
Organic Chemistry; Modified MasteringChemistry with Pearson eText -- ValuePack Access Card; Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
7th Edition
ISBN: 9780134240152
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17.1, Problem 3P
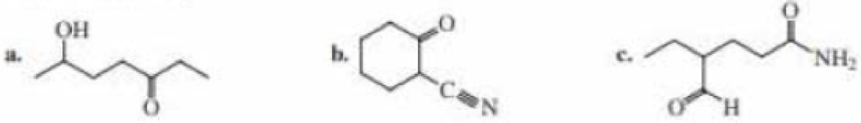
Name the following:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Identify the mechanism through which the following reaction will proceed and draw the major product.
Part 1 of 2
Br
KOH
EtOH
Through which mechanism will the reaction proceed? Select the single best answer.
E1
E2
neither
Part: 1/2
Part 2 of 2
Draw the major product formed as a result of the reaction.
Click and drag to start drawing a
structure.
X
What is single-point calibration? Provide an example.
Draw the major product formed via an E1 pathway.
Chapter 17 Solutions
Organic Chemistry; Modified MasteringChemistry with Pearson eText -- ValuePack Access Card; Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
Ch. 17.1 - Prob. 1PCh. 17.1 - Give two names for each of the following:Ch. 17.1 - Name the following:Ch. 17.2 - Prob. 4PCh. 17.4 - What products are formed when the following...Ch. 17.4 - We saw on the previous page that...Ch. 17.4 - a. How many stereoisomers are obtained from the...Ch. 17.4 - Prob. 9PCh. 17.4 - Write the mechanism for the reaction of acetyl...Ch. 17.4 - Prob. 11P
Ch. 17.4 - Show how the following compounds can be...Ch. 17.5 - Prob. 13PCh. 17.5 - Prob. 14PCh. 17.6 - In the mechanism for cyanohydrin formation, why is...Ch. 17.6 - Prob. 16PCh. 17.6 - Prob. 17PCh. 17.6 - Show two ways to convert an alkyl halide into a...Ch. 17.7 - Prob. 20PCh. 17.7 - Prob. 21PCh. 17.7 - Prob. 22PCh. 17.7 - Prob. 23PCh. 17.8 - Prob. 24PCh. 17.9 - What reducing agents should be used to obtain the...Ch. 17.9 - Prob. 26PCh. 17.9 - Prob. 27PCh. 17.10 - Prob. 28PCh. 17.10 - Prob. 29PCh. 17.10 - Prob. 30PCh. 17.10 - The pKa of protonated acetone is about 7.5. and...Ch. 17.10 - Prob. 32PCh. 17.10 - Prob. 33PCh. 17.10 - Prob. 34PCh. 17.10 - Excess ammonia must be used when a primary amine...Ch. 17.10 - The compounds commonly known as amino acids are...Ch. 17.11 - Hydration of an aldehyde is also catalyzed by...Ch. 17.11 - Which ketone forms the most hydrate in an aqueous...Ch. 17.11 - When trichloroacetaldehyde is dissolved in water,...Ch. 17.12 - Which of the following are a. hermiacetals? b....Ch. 17.12 - Prob. 41PCh. 17.12 - Explain why an acetal can be isolated but most...Ch. 17.13 - Prob. 43PCh. 17.13 - Prob. 44PCh. 17.13 - What products would be formed from the proceedings...Ch. 17.13 - a. In a six-step synthesis, what is the yield of...Ch. 17.13 - Show how each of the following compounds could be...Ch. 17.15 - Prob. 48PCh. 17.17 - Prob. 50PCh. 17.18 - Prob. 51PCh. 17.19 - Prob. 52PCh. 17 - Draw the structure for each of the following: a....Ch. 17 - Prob. 54PCh. 17 - Prob. 55PCh. 17 - a. Show the reagents required to form the primary...Ch. 17 - Prob. 57PCh. 17 - Using cyclohexanone as the starting material,...Ch. 17 - Prob. 59PCh. 17 - 60. Show how each of the following compounds could...Ch. 17 - Fill in the boxes:Ch. 17 - Prob. 62PCh. 17 - Identify A through O:Ch. 17 - Prob. 64PCh. 17 - Prob. 65PCh. 17 - Prob. 66PCh. 17 - How many signals would the product of the...Ch. 17 - Prob. 68PCh. 17 - Prob. 69PCh. 17 - Prob. 70PCh. 17 - Prob. 71PCh. 17 - Prob. 72PCh. 17 - Prob. 73PCh. 17 - Prob. 74PCh. 17 - Prob. 75PCh. 17 - Prob. 76PCh. 17 - Prob. 77PCh. 17 - A compound gives the following IR spectrum. Upon...Ch. 17 - a. Propose a mechanism for the following reaction:...Ch. 17 - Prob. 80PCh. 17 - Prob. 81PCh. 17 - Prob. 82PCh. 17 - Prob. 83PCh. 17 - Prob. 84PCh. 17 - In the presence of an acid catalyst, acetaldehyde...Ch. 17 - Prob. 86PCh. 17 - Prob. 87PCh. 17 - Prob. 88PCh. 17 - A compound reacts with methylmagnesium bromide...Ch. 17 - Show how each of the following compounds can be...Ch. 17 - Prob. 91PCh. 17 - Prob. 92PCh. 17 - The pKa values of the carboxylic acid groups of...Ch. 17 - The Baylis-Hillman reaction is a DABCO...Ch. 17 - Prob. 95PCh. 17 - Prob. 96P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Part 9 of 9 Consider the products for the reaction. Identify the major and minor products. HO Cl The E stereoisomer is the major product and the Z stereoisomer is the minor product ▼ S major product minor productarrow_forwardConsider the reactants below. Answer the following questions about the reaction mechanism and products. HO Clarrow_forwardjulietteyep@gmail.com X YSCU Grades for Juliette L Turner: Orc X 199 A ALEKS - Juliette Turner - Modul X A ALEKS - Juliette Turner - Modul x G butane newman projection - Gox + www-awa.aleks.com/alekscgi/x/Isl.exe/10_u-IgNslkr7j8P3jH-IBxzaplnN4HsoQggFsejpgqKoyrQrB2dKVAN-BcZvcye0LYa6eXZ8d4vVr8Nc1GZqko5mtw-d1MkNcNzzwZsLf2Tu9_V817y?10Bw7QYjlb il Scribbr citation APA SCU email Student Portal | Main Ryker-Learning WCU-PHARM D MySCU YSCU Canvas- SCU Module 4: Homework (Ch 9-10) Question 28 of 30 (1 point) | Question Attempt: 1 of Unlimited H₂SO heat OH The mechanism of this reaction involves two carbocation intermediates, A and B. Part 1 of 2 KHSO 4 rearrangement A heat B H₂O 2 OH Draw the structure of A. Check Search #t m Save For Later Juliet Submit Assignm 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessarrow_forward
- The electrons flow from the electron-rich atoms of the nucleophile to the electrons poor atoms of the alkyl halide. Identify the electron rich in the nucleophile. Enter the element symbol only, do not include any changes.arrow_forwardHello, I am doing a court case analysis in my Analytical Chemistry course. The case is about a dog napping and my role is prosecution of the defendant. I am tasked in the Area of Expertise in Neutron Activation and Isotopic Analysis. Attached is the following case study reading of my area of expertise! The landscaping stone was not particularly distinctive in its decoration but matched both the color and pattern of the Fluential’s landscaping stone as well as the stone in the back of the recovered vehicle. Further analysis of the stone was done using a technique called instrumental neutron activation analysis. (Proceed to Neutron Activation data) Photo Notes: Landscaping stone recovered in vehicle. Stone at Fluential’s home is similar inappearance. Finally, the white paint on the brick was analyzed using stable isotope analysis. The brick recovered at the scene had smeared white paint on it. A couple of pieces of brick in the back of the car had white paint on them. They…arrow_forwardCite the stability criteria of an enamine..arrow_forward
- What would you expect to be the major product obtained from the following reaction? Please explain what is happening here. Provide a detailed explanation and a drawing showing how the reaction occurs. The correct answer to this question is V.arrow_forwardPlease answer the question for the reactions, thank youarrow_forwardWhat is the product of the following reaction? Please include a detailed explanation of what is happening in this question. Include a drawing showing how the reagent is reacting with the catalyst to produce the correct product. The correct answer is IV.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning


Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY