
(a)
Interpretation:
The compounds mesitylene, toluene, and
Concept introduction:
The replacement of hydrogen atom attached to a carbon atom of electron-rich benzene ring by an electrophile is known as electrophilic aromatic substitution reaction. The rate of electrophilic aromatic substitution reaction depends on the substituted group on the aromatic ring. The ring deactivating group retards the electrophilic aromatic substitution reaction and ring activating group enhances the electrophilic aromatic substitution reaction.
Answer to Problem 16.44AP
The compounds mesitylene, toluene, and
Explanation of Solution
The structure of mesitylene, toluene, and
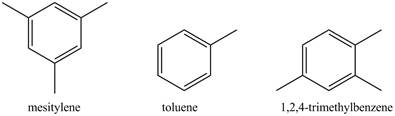
Figure 1
The reaction of any
The methyl group is electron-donating group. It activates the phenyl ring. The toluene has only one methyl group attached to it. Therefore, it will be least reactive towards
The order of reactivity toward nitration reaction is shown below.
The increasing order of reactivity towards toward
(b)
Interpretation:
The compounds chlorobenzene, benzene, and nitrobenzene are to be arranged in increasing order of increasing reactivity toward
Concept introduction:
The replacement of hydrogen atom attached to a carbon atom of electron-rich benzene ring by an electrophile is known as electrophilic aromatic substitution reaction. The rate of electrophilic aromatic substitution reaction depends on the substituted group on the aromatic ring. The ring deactivating group retards the electrophilic aromatic substitution reaction and ring activating group enhances the electrophilic aromatic substitution reaction.
Answer to Problem 16.44AP
The compounds chlorobenzene, benzene, and nitrobenzene are arranged in increasing order of increasing reactivity toward
Explanation of Solution
The structure of chlorobenzene, benzene, and nitrobenzene are shown below.
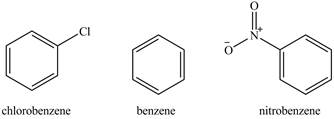
Figure 2
The reaction of any aromatic compound with
The nitro and chloro groups are electron-withdrawing groups. Therefore, the reactivity of the chlorobenzene and nitrobenzene will be less than that of benzene. The nitro group is stronger deactivating group than chloro group. Therefore, the order of reactivity toward nitration reaction is shown below.
The increasing order of reactivity towards toward
(c)
Interpretation:
The compounds
Concept introduction:
The replacement of hydrogen atom attached to a carbon atom of electron-rich benzene ring by an electrophile is known as electrophilic aromatic substitution reaction. The rate of electrophilic aromatic substitution reaction depends on the substituted group on the aromatic ring. The ring deactivating group retards the electrophilic aromatic substitution reaction and ring activating group enhances the electrophilic aromatic substitution reaction.
Answer to Problem 16.44AP
The compounds
Explanation of Solution
The structure of
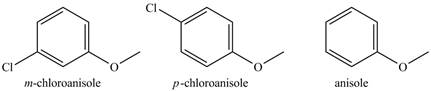
Figure 3
The reaction of any aromatic compound with
The methoxy group and chloro groups are ortho and para directing groups. The methoxy group is electron releasing group and chloro group is electron-withdrawing group.
Therefore, the reactivity of anisole will be highest among the rest of the compound toward
The order of reactivity toward nitration reaction is shown below.
The increasing order of reactivity towards toward
(d)
Interpretation:
The compounds acetophenone,
Concept introduction:
The replacement of hydrogen atom attached to a carbon atom of electron-rich benzene ring by an electrophile is known as electrophilic aromatic substitution reaction. The rate of electrophilic aromatic substitution reaction depends on the substituted group on the aromatic ring. The ring deactivating group retards the electrophilic aromatic substitution reaction and ring activating group enhances the electrophilic aromatic substitution reaction.
Answer to Problem 16.44AP
The compounds acetophenone,
Explanation of Solution
The structure of acetophenone,
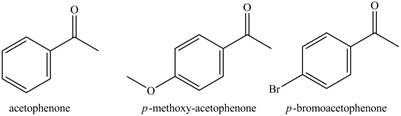
Figure 4
The reaction of any aromatic compound with
The acetyl group and bromo group is electron-withdrawing groups and methoxy group is electron releasing group. Acetophenone has a ring activating group attached on it. Therefore, it is most reactive toward nitration reaction among the rest of the compound. The compound
The increasing order of reactivity towards toward
Want to see more full solutions like this?
Chapter 16 Solutions
ORGANIC CHEM +SG +SAPLING >IP<
- Help me solve this problem.arrow_forwardDraw a mechanism for the following synthetic transformation including reagents and any isolable intermediates throughout the process. Please clearly indicate bond cleavage/formation using curly arrows. MeO2Carrow_forwardCHEM 310 Quiz 8 Organic Chemistry II Due: Tuesday, April 25th, at 11:59 pm. This quiz is open textbook / open notes - but you must work alone. You cannot use the internet or the solutions manual for the book. Scan in your work and record an explanation of your mechanism. You may record this any way that you like. One way would be to start an individual Zoom meeting, start recording, "share your screen" and then talk through the problem. This will be converted to an .mp4 file that you can upload into Canvas using the "record/upload media" feature. Pyridine, benzoic acid and benzene are dissolved in ethyl acetate. Design and provide a plan / flow chart for separating and isolating each of these components. Pyridine and benzene are liquids at room temperature. Benzoic acid is a solid. You have ethyl acetate, 2M NaOH, 2M HCI and anhydrous MgSO4 available, as well as all the glassware and equipment that you used in the organic lab this year. Provide accurate acid/base reactions for any…arrow_forward
- Can anyone help me solve this step by step. Thank you in advaarrow_forwardPlease draw the mechanism for this Friedel-crafts acylation reaction using arrowsarrow_forwardDraw the Fischer projection of D-fructose. Click and drag to start drawing a structure. Skip Part Check AP 14 tv SC F1 F2 80 F3 a F4 ! 2 # 3 CF F5 75 Ax MacBook Air 894 $ 5olo % Λ 6 > W F6 K F7 &arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





