
Concept explainers
GO Figure 18-46 shows the cross section of a wall made of three layers. The layer thicknesses are L1, L2 = 0.700L1, and L3 = 0.350L1. The thermal conductivities are k1, k2 = 0.900 k1, and k3 = 0.800 k1. The temperatures at the left side and right side of the wall are TH = 30.0°C and TC = −15.0°C, respectively. Thermal
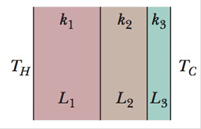
Figure 18-56 Problem 60.
Trending nowThis is a popular solution!

Chapter 18 Solutions
FUNDAMENTALS OF PHYSICS
Additional Science Textbook Solutions
Cosmic Perspective Fundamentals
Campbell Biology in Focus (2nd Edition)
Introductory Chemistry (6th Edition)
Biology: Life on Earth with Physiology (11th Edition)
College Physics: A Strategic Approach (3rd Edition)
The Cosmic Perspective (8th Edition)
- Pure membranes of dipalmitoyl lecithin phospholipids are models of biological membranes. They melt = 41°C. Reversible melting experiments indicate that at Tm AHm=37.7 kJ mol-1. Calculate: A. The entropy of melting, ASm- B. The Gibbs free energy of melting, AGm- C. Does the membrane become more or less ordered upon melting? D. There are 32 rotatable CH2 CH2 bonds in each molecule that can rotate more freely if the membrane melts. What is the increase in multiplicity on melting a mole of bonds?arrow_forward5. Heat capacity often has a temperature dependence for real molecules, particularly if you go over a large temperature range. The heat capacity for liquid n-butane can be fit to the equation Cp(T) = a + bT where a = 100 J K₁₁ mol¹ and b = 0.1067 J K² mol¹ from its freezing point (T = 140 K) to its boiling point (T₁ = 270 K). A. Compute AH for heating butane from 170 K to 270 K. B. Compute AS for the same temperature range.arrow_forward4. How much energy must be transferred as heat to cause the quasi-static isothermal expansion of one mole of an ideal gas at 300 K from PA = 1 bar to PB = 0.5 bar? A. What is VA? B. What is VB? C. What is AU for the process? D. What is AH for the process? E. What is AS for the process?arrow_forward
- 1. The diagram shows the tube used in the Thomson experiment. a. State the KE of the electrons. b. Draw the path of the electron beam in the gravitational field of the earth. C. If the electric field directed upwards, deduce the direction of the magnetic field so it would be possible to balance the forces. electron gun 1KVarrow_forwardas a hiker in glacier national park, you need to keep the bears from getting at your food supply. You find a campground that is near an outcropping of ice. Part of the outcropping forms a feta=51.5* slopeup that leads to a verticle cliff. You decide that this is an idea place to hang your food supply out of bear reach. You put all of your food into a burlap sack, tie a rope to the sack, and then tie a bag full of rocks to the other end of the rope to act as an anchor. You currently have 18.5 kg of food left for the rest of your trip, so you put 18.5 kg of rocks in the anchor bag to balance it out. what happens when you lower the food bag over the edge and let go of the anchor bag? Determine the acceleration magnitude a of the two-bag system when you let go of the anchor bag?arrow_forward2. A thin Nichrome wire is used in an experiment to test Ohm's law using a power supply ranging from 0 to 12 V in steps of 2 V. Why isn't the graph of I vs V linear? 1. Nichrome wire does obey Ohm's law. Explain how that can that be true given the results abovearrow_forward
- 1. The average KE and temperature in Kelvin of the molecules of a gas are related by the equation KE = 3/2 KT where k is the Boltzmann constant 1.38 x 10 m² kg s². The diagram shows the energy levels for a Hydrogen atom. Energy/eV 0.00 -1.51 3.39 13.58 Use this information to show that Hydrogen at room temperature will not emit light. 2. When hydrogen burns in oxygen 241.8 kJ of energy are released per mole. Show that this reaction can produce light.arrow_forward3. By using the fact that around any closed loop the sum of the EMFS = the sum of the PDs. Write equations for the two loops shown in the cct below. 40 ΔΩ I₂ 4V (loop1 20 (loop2) 2v I+12 Use these equations to show that the current flowing through the 20 resistor is 0.75Aarrow_forward5. A potential divider circuit is made by stretching a 1 m long wire with a resistance of 0.1 per cm from A to B as shown. 8V A 100cm B sliding contact 5Ω A varying PD is achieved across the 5 Q resistor by moving the slider along the resistance wire. Calculate the distance from A when the PD across the 5 Q resistor is 6 V.arrow_forward
- 4. A voltmeter with resistance 10 kQ is used to measure the pd across the 1 kQ resistor in the circuit below. 6V 5ΚΩ 1ΚΩ V Calculate the percentage difference between the value with and without the voltmeter.arrow_forward1. A 9V battery with internal resistance 5 2 is connected to a 100 2 resistor. Calculate: a. the Power dissipated in the 100 2 resistor b. The heat generated per second inside the battery. C. The rate of converting chemical to electrical energy by the battery. 2. A 230 V kettle is rated at 1800 W. Calculate the resistance of the heating element.arrow_forward2. If each of the resistors in the circuit below has resistance R show that the total resistance between A and B is 5R/11 A Barrow_forward
 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning





