ideal gas are taken around cycle...Ch. 19 - Figure P19.43 shows a pV-diagram for 0.0040 mol of...Ch. 19 - (a) Onc-third of a mole of He gas is taken along...Ch. 19 - Starting with 2.50 mol of N2 gas (assumed to be...Ch. 19 - Nitrogen gas in an expandable container is cooled...Ch. 19 - CALC A cylinder with a frictionless, movable...Ch. 19 - CP A Thermodynamic Process in a Solid. A cube of...Ch. 19 - Prob. 19.49PCh. 19 - High-Altitude Research. A large research balloon...Ch. 19 - An air pump has a cylinder 0.250 m long with a...Ch. 19 - A certain ideal gas has molar heat capacity at...Ch. 19 - A monatomic ideal gas expands slowly to twice its...Ch. 19 - CALC A cylinder with a piston contains 0.250 mol...Ch. 19 - Use the conditions and processes of Problem 19.54...Ch. 19 - CALC A cylinder with a piston contains 0.150 mol...Ch. 19 - Use the conditions and processes of Problem 19.56...Ch. 19 - Comparing Thermodynamic Processes. In a cylinder,...Ch. 19 - DATA You have recorded measurements of the heat...Ch. 19 - DATA You compress a gas in an insulated cylinderno...Ch. 19 - DATA You place a quantity of gas into a metal...Ch. 19 - Prob. 19.62CPCh. 19 - BIO ANESTHETIC GASES. One type of gas mixture used...Ch. 19 - BIO ANESTHETIC GASES. One type of gas mixture used...Ch. 19 - BIO ANESTHETIC GASES. One type of gas mixture used...Ch. 19 - BIO ANESTHETIC GASES. One type of gas mixture used...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Description of Motion: Move toward the detector with decreasing speed, then just as you have come to rest, move...
Tutorials in Introductory Physics
3. What is free-fall, and why does it make you weightless? Briefly describe why astronauts are weightless in th...
The Cosmic Perspective
(a) A daredevil is attempting to jump his motorcycle over a line of buses parked end to end by driving up a 32°...
College Physics
67. Why does the gravitational attraction between the Sun and Mercury vary?
Conceptual Physical Science (6th Edition)
sum of the coefficient on the reactant side of a balance equation doesn’t have to equal the sum of the coeffici...
Glencoe Physical Science 2012 Student Edition (Glencoe Science) (McGraw-Hill Education)
Due to the light absorbed by Earth’s surface that was emitted by Earth’s atmosphere, is Earth’s temperature nea...
Lecture- Tutorials for Introductory Astronomy
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- A cylinder is closed at both ends and has insulating EZZ3 walls. It is divided into two compartments by an insulating piston that is perpendicular to the axis of the cylinder as shown in Figure P21.75a. Each compartment contains 1.00 mol of oxygen that behaves as an ideal gas with = 1.40. Initially, the two compartments haw equal volumes and their temperatures are 550 K and 250 K. The piston is then allowed to move slowly parallel to the axis of the cylinder until it comes to rest at an equilibrium position (Fig. P2l.75b). Find the final temperatures in the two compartments.arrow_forwardA sample of a monatomic ideal gas occupies 5.00 L at atmospheric pressure and 300 K (point A in Fig. P21.65). It is warmed at constant volume to 3.00 atm (point B). Then it is allowed to expand isothermally to 1.00 atm (point C) and at last compressed isobarically to its original state, (a) Find the number of moles in the sample. Find (b) the temperature at point B, (c) the temperature at point C, and (d) the volume at point C. (e) Now consider the processes A B, B C, and C A. Describe how to carry out each process experimentally, (f) Find Q, W, and Eint for each of the processes, (g) For the whole cycle A B C A, find Q, W, and Eint.arrow_forwardIn Figure P17.32, the change in internal energy of a gas that is taken from A to C along the blue path is +800 J. The work done on the gas along the red path ABC is 500 J. (a) How much energy must be added to the system by heat as it goes from A through B to C? (b) If the pressure at point A is five times that of point C, what is the work done on the system in going from C to D? (c) What is the energy exchanged with the surroundings by heat as the gas goes from C to A along the green path? (d) If the change in internal energy in going from point D to point A is +500 J, how much energy must be added to the system by heat as it goes from point C to point D? Figure P17.32arrow_forward
- Figure P21.45 shows a cyclic process ABCDA for 1.00 mol of an ideal gas. The gas is initially at Pi = 1.50 105 Pa, Vi = 1.00 103 m3 (point A in Fig. P21.45). a. What is the net work done on the gas during the cycle? b. What is the net amount of energy added by heat to this gas during the cycle? FIGURE P21.45arrow_forwardA gas in a cylindrical closed container is adiabatically and quasi-statically expanded from a state A (3 MPa, 2 L) to a state B with volume of 6 L along the path 1.8pV= constant. (a) Plot the path in the pV plane. (b) Find the amount of work done by the gas and the change in the internal energy of the gas during the process.arrow_forwarda) At what temperature do oxygen molecules have the same average speed as helium atoms ( M=4.00 g/mol) have at 300 K? b) What is answer to the same question about most probable speeds? c) What is the answer to the same question about rms speeds?arrow_forward
- A 2.00-mol sample of a diatomic ideal gas expands slowly and adiabatically from a pressure of 5.00 atm and a volume of 12.0 L to a final volume of 30.0 L. (a) What is the final pressure of the gas? (b) What are the initial and final temperatures? Find (c) Q, (d) Eint, and (e) W for the gas during this process.arrow_forwardOne cylinder contains helium gas and another contains krypton gas at the same temperature. Mark each of these statements true, false, or impossible to determine from the given information. (a) The rms speeds of atoms in the two gases are the same. (b) The average kinetic energies of atoms in the two gases are the same. (c) The internal energies of 1 mole of gas in each cylinder are the same. (d) The pressures in the two cylinders ale the same.arrow_forward(a) Determine the work done on a gas that expands from i to f as indicated in Figure P19.16. (b) What If? How much work is done on the gas if it is compressed from f to i along the same path? Figure P19.16arrow_forward
- Suppose 26.0 g of neon gas are stored in a tank at a temperature of 152C. (a) What is the temperature of the gas on the Kelvin scale? (See Section 10.2.) (b) How many moles of gas are in the tank? (See Section 10.4.) (c) What is the internal energy of the gas? (See Section 10.5.)arrow_forwardA sample of a monatomic ideal gas occupies 5.00 L at atmospheric pressure and 300 K (point A in Fig. P17.68). It is warmed at constant volume to 3.00 atm (point B). Then it is allowed to expand isothermally to 1.00 atm (point C) and at last compressed isobarically to its original state. (a) Find the number of moles in the sample. Find (b) the temperature at point B, (c) the temperature at point C, and (d) the volume at point C. (e) Now consider the processes A B, B C, and C A. Describe how to carry out each process experimentally. (f) Find Q, W, and Eint for each of the processes. (g) For the whole cycle A B C A, find Q, W, and Eint. Figure P17.68arrow_forwardA tank contains 111.0 g chlorine gas l2), which is at temperature 82.0 and absolute pressure 5.70105 Pa. The temperature of the air outside the tank is 20.0 . The molar mass of Cl2 is 70.9 g/mol. (a) What is the volume of the tank? (b) What is the internal energy of the gas? (c) What is the work done by the gas if the temperature and pressure inside the tank drop to 31.0 and 3.80105 Pa, respectively, due to a leak?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning

Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

Physics for Scientists and Engineers, Technology ...
Physics
ISBN:9781305116399
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers
Physics
ISBN:9781337553278
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers with Modern ...
Physics
ISBN:9781337553292
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

College Physics
Physics
ISBN:9781285737027
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning
Thermodynamics: Crash Course Physics #23; Author: Crash Course;https://www.youtube.com/watch?v=4i1MUWJoI0U;License: Standard YouTube License, CC-BY