
Concept explainers
Draw the structure for each of the following:
- a. isobutyraldehyde
- b. 4-hexenaI
- c. diisopentyl
ketone - d. 3-methylcyclohexanone
- e. 2.4-pentanedione
- f. 4-bromo-3-heptanone
- g. γ-bromocaproaldehyde
- h. 2-ethylcyclopentanecarbaldehyde
- i. 4-methyl-5-oxohexanal
(a)
Interpretation:
The structure of Isobutyraldehyde has to be drawn.
Answer to Problem 53P
The structure of Isobutyraldehyde is given below:
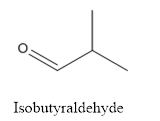
Explanation of Solution
The compound has aldehyde group that is attached to the isobutyl group.
The structure of isobutyraldehyde is as follows.

(b)
Interpretation:
The structure of 4-hexenal has to be drawn.
Answer to Problem 53P
The structure of 4-hexenal is given below:
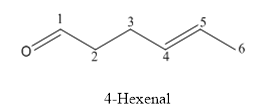
Explanation of Solution
The compound has aldehyde functional group and double bond at the 4th position of the compound.
The structure is as follows.

(c)
Interpretation:
The structure of diisopentyl ketone has to be drawn.
Answer to Problem 53P
The structure of diisopentyl ketone is given below:
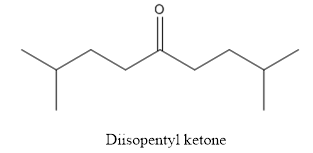
Explanation of Solution
The compound has ketone functional group. Two isopentyl groups are attached to the keto group.
The structure of diisopentyl ketone is as follows.

(d)
Interpretation:
The structure of 3-methylcyclohexanone has to be drawn.
Answer to Problem 53P
The structure of 3-methylcyclohexanoneis given below:
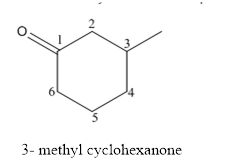
Explanation of Solution
The compound has ketone functional and group and it is called as cyclohexanone and it has one methyl group at 3rd position of the ring.
The structure of 3-methyl cyclohexanone is as follows.

(e)
Interpretation:
The structure of 2,4-pentanedione has to be drawn.
Answer to Problem 53P
The structure of 2,4-pentanedione is given below:
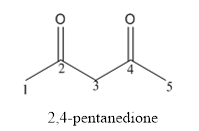
Explanation of Solution
The compound has two keto functional groups and these are attached to the pentane carbon chain.

(f)
Interpretation:
The structure of 4-bromo-3-heptanone has to be drawn.
Answer to Problem 53P
The structure of 4-bromo-3-heptanone is given below:
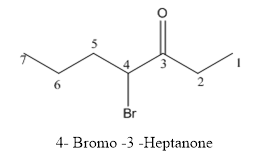
Explanation of Solution
The compound has keto functional group and it is attached to the heptanes carbon chain. At the 4th position of the carbon chain has bromine attachment.

(g)
Interpretation:
The structure of
Answer to Problem 53P
The structure of
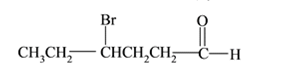
Explanation of Solution
Compound has aldehyde functional group and at the
The structure of
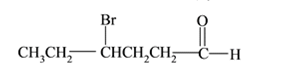
(h)
Interpretation:
The structure of 2-ethylcyclopentanecarbaldehyde has to be drawn.
Answer to Problem 53P
The structure of 2-ethylcyclopentanecarbaldehyde is given below:
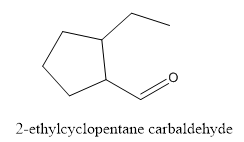
Explanation of Solution
In this compound, pentane ring has ethyl group and aldehyde functional group.
The structure of 2-ethylcyclopentane carbaldehyde.

(i)
Interpretation:
The structure of 4-methyl-5-oxohexanal has to be drawn.
Answer to Problem 53P
The structure of 4-methyl-5-oxohexanal is givenbelow:
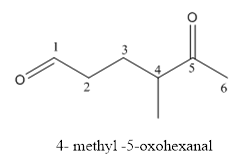
Explanation of Solution
The compound has aldehyde functional group at the 5th position of the carbon chain as keto group it is named as oxo-because it is referred to an attachment.
The structure of 4- methyl-5-oxohexanal.

Want to see more full solutions like this?
Chapter 17 Solutions
Organic Chemistry; Modified MasteringChemistry with Pearson eText -- ValuePack Access Card; Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
- B 1 of 2 Additional problems in preparation to Midterm #1: 1.) How can the following compounds be prepared using Diels-Alder reaction: CH3 O CN (a) (b) CN CH3 2.) What is the missing reagent in the shown reaction? H3C + ? H3C H3C CN H3C ''CN (၁) H 3.) Write the products 1,2-addition and 1,4-addition of DBr to 1,3-cyclohexadiene. Remember, D is deuterium, a heavy isotope of hydrogen. It reacts exactly like hydrogen. 4.) In the shown reaction, which will be the kinetic product and which will be the thermodynamic product? H3C CI H3C HCI H3C + 5.) Which of the following molecules is aromatic? (a) (b) (c) H 6.) Which of the following molecules is aromatic? (a) (b) (c) 7.) Write the mechanism for the shown reaction. + Ха AICI 3 CI 8.) Suggest reagents that would convert benzene into the shown compounds. CI NO2 -8-6-6-8-a (a) (b) (c) (d) (e) (a) SO3H Brarrow_forwardThe number of 2sp^2 hybridized atoms in is: A. 8; B. 6; C.4; D.2; E.0;arrow_forwardThe highest boiling compound from among the following isA. 2-methylheptane; B. 3-methylheptane; C. 2,2-dimethylhexane;D. octane; E. 2,2,3-trimethylpentanearrow_forward
- Which of the following features are found in the most stable structure ofCH5NO that does not have a CO bond?w. a π bond, x. two NH bonds, y. one OH bond, z. 3 lone pairsA. w, x; B. x, y; C. y, z; D. x, y, z; E. all of them.arrow_forwardWhich one of the following functional groups is not present in thecompound shownA. amine; B. aldehyde, C. ether; D. amide. E. ketonearrow_forwardWhich of the following formulas correspond to at least one compound inwhich resonance is important?w. C2H5N x. C3H5Br; y. C3H4; z. C4H6.A. w, x, y; B. x, y, z; C. w, x, z; D. w, y, z; E. all of themarrow_forward
- Predict the product(s) that are formed after each step for reactions 1-4. In each case, consider formation of any chiral center(s) and draw all expected stereoisomers. 1) OH 1) HBr (SN2) 2) NaOH, heat 3) BH3, THF 4) H2O2, NaOH 2) OH 1) SOCI 2, py 2) NaOEt 3) Br2, H₂O 3) OH 1) H2SO4 conc. 2) HBr, ROOR 3) KOtBu 4) OH 1) TsCl, py 2) NaOEt 3) 03 4) DMSarrow_forwardWhich of the following rings has the least strain in its most stableconformation?A. Cyclobutane; B. Cyclopentane; C. Cyclohexane; D. Cycloheptane;E. Cyclooctanearrow_forwardThe number of different carbon skeletons that have a main chain of 9carbons and an ethyl branch isA 3; B. 4; C. 5; D. 6; E. 7arrow_forward
- Q5: Classify the following pair of molecules as constitutional isomers, enantiomers, diastereomers, the same molecule, or completely different molecules. Br O CI Br OH OH 111 Br .!!!/Br F OH and ...m Br Br OH CI Br OH ་་་་་" ། ་arrow_forwardConsidering only rotation around the 1-2 bond, how many differentstaggered conformations are there of 1,2-dibromo-1,2-dichloropropane?A: 2; B. 3; C. 4; D. 6; E. more than 6.arrow_forwardcan you help me solve thisarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





