
Organic Chemistry; Modified MasteringChemistry with Pearson eText -- ValuePack Access Card; Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
7th Edition
ISBN: 9780134240152
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17.13, Problem 47P
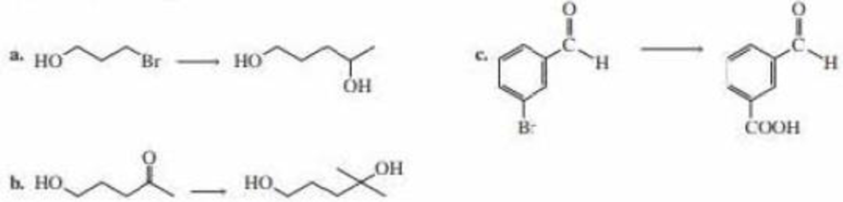
Show how each of the following compounds could be prepared from the given starting material. Each requires a protecting group.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Help w c!
Can someone help me understand this?
help w d!
Chapter 17 Solutions
Organic Chemistry; Modified MasteringChemistry with Pearson eText -- ValuePack Access Card; Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
Ch. 17.1 - Prob. 1PCh. 17.1 - Give two names for each of the following:Ch. 17.1 - Name the following:Ch. 17.2 - Prob. 4PCh. 17.4 - What products are formed when the following...Ch. 17.4 - We saw on the previous page that...Ch. 17.4 - a. How many stereoisomers are obtained from the...Ch. 17.4 - Prob. 9PCh. 17.4 - Write the mechanism for the reaction of acetyl...Ch. 17.4 - Prob. 11P
Ch. 17.4 - Show how the following compounds can be...Ch. 17.5 - Prob. 13PCh. 17.5 - Prob. 14PCh. 17.6 - In the mechanism for cyanohydrin formation, why is...Ch. 17.6 - Prob. 16PCh. 17.6 - Prob. 17PCh. 17.6 - Show two ways to convert an alkyl halide into a...Ch. 17.7 - Prob. 20PCh. 17.7 - Prob. 21PCh. 17.7 - Prob. 22PCh. 17.7 - Prob. 23PCh. 17.8 - Prob. 24PCh. 17.9 - What reducing agents should be used to obtain the...Ch. 17.9 - Prob. 26PCh. 17.9 - Prob. 27PCh. 17.10 - Prob. 28PCh. 17.10 - Prob. 29PCh. 17.10 - Prob. 30PCh. 17.10 - The pKa of protonated acetone is about 7.5. and...Ch. 17.10 - Prob. 32PCh. 17.10 - Prob. 33PCh. 17.10 - Prob. 34PCh. 17.10 - Excess ammonia must be used when a primary amine...Ch. 17.10 - The compounds commonly known as amino acids are...Ch. 17.11 - Hydration of an aldehyde is also catalyzed by...Ch. 17.11 - Which ketone forms the most hydrate in an aqueous...Ch. 17.11 - When trichloroacetaldehyde is dissolved in water,...Ch. 17.12 - Which of the following are a. hermiacetals? b....Ch. 17.12 - Prob. 41PCh. 17.12 - Explain why an acetal can be isolated but most...Ch. 17.13 - Prob. 43PCh. 17.13 - Prob. 44PCh. 17.13 - What products would be formed from the proceedings...Ch. 17.13 - a. In a six-step synthesis, what is the yield of...Ch. 17.13 - Show how each of the following compounds could be...Ch. 17.15 - Prob. 48PCh. 17.17 - Prob. 50PCh. 17.18 - Prob. 51PCh. 17.19 - Prob. 52PCh. 17 - Draw the structure for each of the following: a....Ch. 17 - Prob. 54PCh. 17 - Prob. 55PCh. 17 - a. Show the reagents required to form the primary...Ch. 17 - Prob. 57PCh. 17 - Using cyclohexanone as the starting material,...Ch. 17 - Prob. 59PCh. 17 - 60. Show how each of the following compounds could...Ch. 17 - Fill in the boxes:Ch. 17 - Prob. 62PCh. 17 - Identify A through O:Ch. 17 - Prob. 64PCh. 17 - Prob. 65PCh. 17 - Prob. 66PCh. 17 - How many signals would the product of the...Ch. 17 - Prob. 68PCh. 17 - Prob. 69PCh. 17 - Prob. 70PCh. 17 - Prob. 71PCh. 17 - Prob. 72PCh. 17 - Prob. 73PCh. 17 - Prob. 74PCh. 17 - Prob. 75PCh. 17 - Prob. 76PCh. 17 - Prob. 77PCh. 17 - A compound gives the following IR spectrum. Upon...Ch. 17 - a. Propose a mechanism for the following reaction:...Ch. 17 - Prob. 80PCh. 17 - Prob. 81PCh. 17 - Prob. 82PCh. 17 - Prob. 83PCh. 17 - Prob. 84PCh. 17 - In the presence of an acid catalyst, acetaldehyde...Ch. 17 - Prob. 86PCh. 17 - Prob. 87PCh. 17 - Prob. 88PCh. 17 - A compound reacts with methylmagnesium bromide...Ch. 17 - Show how each of the following compounds can be...Ch. 17 - Prob. 91PCh. 17 - Prob. 92PCh. 17 - The pKa values of the carboxylic acid groups of...Ch. 17 - The Baylis-Hillman reaction is a DABCO...Ch. 17 - Prob. 95PCh. 17 - Prob. 96P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- a. Explain Why electron withdrawing groupe tend to be meta-Directors. Your answer Should lyclude all apropriate. Resonance contributing Structures 6. Explain why -ll is an ortho -pura drccton evon though chlorine has a very High Electronegativityarrow_forwardC. Ν Harrow_forwarda. H3C. N H3C CH3 HCNarrow_forward
- ол 2. восцапан (46:00) Curtius rearrangment 1. NaN3, heat -OHarrow_forwardQuestion 1. Please predict the products for each of the following reactions. Clearly show the regiochemistry (Markovnikov vs anti-Markovnikov) and stereochemistry (syn- vs anti- or both). If a mixture of enantiomers is formed, please draw all the enantiomers.arrow_forwardElectrochemistry. Briefly describe the Donnan potential.arrow_forward
- Indicate what the Luther equation is used for?arrow_forwardIndicate one aspect that benefits and another that makes it difficult to use the hydroquinone electrode to measure pH.arrow_forwardAt an electrified interface according to the Gouy-Chapman model, what types of interactions do NOT occur between the ions and the solvent according to this theory?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY