
Organic Chemistry Plus Masteringchemistry With Pearson Etext, Global Edition
9th Edition
ISBN: 9781292151229
Author: Wade, LeRoy G.
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 17.9, Problem 17.13P
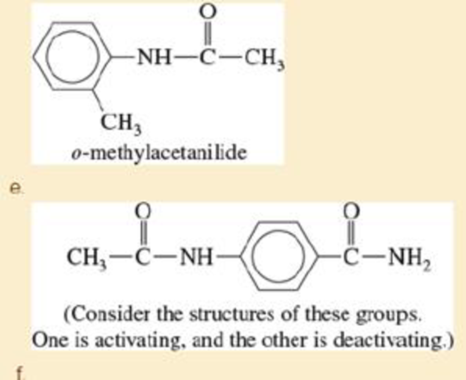
Predict the mononitration products of the following
- a. p-methylanisole
- b. m-nitrochlorobenzene
- c. p-chlorophenol
- d. m-nitroanisole
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
At 25°C, the reaction Zn2+ + 2e ⇄ Zn has a normal equilibrium potential versus the saturated calomel electrode of -1.0048 V. Determine the normal equilibrium potential of Zn versus the hydrogen electrode.Data: The calomel electrode potential is E° = 0.2420 V versus the normal hydrogen electrode.
Electrochemistry. State the difference between E and E0.
In an electrolytic cell, the positive pole is always assumed to be on the right side of the battery notation. Is that correct?
Chapter 17 Solutions
Organic Chemistry Plus Masteringchemistry With Pearson Etext, Global Edition
Ch. 17.1 - Prob. 17.1PCh. 17.2 - Prob. 17.2PCh. 17.3 - Prob. 17.3PCh. 17.4 - Use resonance forms to show that the dipolar sigma...Ch. 17.6A - Prob. 17.5PCh. 17.6A - Prob. 17.6PCh. 17.6B - Propose a mechanism for the brommation of...Ch. 17.6B - Prob. 17.8PCh. 17.6B - Prob. 17.9PCh. 17.7 - Prob. 17.10P
Ch. 17.8 - Draw all the resonance forms of the sigma complex...Ch. 17.9 - Predict the mononitration products of the...Ch. 17.9 - Predict the mononitration products of the...Ch. 17.9 - Prob. 17.14PCh. 17.10 - Propose products (if any) and mechanisms for the...Ch. 17.10 - Predict the products (if any) of the following...Ch. 17.10 - Which reactions will produce the desired product...Ch. 17.10 - Prob. 17.19PCh. 17.11C - Prob. 17.20PCh. 17.12A - Prob. 17.21PCh. 17.12B - Propose a mechanism that shows why p-chlorotoluene...Ch. 17.12B - Propose mechanisms and show the expected products...Ch. 17.12B - Prob. 17.24PCh. 17.13A - What products would you expect from the following...Ch. 17.13A - What organocuprate reagent would you use for the...Ch. 17.13B - What products would you expect from the following...Ch. 17.13B - Prob. 17.28PCh. 17.13C - What products would you expect from the following...Ch. 17.13C - Prob. 17.30PCh. 17.14C - Prob. 17.31PCh. 17.14C - Predict the major products of the following...Ch. 17.15A - Predict the major products of treating the...Ch. 17.15B - Prob. 17.34PCh. 17.15B - Prob. 17.35PCh. 17.15B - Predict the major products when the following...Ch. 17.15C - Prob. 17.37PCh. 17.15C - a. Based on what you know about the relative...Ch. 17.15C - Show how you would synthesize the following...Ch. 17.16A - The bombardier beetle defends itself by spraying a...Ch. 17.16B - Predict the products formed when m-cresol...Ch. 17.16B - Prob. 17.42PCh. 17.16B - Prob. 17.43PCh. 17.16B - Predict the site(s) of electophilic attack on...Ch. 17.16B - Prob. 17.45PCh. 17.16B - Prob. 17.46PCh. 17.16B - Propose a synthetic sequence of this...Ch. 17.16B - Prob. 17.48PCh. 17.16B - Starting from toluene, propose a synthesis of this...Ch. 17 - Prob. 17.50SPCh. 17 - Prob. 17.51SPCh. 17 - Show how you would synthesize the following...Ch. 17 - Predict the major products of the following...Ch. 17 - Predict the major products of bromination of the...Ch. 17 - What products would you expect from the following...Ch. 17 - Prob. 17.56SPCh. 17 - Prob. 17.57SPCh. 17 - The following compound reacts with a hot,...Ch. 17 - Prob. 17.59SPCh. 17 - Electrophilic aromatic substitution usually occurs...Ch. 17 - Prob. 17.62SPCh. 17 - The most common selective herbicide for killing...Ch. 17 - Furan undergoes electrophilic aromatic...Ch. 17 - Prob. 17.65SPCh. 17 - Bisphenol A is an important component of many...Ch. 17 - Prob. 17.67SPCh. 17 - Prob. 17.68SPCh. 17 - Prob. 17.69SPCh. 17 - In Chapter14, we saw that Agent Orange contains...Ch. 17 - Phenol reacts with three equivalents of bromine in...Ch. 17 - Prob. 17.72SPCh. 17 - Prob. 17.73SPCh. 17 - A common illicit synthesis of methamphetamine...Ch. 17 - Prob. 17.75SPCh. 17 - Prob. 17.76SPCh. 17 - Prob. 17.77SPCh. 17 - Prob. 17.78SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- In an electrolytic cell, the positive pole is always assumed to be on the right side of the battery. Is that correct?arrow_forwardCalculate the free energy of formation of 1 mol of Cu in cells where the electrolyte is 1 mol dm-3 Cu2+ in sulfate solution, pH 0. E° for the Cu2+/Cu pair in this medium is +142 mV versus ENH.Assume the anodic reaction is oxygen evolution.Data: EH2 = -0.059 pH (V) and EO2 = 1.230 - 0.059 pH (V); 2.3RT/F = 0.059 Varrow_forwardIf the normal potential for the Fe(III)/Fe(II) pair in acid at zero pH is 524 mV Hg/Hg2Cl2 . The potential of the saturated calomel reference electrode is +246 mV versus the NHE. Calculate E0 vs NHE.arrow_forward
- Given the galvanic cell whose scheme is: (-) Zn/Zn2+ ⋮⋮ Ag+/Ag (+). If we know the normal potentials E°(Zn2+/Zn) = -0.76V and E°(Ag+/Ag) = 0.799 V. Indicate the electrodes that are the anode and the cathode and calculate the E0battery.arrow_forwardIndicate the functions that salt bridges have in batteries.arrow_forwardIn the battery:Pt | H2 (g) | H+ (aq) | Fe2+ (aq) | FeIndicate the cathode and anode.arrow_forward
- Write the equations that occur when the electrode Pb (s) | PbI2 (s) | KI (ac) in a galvanic cell. a) It functions as a positive electrode b) It functions as a negative electrode c) What is the ion with respect to which this electrode is reversible?arrow_forwardState the formula to find the electromotive force of a battery as a function of the potential of the anode and the cathode.arrow_forwardWhy are normal electrode potentials also called relative electrode potentials?arrow_forward
- Easily differentiate between electrochemical potential and Galvani potential.arrow_forwardConstruct a molecular orbital diagram for carbon monoxide. Identify the relevant point group,include all of the appropriate symmetry labels and pictures, and fill in the electrons. Make sure toaccount for the difference in electronegativity between C and O. Hint: CO is substantiallyisoelectronic to N2. (PLEASE DRAW THE ENTIRE MO DIAGRAM!!!)arrow_forwardplease help with hwarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
Seven Name Reactions in One - Palladium Catalysed Reaction (047 - 053); Author: Rasayan Academy - Jagriti Sharma;https://www.youtube.com/watch?v=5HEKTpDFkqI;License: Standard YouTube License, CC-BY